Optimizing Ambroxol Hydrochloride Production: A Green Three-Step Synthetic Route for Industrial Scale
Optimizing Ambroxol Hydrochloride Production: A Green Three-Step Synthetic Route for Industrial Scale
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical respiratory therapeutics. A significant breakthrough in this domain is detailed in patent CN111072499A, which discloses a novel synthesis process for Ambroxol Hydrochloride, a widely used mucolytic agent. This technology represents a paradigm shift from traditional aldehyde-based condensation methods to a more stable, ester-based synthetic strategy. By utilizing methyl anthranilate as the starting material, the process circumvents the inherent instability of Schiff base intermediates found in legacy routes. The methodology employs a green bromination system coupled with a biomimetic reduction step, achieving exceptional purity levels exceeding 99% while maintaining mild reaction conditions. For global procurement teams and R&D directors, this patent offers a compelling blueprint for enhancing supply chain resilience and reducing the environmental footprint of API manufacturing.
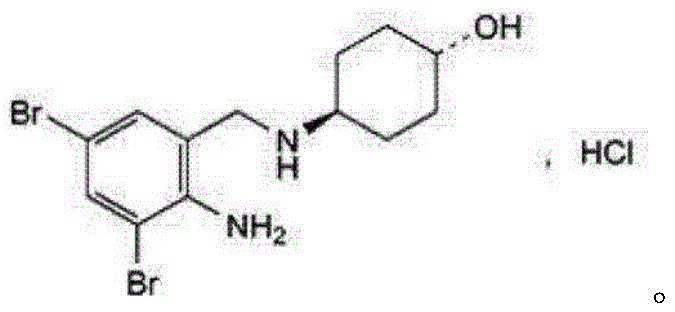
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ambroxol Hydrochloride has relied heavily on the condensation of 2-amino-3,5-dibromobenzaldehyde with trans-4-aminocyclohexanol. While conceptually straightforward, this pathway is plagued by significant operational deficiencies that hinder large-scale efficiency. The primary bottleneck lies in the formation of the Schiff base intermediate, trans-4-[(2-amino-3,5-dibromobenzylidene)amino]cyclohexanol. As illustrated in prior art routes, this intermediate is chemically unstable and highly susceptible to hydrolysis, making isolation, storage, and quality control exceptionally difficult. Furthermore, the subsequent reduction step typically necessitates the use of hazardous reagents such as sodium borohydride or lithium aluminum hydride. These reducing agents are not only moisture-sensitive and prone to oxidation but also introduce severe safety risks due to their potential for violent exothermic reactions. Additionally, the generation of corrosive hydrogen bromide during traditional bromination processes accelerates equipment degradation, leading to increased maintenance downtime and capital expenditure for reactor lining replacements.
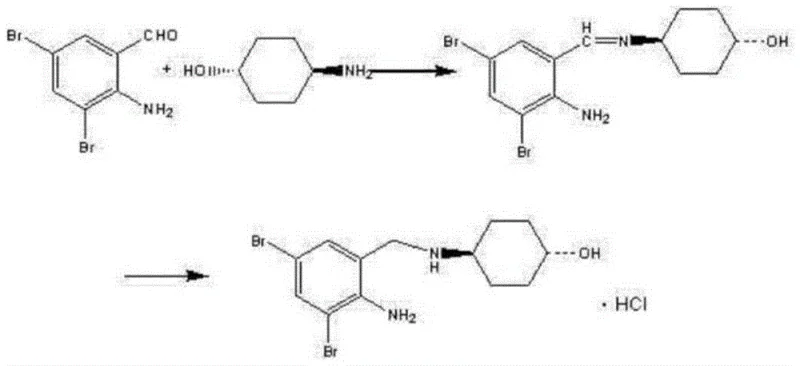
The Novel Approach
In stark contrast to these legacy challenges, the process disclosed in CN111072499A introduces a streamlined three-step sequence that fundamentally alters the chemical logic of the synthesis. By initiating the synthesis with methyl anthranilate rather than the aldehyde, the process avoids the formation of the unstable imine linkage entirely. Instead, it proceeds through a stable amide intermediate, which is far more robust against hydrolysis and easier to purify. The innovation extends to the bromination step, where molecular bromine is utilized in conjunction with hydrogen peroxide. This dual-reagent system creates a catalytic cycle where hydrogen bromide byproducts are oxidized back into molecular bromine, theoretically saving 50% of the bromine reagent and eliminating corrosive waste. The final reduction utilizes Hantzsch ester, a mild organic hydride donor, which operates safely at room temperature. This holistic redesign results in a process that is not only higher yielding but also inherently safer and more compatible with continuous manufacturing environments.
