Advanced Synthetic Route for Ivabradine: Enhancing Purity and Commercial Scalability for Global Pharma
Advanced Synthetic Route for Ivabradine: Enhancing Purity and Commercial Scalability for Global Pharma
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for more efficient manufacturing processes of critical active pharmaceutical ingredients (APIs). Patent CN102498102A introduces a groundbreaking methodology for the synthesis of Ivabradine and its pharmaceutically acceptable acid addition salts, specifically addressing the longstanding inefficiencies in prior art. This innovation is particularly relevant for R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier who can guarantee both high purity and supply continuity. By re-engineering the alkylation steps and optimizing reaction conditions, this new route offers a compelling alternative to legacy methods that have historically plagued production lines with low yields and complex purification burdens.
The core of this technological advancement lies in the strategic construction of the molecular scaffold using novel intermediates that facilitate cleaner coupling reactions. Unlike earlier attempts that struggled with reproducibility and yield attrition, the process detailed in this patent leverages specific halogenated or sulfonated propyl amine derivatives to achieve superior conversion rates. For supply chain heads, this translates to a more predictable manufacturing timeline and reduced risk of batch failures. The ability to produce Ivabradine hydrochloride with consistent quality metrics is essential for maintaining the integrity of the global drug supply, especially for medications treating myocardial ischemia and heart failure where dosage precision is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ivabradine has been fraught with significant technical hurdles that impact both cost and availability. The method described in European patent specification EP 0 534 859, while foundational, suffers from a critical disadvantage: the overall yield of Ivabradine after three synthetic steps is less than 17%. Such low efficiency is commercially unsustainable, leading to excessive waste of raw materials and inflated production costs that ultimately burden the healthcare system. Furthermore, subsequent attempts to improve this route, such as those disclosed in international application WO 2008/065681, have proven difficult to reproduce reliably. Applicants found that replicating the WO 2008/065681 method failed to produce the target hydrochloride salt effectively, highlighting the fragility of these older protocols when transferred from laboratory scale to industrial manufacturing environments.
The Novel Approach
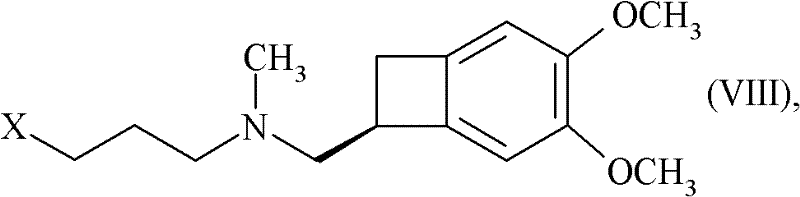
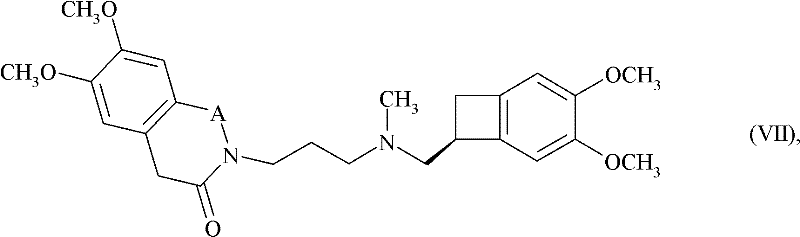
In stark contrast, the method presented in CN102498102A overcomes these barriers through a refined alkylation strategy that ensures robust reaction kinetics and easier purification. The process utilizes a compound of Formula (VIII), where X represents a halogen atom, mesylate, or tosylate group, reacting with a benzazepinone derivative of Formula (IX). This specific pairing allows for a highly selective nucleophilic substitution that minimizes side reactions. By operating under mild conditions, often at room temperature in solvents like dimethyl sulfoxide (DMSO), the new approach drastically simplifies the operational complexity. This shift not only enhances the chemical yield but also streamlines the downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent quality standards required for cardiovascular drugs.
Mechanistic Insights into Base-Mediated Alkylation and Cyclization
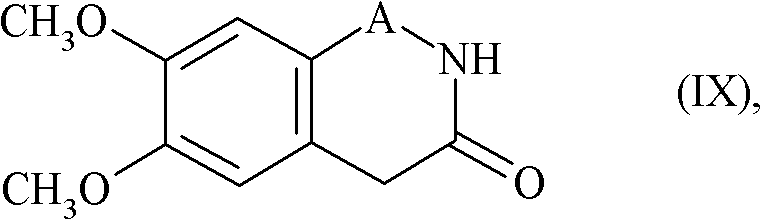
The mechanistic elegance of this synthesis rests on the precise activation of the nitrogen nucleophile within the benzazepinone ring system. In the key coupling step, a strong base such as potassium tert-butoxide is employed to deprotonate the lactam nitrogen of the 7,8-dimethoxy-1,3,4,5-tetrahydro-2H-3-benzazepin-2-one. This generates a highly reactive anionic species that attacks the electrophilic carbon of the propyl chain in Formula (VIII). The choice of base is critical; while sodium hydride or potassium carbonate could theoretically function, potassium tert-butoxide in DMSO provides the optimal balance of solubility and basicity to drive the reaction to completion within a reasonable timeframe, typically overnight at room temperature. This mechanistic pathway avoids the need for harsh thermal conditions that might degrade the sensitive methoxy-substituted aromatic rings.
