Advanced Lewis Acid Catalysis for Scalable Dihydrofuran Derivative Production
Advanced Lewis Acid Catalysis for Scalable Dihydrofuran Derivative Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and minimal environmental impact. A significant breakthrough in this domain is documented in patent CN110938048B, which discloses a novel synthesis method for efficiently forming carbon-carbon bonds and cyclizing dihydrofuran derivatives. This technology leverages a Lewis acid-catalyzed insertion reaction where ionic liquids serve as the solvent and sulfur ylides act as the nucleophilic reagent. For R&D directors and procurement managers alike, this represents a paradigm shift away from traditional, hazardous synthetic routes towards a greener, more economically viable manufacturing process. The ability to construct the dihydrofuran core—a motif prevalent in numerous bioactive natural products and pharmaceutical agents—under mild conditions addresses critical pain points regarding safety, cost, and scalability in modern chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan and its derivatives has relied on classical methodologies such as the Paal-Knorr Furan synthesis or the Nickl Benzofuran Synthesis. While these methods established the foundational chemistry for heterocycle formation, they are increasingly viewed as obsolete for modern large-scale applications due to inherent inefficiencies. These traditional pathways often necessitate harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can lead to the degradation of sensitive functional groups and result in poor substrate compatibility. Furthermore, the atom economy of these older processes is frequently suboptimal, generating substantial quantities of chemical waste that require costly disposal procedures. Perhaps most critically, many contemporary approaches to constructing substituted furans rely on diazo compounds as carbene precursors. Diazo compounds are notoriously unstable, toxic, and potentially explosive, posing severe safety risks during storage, handling, and scale-up, thereby complicating regulatory compliance and increasing insurance and operational overheads for manufacturing facilities.
The Novel Approach
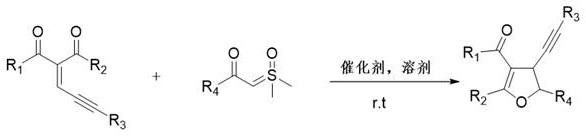
In stark contrast to these legacy methods, the technology described in patent CN110938048B introduces a streamlined, one-step cyclization strategy that utilizes sulfur ylides as stable and safe alternatives to diazo reagents. This innovative approach employs enynone compounds as starting materials, which undergo a Michael addition-type insertion reaction catalyzed by inexpensive Lewis acids. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling systems. By utilizing ionic liquids as the reaction medium, the process avoids the use of volatile organic compounds (VOCs), aligning with stringent environmental regulations and green chemistry principles. The versatility of this method is demonstrated by its tolerance to various substituents, allowing for the rapid generation of diverse dihydrofuran libraries. This simplicity not only accelerates the R&D timeline for new drug candidates but also simplifies the technological transfer to commercial production scales, ensuring a reliable supply of high-purity intermediates.
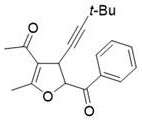
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic innovation lies in the precise activation of the enynone substrate by the Lewis acid catalyst. When a Lewis acid such as zinc chloride (ZnCl2) or copper chloride is introduced into the ionic liquid medium, it coordinates with the carbonyl oxygen of the enynone. This coordination significantly increases the electrophilicity of the beta-carbon, facilitating a nucleophilic attack by the sulfur ylide. Unlike traditional carbene transfer reactions that require transition metals like gold or rhodium to generate free carbenes, this mechanism likely proceeds through a concerted or stepwise insertion pathway where the sulfur ylide acts directly as a nucleophile in a Michael addition fashion. Following the initial C-C bond formation, an intramolecular cyclization occurs, driven by the nucleophilic attack of the enolate oxygen onto the activated alkyne or adjacent electrophilic center, ultimately closing the dihydrofuran ring. This mechanistic pathway avoids the formation of highly reactive free carbene species, thereby minimizing side reactions such as dimerization or non-selective C-H insertion, which are common pitfalls in metal-carbene chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical manufacturing. The mildness of the room temperature reaction conditions suppresses thermal decomposition pathways that often lead to complex, difficult-to-remove impurity profiles. Additionally, the use of sulfur ylides, which are thermally stable solids, ensures consistent reagent quality and reduces the risk of batch-to-batch variability caused by reagent degradation. The ionic liquid solvent further contributes to purity by providing a homogeneous reaction environment that stabilizes charged intermediates, preventing premature precipitation or aggregation that could trap impurities. Consequently, the crude reaction mixtures obtained from this process are typically cleaner, requiring less aggressive purification protocols. This translates to higher overall yields and reduced solvent consumption during the workup phase, directly impacting the cost of goods sold (COGS) and enhancing the sustainability profile of the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize Dihydrofuran Derivatives Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to minimize operational complexity while maximizing output. The protocol involves the sequential addition of the enynone substrate, the sulfur ylide nucleophile, and the Lewis acid catalyst into a reactor containing the ionic liquid solvent. The mixture is then agitated at ambient temperature, allowing the reaction to proceed to completion overnight without the need for external heating or specialized pressure equipment. This operational simplicity makes the process highly attractive for both laboratory-scale optimization and pilot-plant trials. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below to guide technical teams in replicating this high-efficiency route.
