Revolutionizing Ambroxol Hydrochloride Production: A Green Three-Step Synthetic Strategy for Commercial Scale-Up
Introduction to Patent CN111072500B
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for producing essential respiratory medications. Patent CN111072500B, published in December 2022, introduces a groundbreaking preparation method for Ambroxol Hydrochloride, a widely used mucolytic agent. This technology addresses critical bottlenecks in traditional synthesis, such as unstable intermediates and hazardous reagents, by proposing a streamlined three-step process starting from o-nitrobenzaldehyde. The core innovation lies in the strategic protection of the aldehyde group prior to reduction, followed by a novel oxidative bromination system that recycles bromine atoms, and finally, a highly selective reductive amination. This approach not only enhances the overall yield and purity but also aligns with modern green chemistry principles, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming for sustainable production.
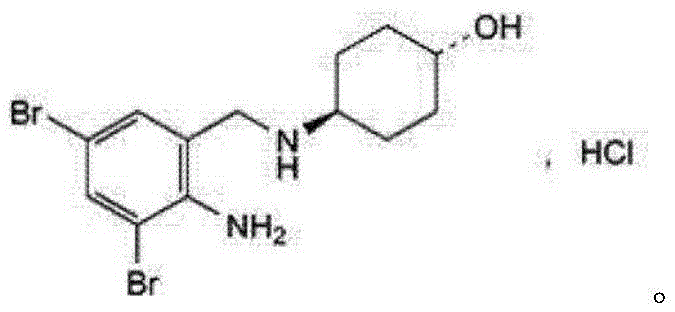
For R&D directors and process chemists, the significance of this patent extends beyond mere yield improvements; it represents a fundamental shift in process safety and scalability. By eliminating the need for isolating unstable Schiff base intermediates and replacing dangerous reducing agents like lithium aluminum hydride with safer catalytic systems, the method drastically reduces operational risks. The detailed reaction conditions provided, including specific mass ratios for catalysts like NiCoB/TiO2 and precise temperature controls, offer a robust framework for technology transfer. As we delve deeper into the technical specifics, it becomes evident that this methodology provides a viable solution for cost reduction in API manufacturing while maintaining the stringent quality standards required for respiratory therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ambroxol Hydrochloride has been plagued by significant technical hurdles that hinder large-scale industrial adoption. Traditional routes, such as those disclosed in earlier literature, often rely on the direct condensation of 2-amino-3,5-dibromobenzaldehyde with trans-4-aminocyclohexanol to form a Schiff base intermediate. As illustrated in prior art reaction schemes, this intermediate is notoriously unstable, prone to hydrolysis, and difficult to store, leading to inconsistent quality and yield losses during isolation. Furthermore, the subsequent reduction step frequently employs sodium borohydride aqueous solutions or lithium aluminum hydride. Sodium borohydride is highly susceptible to oxidation and moisture absorption, requiring strict storage conditions, while lithium aluminum hydride poses severe safety risks due to its pyrophoric nature and violent reactivity with water. Additionally, conventional bromination processes using molecular bromine alone generate stoichiometric amounts of corrosive hydrogen bromide gas, which severely damages industrial equipment and necessitates costly waste treatment protocols.
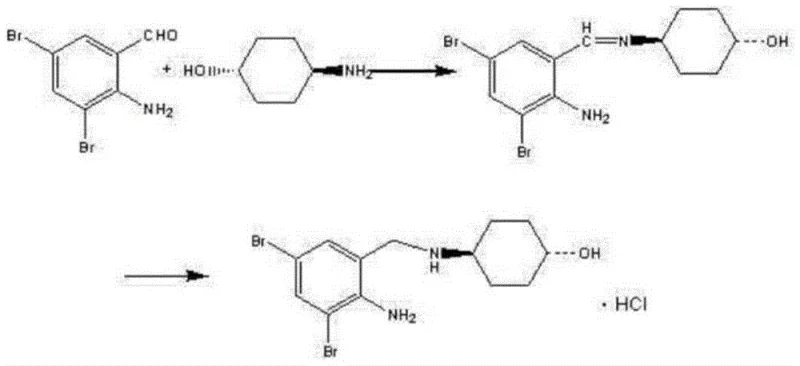
The Novel Approach
In stark contrast, the methodology outlined in CN111072500B circumvents these pitfalls through a clever sequence of protection, reduction, and functionalization. Instead of starting with the sensitive amino-aldehyde, the process begins with o-nitrobenzaldehyde, which is first protected as an acetal. This protection step stabilizes the reactive aldehyde functionality, allowing for subsequent transformations without degradation. The nitro group is then reduced using a NiCoB/TiO2 catalyst and hydrazine hydrate, a combination that is far safer and more controllable than metal hydrides. Following this, the bromination is conducted in the presence of hydrogen peroxide, which acts as an oxidant to recycle the generated hydrogen bromide back into molecular bromine. This innovative loop not only improves atom economy theoretically saving half the bromine reagent but also mitigates equipment corrosion. Finally, the deprotected aldehyde undergoes reductive amination with trans-4-aminocyclohexanol using sodium triacetoxyborohydride, a mild reducing agent that ensures high selectivity and avoids the formation of toxic by-products, thereby facilitating a smoother path to commercial scale-up of complex pharmaceutical intermediates.
