Scalable Manufacturing of Ivabradine Intermediates via Optimized Catalytic Hydrogenation
The pharmaceutical industry continuously seeks robust synthetic routes for cardiovascular therapeutics, and the preparation of Ivabradine (S 16257-2) represents a critical area of innovation for reducing heart rate in ischemic conditions. Patent CN101768116A discloses a highly efficient preparation method that fundamentally alters the manufacturing landscape for this active pharmaceutical ingredient (API) and its intermediates. This novel approach replaces traditional, labor-intensive purification techniques with a streamlined catalytic hydrogenation process followed by a direct coupling reaction. By eliminating the need for column chromatography and high-pressure autoclaves, this technology offers a pathway to significantly lower production costs while maintaining stringent quality standards required for global regulatory compliance. The core innovation lies in the ability to perform the key reduction step under mild, atmospheric conditions, thereby enhancing operational safety and equipment longevity in large-scale facilities.
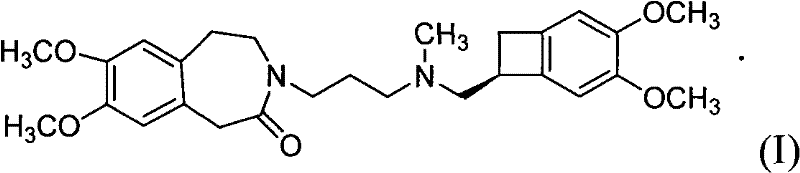
For procurement managers and supply chain directors, the implications of this patent are profound, as it addresses the twin challenges of cost reduction in pharmaceutical intermediates manufacturing and the reliability of supply chains. The method described allows for the direct synthesis of the target compound from readily available precursors, minimizing the number of unit operations and the associated logistical overhead. This efficiency translates into a more resilient supply chain capable of meeting the demands of multinational pharmaceutical companies without the bottlenecks typically associated with complex multi-step syntheses. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advantages is essential for projecting long-term availability and pricing stability for clients dependent on Ivabradine for their cardiovascular drug portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing S 16257-2, such as those described in European patent EP 0534859, suffered from significant industrial drawbacks that hindered cost-effective mass production. The most glaring limitation was the reliance on column chromatography for purification, a technique that is notoriously difficult to scale beyond laboratory quantities due to excessive solvent consumption and low throughput. Furthermore, alternative routes reported in Chinese patent CN200510051779.0 utilized noble metal palladium charcoal catalysts under high-pressure conditions, necessitating expensive specialized reactor vessels and rigorous safety protocols. These high-pressure hydrogenation steps not only increased capital expenditure (CAPEX) for manufacturing plants but also introduced potential safety hazards and longer cycle times due to the pressurization and depressurization phases. Consequently, the overall yield was often compromised by the complexity of the workup procedures, leading to higher waste generation and elevated environmental compliance costs.
The Novel Approach
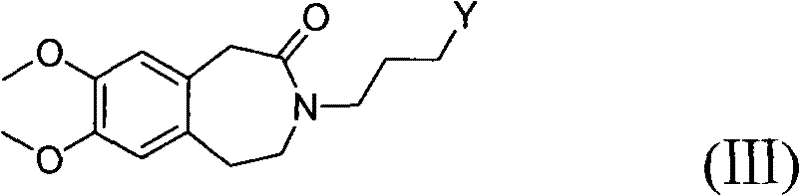
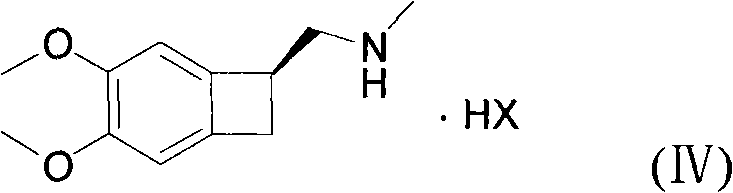
In stark contrast, the method disclosed in CN101768116A introduces a paradigm shift by utilizing catalytic hydrogenation at normal atmospheric pressure, typically ranging from 1 to 10 atm but preferably at 1 atm. This modification drastically simplifies the reaction engineering requirements, allowing the use of standard glass-lined or stainless steel reactors rather than specialized high-pressure autoclaves. The process involves the hydrogenation of a Formula (II) compound to generate a Formula (III) intermediate, which is then directly coupled with a Formula (IV) amine salt without the need for intermediate isolation. This telescoping of steps not only improves the overall mass balance and yield but also significantly reduces the volume of organic solvents required, aligning with green chemistry principles. The elimination of column chromatography in favor of simple filtration and recrystallization represents a major breakthrough in process intensification, making the commercial scale-up of complex pharmaceutical intermediates far more economically viable.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and N-Alkylation
The core of this synthetic strategy relies on the selective reduction of the olefinic double bond within the benzazepine ring system of the Formula (II) precursor. As illustrated in the reaction scheme below, the use of a heterogeneous catalyst such as 10% Palladium on Carbon (Pd/C) facilitates the addition of hydrogen across the double bond under mild thermal conditions, typically between 20°C and 60°C. The preference for methanol as the solvent in this step is driven by its ability to dissolve the substrate effectively while maintaining compatibility with the hydrogenation catalyst. The mechanism likely involves the adsorption of both hydrogen gas and the alkene substrate onto the metal surface, followed by sequential hydrogen transfer to form the saturated Formula (III) compound. This saturation is crucial as it establishes the correct stereochemical and conformational properties required for the subsequent biological activity of the final drug molecule.