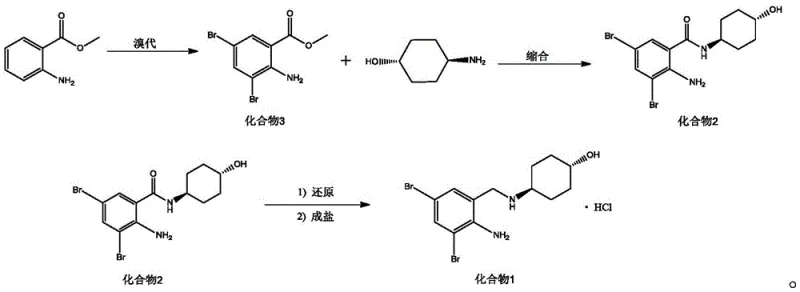
Mechanistic Insights into Green Bromination and Biomimetic Reduction
The core chemical innovation of this process lies in its sophisticated management of redox chemistry and coupling efficiency. In the initial bromination stage, the reaction system leverages the oxidative power of hydrogen peroxide (30% mass fraction) to regenerate elemental bromine in situ. Mechanistically, as the electrophilic aromatic substitution proceeds on the methyl anthranilate ring, hydrogen bromide is released as a stoichiometric byproduct. In conventional systems, this HBr accumulates, lowering pH and corroding steel reactors. However, in this novel protocol, the peroxide oxidizes the HBr back to Br2, which re-enters the reaction cycle. This atom-economic loop ensures that the utilization rate of bromine atoms approaches theoretical maximums, drastically reducing raw material consumption. The reaction is conducted in a biphasic dichloromethane-water system, which facilitates efficient heat dissipation and simplifies the workup procedure, yielding the dibromo-intermediate with purity levels reaching 99.92% after simple recrystallization.
Furthermore, the reduction mechanism employing Hantzsch ester (1,4-dihydropyridine derivative) represents a significant advancement in functional group tolerance and safety. Catalyzed by trifluoromethanesulfonic anhydride, the Hantzsch ester acts as a hydride source to reduce the amide carbonyl to the corresponding methylene amine. Unlike metal hydrides that require strict anhydrous conditions and cryogenic temperatures to prevent runaway reactions, this organic reduction proceeds smoothly at room temperature. The catalyst activates the carbonyl oxygen, making it susceptible to nucleophilic attack by the dihydropyridine hydride. This mildness preserves the stereochemical integrity of the trans-4-aminocyclohexanol moiety, preventing racemization which is a common impurity concern in high-temperature reductions. The result is a final product profile with minimal impurities, reducing the burden on downstream purification units and ensuring consistent batch-to-batch quality for regulatory compliance.
How to Synthesize Ambroxol Hydrochloride Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and reaction parameters to maximize the benefits of the green chemistry principles employed. The process is divided into three distinct operational phases: the oxidative bromination of the starting ester, the peptide-like coupling with the cyclohexyl amine, and the final catalytic reduction. Each step has been optimized in the patent examples to demonstrate scalability, with yields consistently exceeding 90% across multi-gram trials. The use of common solvents like dichloromethane, DMF, and toluene ensures that the process can be adapted to existing infrastructure without requiring exotic equipment. For technical teams looking to adopt this methodology, adherence to the specific molar ratios of the coupling agents (DIC/DMAP) and the careful addition of the oxidant are critical for success. Detailed standardized operating procedures for each unit operation are essential to maintain the high purity specifications required for pharmaceutical intermediates.