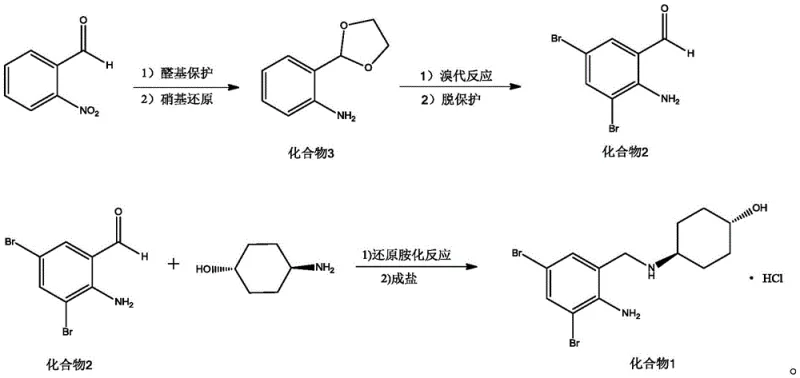
Mechanistic Insights into NiCoB/TiO2 Catalyzed Reduction and Oxidative Bromination
The heart of this novel synthesis lies in the sophisticated application of heterogeneous catalysis and oxidative recycling mechanisms. The use of NiCoB/TiO2 as a catalyst for the reduction of the nitro group is particularly noteworthy. This amorphous alloy catalyst, supported on titanium dioxide, provides a high surface area and unique electronic properties that facilitate the activation of hydrazine hydrate as a hydrogen source. Unlike traditional noble metal catalysts which can be cost-prohibitive or susceptible to poisoning, the NiCoB system offers robust performance at moderate temperatures of 50-55°C. The mechanism likely involves the adsorption of the nitro compound onto the catalyst surface, where hydrazine decomposes to provide active hydrogen species that sequentially reduce the nitro group to an amine without affecting the protected aldehyde moiety. This chemoselectivity is crucial for maintaining the integrity of the molecule throughout the synthesis, ensuring that the final product meets high-purity ambroxol hydrochloride specifications without requiring extensive purification steps that would otherwise erode profit margins.
Equally important is the mechanistic advantage of the bromination step utilizing molecular bromine and hydrogen peroxide. In standard electrophilic aromatic substitution, the replacement of a hydrogen atom with bromine releases a proton and a bromide ion, forming hydrogen bromide (HBr). In this patented process, hydrogen peroxide serves as an oxidizing agent that converts the liberated HBr back into elemental bromine (Br2) in situ. This regeneration cycle means that the effective concentration of the brominating agent is maintained throughout the reaction, driving the equilibrium towards completion and minimizing the waste of expensive bromine reagents. From a process safety perspective, this also means that the accumulation of corrosive acidic gases is significantly reduced, protecting the reactor lining and downstream piping. The subsequent deprotection of the acetal group using p-toluenesulfonic acid in acetone is a straightforward acid-catalyzed hydrolysis that cleanly reveals the aldehyde, readying it for the final coupling reaction with high fidelity.
How to Synthesize Ambroxol Hydrochloride Efficiently
Implementing this synthesis requires careful attention to the specific stoichiometric ratios and solvent systems detailed in the patent to maximize yield and safety. The process is divided into three distinct operational stages: the formation of the protected amino-intermediate, the oxidative bromination and deprotection to generate the key aldehyde building block, and the final reductive amination to construct the target molecule. Each step has been optimized to balance reaction kinetics with practical handling requirements, ensuring that the procedure is robust enough for multi-kilogram production. For process engineers looking to adopt this technology, understanding the nuances of the catalyst preparation and the precise addition rates of oxidants is critical for reproducibility. The detailed standardized synthesis steps see the guide below outline the exact parameters required to achieve the reported yields of over 96% in the final step.