Furthermore, the process incorporates a strategic flexibility regarding the saturation of the propyl linker. In embodiments where the linker contains unsaturation (A represents HC=CH), a subsequent catalytic hydrogenation step using Pd(OH)2 under hydrogen pressure (e.g., 5 bar) is utilized to saturate the bond. This two-stage approach—alkylation followed by optional hydrogenation—allows for fine-tuning of the impurity profile. The final conversion to the hydrochloride salt is achieved using ethanolic hydrogen chloride, which induces crystallization. This crystallization step is vital for impurity rejection, ensuring that the final API intermediate meets the rigorous purity specifications demanded by regulatory bodies. The ability to control the solid-state form through salt formation is a key advantage for long-term stability and bioavailability.
How to Synthesize Ivabradine Hydrochloride Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent quality to maximize the benefits of the novel route. The process begins with the preparation of the chloro-amine intermediate, followed by the crucial coupling with the benzazepinone core. Detailed operational parameters, including specific molar ratios and workup procedures involving pH adjustments and silica gel chromatography, are essential for achieving the reported high purity levels. Manufacturers looking to adopt this technology should note that the purification steps, such as the extraction with 2N HCl and subsequent basification, are designed to remove unreacted starting materials effectively. For a comprehensive guide on executing this protocol with precision, the detailed standardized synthesis steps are outlined below.
- Prepare the key intermediate by reacting (1S)-4,5-dimethoxy-1-(methylaminomethyl)benzocyclobutane with 1-bromo-3-chloropropane using potassium carbonate.
- Perform the core coupling reaction by alkylating 7,8-dimethoxy-1,3,4,5-tetrahydro-2H-3-benzazepin-2-one with the prepared intermediate using potassium tert-butoxide in DMSO.
- Complete the synthesis by converting the free base into its hydrochloride salt using ethanolic hydrogen chloride, followed by crystallization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain leaders focused on resilience and efficiency. The primary advantage lies in the substantial improvement in overall yield compared to legacy methods. By moving away from processes that yield less than 17% of the target material, manufacturers can significantly reduce the volume of raw materials required per kilogram of finished product. This efficiency gain directly correlates to a reduction in the cost of goods sold (COGS), allowing for more competitive pricing in the global market. Additionally, the use of common, non-proprietary reagents like potassium tert-butoxide and DMSO ensures that the supply chain is not vulnerable to bottlenecks associated with exotic or scarce catalysts.
- Cost Reduction in Manufacturing: The elimination of low-yielding steps and the avoidance of complex, multi-stage purifications inherent in older patents lead to a leaner manufacturing process. By achieving higher conversion rates in the alkylation step, the burden on downstream purification equipment is lessened, reducing energy consumption and solvent waste disposal costs. This streamlined workflow means that production facilities can achieve higher throughput without requiring capital-intensive expansions, thereby driving down the unit cost of the API intermediate significantly.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which often proceed effectively at room temperature, reduces the risk of batch-to-batch variability caused by thermal control issues. This reliability is crucial for maintaining continuous supply to downstream formulation partners. Furthermore, the intermediates generated are stable and can be stored or transported with minimal degradation risk, providing greater flexibility in logistics planning and inventory management for global distribution networks.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as liquid-liquid extraction and crystallization that are well-understood in industrial chemistry. The reduction in waste generation due to higher yields aligns with modern green chemistry principles, simplifying environmental compliance and waste treatment protocols. This makes the technology not only economically attractive but also environmentally sustainable, a key factor for modern pharmaceutical supply chains aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ivabradine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis method improve upon previous patents like EP 0 534 859?
A: Previous methods described in EP 0 534 859 suffered from extremely low overall yields of less than 17% after three steps. The novel process disclosed in CN102498102A utilizes a more efficient alkylation strategy with optimized bases and solvents, significantly boosting the overall yield and purity of the final Ivabradine hydrochloride.
Q: What are the critical quality parameters achieved in this synthetic route?
A: The process demonstrates robust control over impurities, achieving HPLC purity levels of 98% for the intermediate oil before salt formation. The final hydrochloride salt is obtained as white crystals with a sharp melting point range of 125-128°C, indicating high chemical stability and consistency suitable for pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the methodology relies on standard industrial reagents such as potassium tert-butoxide and dimethyl sulfoxide (DMSO) and operates primarily at room temperature. This eliminates the need for complex cryogenic conditions or exotic catalysts, making the commercial scale-up of complex pharmaceutical intermediates significantly more feasible and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ivabradine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. We are committed to delivering high-purity Ivabradine intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify every batch. Our capability to handle complex chemistries, such as the base-mediated alkylation and catalytic hydrogenation described in CN102498102A, positions us as a strategic partner for your long-term supply needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project goals. Let us help you secure a stable, high-quality supply of Ivabradine intermediates for your next generation of pharmaceutical products.
