Advanced Manufacturing of Ambroxol Hydrochloride via Green Aqueous Chemistry and Safe Borane Reduction
Advanced Manufacturing of Ambroxol Hydrochloride via Green Aqueous Chemistry and Safe Borane Reduction
The pharmaceutical industry continuously seeks robust manufacturing pathways for essential respiratory therapeutics, and the synthesis of Ambroxol Hydrochloride stands as a prime example of process optimization. Patent CN112341347A, published in early 2021, introduces a transformative synthetic methodology that addresses long-standing safety and efficiency bottlenecks in producing this mucolytic agent. By shifting the starting material strategy to isatoic anhydride and utilizing an aqueous reaction medium for the initial acylation, this technology offers a compelling alternative to legacy processes reliant on hazardous reagents. For R&D directors and supply chain managers, understanding the nuances of this patent is critical for securing a reliable ambroxol hydrochloride supplier capable of delivering high-purity intermediates at scale.
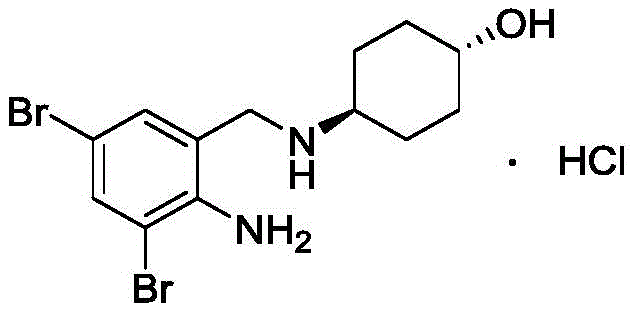
This technical insight report dissects the proprietary route disclosed in the patent, highlighting its superiority over classical methods that suffer from low atom economy and dangerous operating conditions. The core innovation lies in the strategic replacement of lithium aluminum hydride with a boron-based reduction system and the elimination of toxic solvents like carbon tetrachloride. These modifications not only enhance the safety profile for industrial mass production but also align with modern environmental compliance standards, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
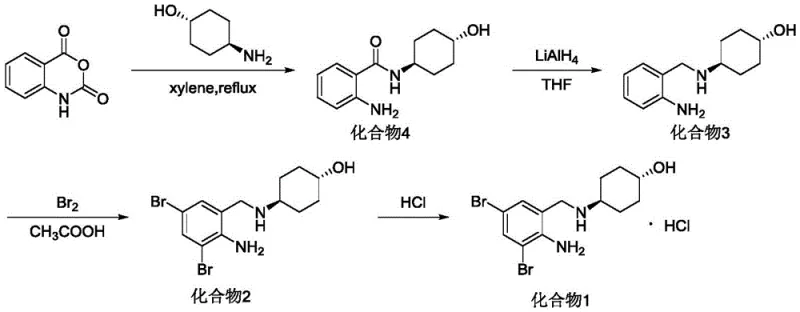
Historically, the synthesis of Ambroxol Hydrochloride has been plagued by inefficient multi-step sequences that compromise both yield and operator safety. As detailed in prior art such as J. Keck's work and US Patent 3536713, traditional routes often necessitate the use of lithium aluminum hydride (LiAlH4) for the critical amide reduction step. This reagent is notoriously pyrophoric, requiring stringent anhydrous conditions and specialized handling equipment, which drastically inflates capital expenditure and operational risk. Furthermore, legacy pathways frequently employ halogenated solvents like carbon tetrachloride or xylene, creating significant environmental liabilities and complicating waste stream management. The cumulative effect of these inefficiencies is often a dismal total yield, reported in some literature to be as low as 4.2%, rendering such processes economically unviable for large-scale commercialization.
The Novel Approach
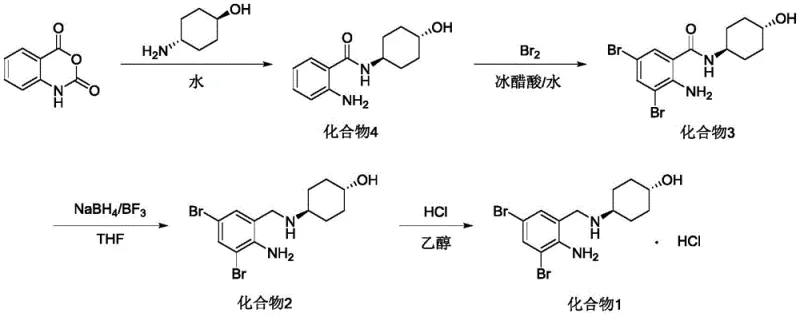
In stark contrast, the methodology outlined in CN112341347A presents a streamlined four-step sequence that prioritizes safety and atom economy without sacrificing chemical fidelity. The process initiates with a ring-opening acylation in water, a benign solvent that eliminates the need for volatile organic compounds during the formation of the key amide intermediate. Subsequent bromination is conducted in a glacial acetic acid and water mixture, achieving near-quantitative conversion rates that minimize raw material waste. Most notably, the reduction step utilizes a generated borane complex from sodium borohydride and boron trifluoride, offering a controllable and less hazardous alternative to LiAlH4. This novel approach ensures a stable supply chain by relying on commercially abundant reagents and simplifies purification protocols, directly addressing the pain points of cost and scalability.