- Charge a clean reactor with enynone compound, sulfur ylide compound, Lewis acid catalyst (e.g., ZnCl2), and ionic liquid solvent sequentially.
- Stir the reaction mixture at room temperature overnight to ensure complete conversion and cyclization.
- Upon completion, purify the crude residue directly using silica gel column chromatography to isolate the high-purity dihydrofuran product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-catalyzed methodology presents a compelling value proposition centered on risk mitigation and cost optimization. The shift from hazardous diazo compounds to stable sulfur ylides fundamentally alters the safety landscape of the manufacturing facility, reducing the need for specialized explosion-proof infrastructure and lowering insurance premiums. Furthermore, the reliance on abundant and inexpensive Lewis acid catalysts, such as zinc or iron salts, instead of precious metals like palladium or gold, drastically reduces raw material costs. The elimination of toxic volatile organic solvents in favor of recyclable ionic liquids not only ensures compliance with increasingly strict environmental regulations but also minimizes the logistical burden associated with solvent recovery and waste disposal. These factors collectively contribute to a more resilient and cost-effective supply chain capable of sustaining long-term production runs.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. By avoiding the use of precious metal catalysts and unstable diazo compounds, manufacturers can achieve substantial savings on raw material procurement. Additionally, the mild reaction conditions eliminate the energy costs associated with heating or cryogenic cooling, while the simplified workup procedure reduces solvent consumption and labor hours required for purification. The potential for recycling the ionic liquid solvent further amplifies these savings over time, creating a compounding effect on the overall cost reduction in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity or regulatory restrictions of key reagents. Sulfur ylides and simple Lewis acids are commodity chemicals with robust global supply chains, ensuring consistent availability and shielding production schedules from market volatility. The stability of these reagents also allows for longer shelf lives and simpler storage requirements, reducing the risk of production stoppages due to reagent spoilage. This reliability is crucial for maintaining just-in-time delivery schedules for downstream pharmaceutical clients who depend on uninterrupted supplies of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges related to heat transfer and mixing, particularly with exothermic reactions involving unstable intermediates. The room temperature nature of this insertion reaction inherently mitigates thermal runaway risks, making the scale-up from grams to tons significantly safer and more predictable. Moreover, the green chemistry attributes of the process, specifically the avoidance of VOCs and the use of non-toxic catalysts, streamline the environmental permitting process. This facilitates faster deployment of commercial capacity and ensures long-term operational viability in regions with stringent environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydrofuran synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the advantages of using sulfur ylides over diazo compounds in this synthesis?
A: Sulfur ylides offer significantly improved safety profiles compared to diazo compounds, which are often unstable and toxic. They act as stable carbene precursors, allowing for milder reaction conditions and easier handling in large-scale manufacturing environments.
Q: Can the ionic liquid solvent be recycled in this process?
A: Yes, the use of ionic liquids such as [BMIM]NTf2 not only provides a green solvent medium with negligible vapor pressure but also facilitates potential recycling strategies, reducing waste generation and lowering overall solvent costs in continuous production.
Q: What types of Lewis acid catalysts are compatible with this method?
A: The method demonstrates broad compatibility with various Lewis acids including zinc chloride, copper salts, and iron chlorides. This flexibility allows manufacturers to select cost-effective and readily available catalysts suitable for their specific supply chain requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrofuran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Lewis acid-catalyzed insertion reaction for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle ionic liquid chemistries and moisture-sensitive reagents with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch. We are committed to delivering dihydrofuran derivatives that meet the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in green catalysis can drive value and efficiency in your supply chain.