- Perform bromination on methyl anthranilate using molecular bromine and hydrogen peroxide in a dichloromethane/water system to generate the dibromo intermediate.
- Conduct ester-amine condensation between the dibromo intermediate and trans-4-aminocyclohexanol using DIC and DMAP catalysts in DMF solvent.
- Execute carbonyl reduction using Hantzsch ester and trifluoromethanesulfonic anhydride catalyst, followed by salification with hydrochloric acid to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic advantages beyond mere technical elegance. The primary value driver is the substantial reduction in raw material volatility and cost. By replacing expensive and hazardous reducing agents like Lithium Aluminum Hydride with stable, commercially available Hantzsch esters, the process mitigates the risk of supply disruptions associated with controlled or dangerous chemicals. Moreover, the improved atom economy in the bromination step directly translates to lower material costs per kilogram of finished product. The elimination of corrosive hydrogen bromide waste also reduces the financial burden associated with waste treatment and neutralization, contributing to a leaner cost structure. These factors combined create a more predictable costing model, allowing for better long-term budget planning and price stability for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization through the recycling of bromine reagents and the use of non-precious catalysts. By avoiding the need for cryogenic cooling systems required by traditional metal hydride reductions, energy consumption is drastically lowered. The high yield of each step minimizes the loss of valuable intermediates, ensuring that the overall throughput of the plant is maximized without increasing the footprint of the reaction vessels. This efficiency allows for competitive pricing strategies in the global market for respiratory drug intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable starting materials like methyl anthranilate and trans-4-aminocyclohexanol ensures a robust supply chain. Unlike aldehyde precursors which can degrade over time, the ester intermediates in this route have extended shelf lives, reducing inventory write-offs. The safety profile of the reagents means that transportation and storage logistics are simplified, as there is no need for specialized hazardous material handling for the reducing agents. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for international clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal residues make this process highly scalable from pilot plant to commercial tonnage. The environmental benefits are profound, as the process generates less hazardous waste and avoids the emission of corrosive gases. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the risk of environmental compliance penalties. For companies aiming to meet ESG (Environmental, Social, and Governance) goals, adopting this synthesis route demonstrates a commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the quality implications of the new route. The answers highlight the specific advantages in terms of purity, safety, and operational simplicity that distinguish this method from conventional practices.
Q: Why is the methyl anthranilate route superior to the traditional aldehyde condensation method?
A: The traditional aldehyde route produces an unstable Schiff base intermediate that is prone to hydrolysis and difficult to store. The methyl anthranilate route utilizes a stable ester intermediate, eliminating isolation steps and significantly improving process robustness and yield.
Q: How does the new bromination method improve equipment longevity?
A: By using hydrogen peroxide as an oxidant alongside molecular bromine, the generated hydrogen bromide is oxidized back into molecular bromine within the reaction system. This prevents the accumulation of corrosive HBr gas, thereby reducing equipment corrosion and maintenance costs.
Q: What are the safety advantages of using Hantzsch ester over Lithium Aluminum Hydride?
A: Hantzsch ester is a mild, non-toxic reducing agent that operates under ambient conditions. Unlike Lithium Aluminum Hydride or Sodium Borohydride, it does not pose risks of violent exothermic reactions, moisture sensitivity, or toxicity, making it ideal for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ambroxol Hydrochloride Supplier
The synthesis technology described in CN111072499A exemplifies the kind of innovation that drives modern pharmaceutical manufacturing forward. At NINGBO INNO PHARMCHEM, we specialize in translating such complex laboratory methodologies into robust, commercial-scale production realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of green chemistry are fully realized in the final product. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Ambroxol Hydrochloride meets the highest international pharmacopoeia standards. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth downstream API synthesis without unexpected impurity profiles.
We invite global partners to collaborate with us to leverage this advanced synthesis route for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your production costs and secure a reliable supply of high-quality pharmaceutical intermediates for the global market.