Mechanistic Insights into Aqueous Acylation and Borane-Mediated Reduction
The mechanistic elegance of this synthesis begins with the nucleophilic attack of trans-4-aminocyclohexanol on the carbonyl carbon of isatoic anhydride. In the presence of water, the anhydride ring opens readily, releasing carbon dioxide and forming the stable trans-4-[(2-aminobenzoyl)amino]cyclohexanol intermediate. The use of water as a solvent is particularly ingenious; it facilitates heat dissipation during the exothermic ring-opening and allows for simple filtration of the product, which precipitates out of the aqueous phase with high purity. This eliminates the need for complex extraction workflows typically associated with organic solvents, thereby reducing processing time and solvent recovery costs significantly.
The subsequent transformation involves a highly selective electrophilic aromatic substitution followed by a sophisticated reduction mechanism. Bromination occurs ortho to the amino group, directed by the activating nature of the nitrogen lone pair, to install the requisite 3,5-dibromo pattern with 100% yield. The final reduction of the amide bond to the amine is achieved through the in situ generation of borane species. Sodium borohydride reacts with boron trifluoride diethyl etherate to produce a potent reducing agent capable of cleaving the robust amide linkage under reflux conditions in tetrahydrofuran. This mechanism avoids the over-reduction or side reactions often seen with harsher hydride sources, ensuring the integrity of the cyclohexanol ring and the stereochemical configuration of the final API intermediate.
How to Synthesize Ambroxol Hydrochloride Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the patented protocol. The process is designed to be scalable, moving seamlessly from laboratory benchtop to kiloton production facilities. Operators must pay close attention to temperature gradients during the bromination exotherm and the stoichiometry of the boron reagents during reduction to ensure consistent quality. The following guide outlines the standardized operational framework derived from the patent examples, serving as a blueprint for process engineers aiming to replicate these high-efficiency results.
- React isatoic anhydride with trans-4-aminocyclohexanol in water at 30-40°C to form trans-4-[(2-aminobenzoyl)amino]cyclohexanol.
- Perform bromination using bromine in glacial acetic acid/water mixture at 5-15°C to obtain the dibromo intermediate.
- Reduce the amide bond using sodium borohydride and boron trifluoride diethyl etherate in THF under reflux.
- Form the hydrochloride salt by treating the free base with concentrated hydrochloric acid in ethanol/ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic advantages beyond mere chemical yield. The shift away from hazardous reagents like lithium aluminum hydride and toxic solvents like carbon tetrachloride fundamentally alters the risk profile of the manufacturing process. By mitigating the dangers associated with pyrophoric materials, facilities can reduce insurance premiums and safety compliance overheads. Furthermore, the reliance on water and acetic acid as primary solvents in the early stages significantly lowers the cost of goods sold (COGS) by minimizing the volume of expensive organic solvents required for reaction and workup.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents such as lithium aluminum hydride creates a direct pathway for substantial cost savings. Sodium borohydride and boron trifluoride complexes are industrially abundant and priced significantly lower than their pyrophoric counterparts. Additionally, the high yield of the bromination step, reported at 100%, ensures that valuable bromine reagents are fully utilized, preventing the financial loss associated with unreacted starting materials and complex recycling streams.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically isatoic anhydride and trans-4-aminocyclohexanol, are commodity chemicals with stable global supply chains. Unlike specialized intermediates required in other synthetic pathways, these precursors are readily available from multiple vendors, reducing the risk of supply disruption. The robustness of the aqueous acylation step also means that production is less sensitive to moisture variations, allowing for more flexible scheduling and reduced downtime due to environmental controls.
- Scalability and Environmental Compliance: The process is inherently designed for industrial mass production, evidenced by the use of simple filtration and crystallization techniques rather than chromatography. The replacement of chlorinated solvents with water and ethanol/ether mixtures simplifies wastewater treatment and aligns with increasingly strict environmental regulations. This green chemistry approach not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this route represents a superior choice for commercial manufacturing.
Q: Why is the new synthesis route safer than conventional methods?
A: Conventional routes often utilize lithium aluminum hydride (LiAlH4), which is pyrophoric and hazardous on large scale. This patent replaces it with a safer sodium borohydride/boron trifluoride system.
Q: How does the aqueous solvent system impact cost?
A: Replacing organic solvents like xylene with water in the initial acylation step significantly reduces solvent procurement costs and simplifies waste treatment protocols.
Q: What is the overall yield advantage of this method?
A: While older literature reports total yields as low as 4.2%, this optimized route achieves individual step yields of up to 100% for bromination and 98.5% for acylation, drastically improving overall throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ambroxol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process validation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ambroxol Hydrochloride meets the exacting standards required for respiratory therapeutic applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this safer, more efficient methodology. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production volumes.