- Protect o-nitrobenzaldehyde with ethylene glycol and reduce the nitro group using NiCoB/TiO2 catalyst and hydrazine hydrate to form Compound 3.
- Perform bromination on Compound 3 using molecular bromine and hydrogen peroxide, followed by acid-catalyzed deprotection to yield 2-amino-3,5-dibromobenzaldehyde (Compound 2).
- Conduct reductive amination between Compound 2 and trans-4-aminocyclohexanol using NaBH(OAc)3 and LiClO4, followed by salt formation with HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented method offers tangible strategic benefits that extend beyond the laboratory bench. The primary advantage lies in the substantial cost savings achieved through reagent efficiency and waste reduction. By recycling bromine atoms within the reaction system, the consumption of molecular bromine is theoretically halved compared to traditional methods. Since bromine is a commodity chemical with fluctuating prices, reducing the dependency on fresh feedstock directly translates to lower variable costs per kilogram of finished product. Furthermore, the elimination of hazardous reagents like lithium aluminum hydride removes the need for specialized storage facilities and expensive quenching procedures, simplifying the operational overhead. The use of common solvents like toluene, dichloromethane, and acetone ensures that raw material sourcing remains stable and resilient against market shocks, enhancing the overall reliability of the supply chain for respiratory drug manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the dual mechanisms of reagent recycling and simplified downstream processing. The oxidative bromination system not only saves on bromine costs but also reduces the burden on waste treatment facilities by minimizing the discharge of corrosive acidic effluents. Additionally, the high selectivity of the NaBH(OAc)3 reduction minimizes the formation of side products, which means less material is lost during purification and recrystallization. This improved mass balance results in a higher overall throughput from the same amount of starting material, effectively lowering the cost of goods sold (COGS). The avoidance of cryogenic conditions or high-pressure hydrogenation equipment further reduces capital expenditure and energy consumption, making the process economically attractive for both pilot and full-scale production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche or hazardous chemicals that have limited suppliers. This method utilizes o-nitrobenzaldehyde and trans-4-aminocyclohexanol, which are widely available bulk chemicals with established global supply chains. The catalyst system, based on nickel and cobalt, avoids the geopolitical risks associated with precious metals like palladium or platinum. Moreover, the stability of the intermediates, particularly the protected forms, allows for potential stockpiling or semi-finished goods inventory, providing a buffer against demand spikes. This flexibility ensures that reducing lead time for high-purity pharmaceutical intermediates becomes a manageable objective rather than a logistical bottleneck, allowing manufacturers to respond swiftly to market needs for generic respiratory medications.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental flaws, but this route is designed with industrial viability in mind. The mild reaction conditions (50-55°C) and ambient pressure operations reduce the risk of thermal runaways, a critical factor for safety audits and insurance compliance. The recycling of HBr significantly lowers the E-factor (environmental factor) of the process by reducing hazardous waste generation. This alignment with green chemistry principles facilitates easier regulatory approval and permits for new production lines. The simplicity of the workup procedures, involving standard extractions and crystallizations, means that existing multipurpose reactors can be easily adapted for this synthesis without major retrofitting, accelerating the timeline from technology transfer to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does the new bromination method improve equipment longevity?
A: By utilizing hydrogen peroxide as an oxidant alongside molecular bromine, the generated hydrogen bromide (HBr) is oxidized back into molecular bromine within the reaction system. This recycling mechanism prevents the accumulation of corrosive HBr, significantly reducing equipment corrosion and extending industrial reactor lifespan.
Q: Why is the NiCoB/TiO2 catalyst preferred over traditional reduction methods?
A: The NiCoB/TiO2 amorphous alloy catalyst offers superior activity and selectivity for nitro reduction using hydrazine hydrate. Unlike hazardous lithium aluminum hydride or unstable sodium borohydride aqueous solutions, this catalytic system operates under mild conditions (50-55°C) without generating harmful by-products, ensuring safer large-scale operations.
Q: What are the purity specifications achievable with this route?
A: Experimental data from the patent indicates that the final Ambroxol Hydrochloride product can achieve purity levels exceeding 99.7% after recrystallization. The use of mild reductive amination reagents like NaBH(OAc)3 minimizes side reactions, resulting in a cleaner impurity profile suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ambroxol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN111072500B for the global respiratory health market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific catalytic and oxidative requirements of this route, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality Ambroxol Hydrochloride intermediates that meet the demanding standards of international pharmacopoeias, providing our partners with a competitive edge in the generic drug sector.
We invite procurement leaders and R&D teams to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this efficient route can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical pharmaceutical ingredient, ensuring your production lines remain efficient and compliant in an evolving regulatory landscape.