Following the hydrogenation, the reaction mixture undergoes a base-catalyzed nucleophilic substitution with the chiral cyclobutane amine derivative, Formula (IV). The presence of inorganic weak bases, specifically anhydrous potassium carbonate, serves to deprotonate the hydroxyl group or activate the leaving group on the side chain, facilitating the attack by the amine nitrogen. The addition of potassium iodide (KI) acts as a catalytic promoter, likely functioning through a Finkelstein-type mechanism to enhance the leaving group ability of halides if present, or simply increasing the ionic strength to accelerate the reaction kinetics in acetone solvent. This coupling step is highly sensitive to moisture and acid levels, necessitating the use of anhydrous conditions to prevent hydrolysis of the sensitive intermediates. The final product is obtained as a pharmaceutically acceptable acid addition salt, typically the hydrochloride, which precipitates upon treatment with HCl and recrystallization, ensuring high optical and chemical purity.
How to Synthesize Ivabradine Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation, particularly during the hydrogenation and coupling phases. Operators must ensure that the hydrogen feed is maintained at a steady rate to keep the catalyst active without causing excessive foaming or safety issues, while the temperature is strictly regulated to prevent side reactions such as over-reduction or decomposition of the methoxy groups. The detailed standardized synthesis steps见下方的指南 outline the specific stoichiometric ratios and workup procedures necessary to achieve the reported 85% yield in the hydrogenation step and 75% in the coupling step. Adherence to these protocols ensures that the final API meets the rigorous specifications demanded by international pharmacopoeias.
- Perform catalytic hydrogenation on the unsaturated benzazepine precursor (Formula II) using Pd/C in methanol at 30-40°C and normal pressure to obtain the saturated intermediate (Formula III).
- React the crude Formula III intermediate directly with the cyclobutane amine salt (Formula IV) in acetone using anhydrous potassium carbonate and potassium iodide as catalysts.
- Filter off the catalyst, adjust pH, and recrystallize the resulting oil from a methanol and ethyl acetate mixture to isolate the pure Ivabradine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers substantial benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply security. By transitioning away from high-pressure processing and chromatographic purification, manufacturers can realize significant cost savings in both utility consumption and raw material usage. The simplified workflow reduces the dependency on specialized equipment vendors and lowers the barrier to entry for secondary suppliers, thereby fostering a more competitive and resilient market environment. For supply chain heads, this means reduced lead time for high-purity pharmaceutical intermediates and a lower risk of production stoppages due to equipment failure or regulatory audits related to high-pressure operations.
- Cost Reduction in Manufacturing: The elimination of high-pressure autoclaves and column chromatography columns results in a drastic reduction in capital investment and operating expenses. Without the need for expensive silica gel and large volumes of elution solvents, the variable cost per kilogram of the product is significantly lowered. Furthermore, the ability to use crude intermediates directly in the second step minimizes material loss and labor costs associated with isolation and drying, creating a leaner manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The use of common, non-hazardous reagents such as potassium carbonate and acetone ensures that raw material sourcing is stable and unaffected by geopolitical fluctuations often seen with exotic catalysts or solvents. The robustness of the atmospheric pressure reaction allows for continuous or semi-continuous processing modes, which can dramatically increase throughput capacity compared to batch-wise high-pressure reactions. This reliability is critical for maintaining uninterrupted supply to downstream formulation partners who depend on just-in-time delivery models for their finished dosage forms.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of complex pressure dynamics and heat transfer limitations associated with exothermic high-pressure hydrogenations. The reduction in solvent waste and the avoidance of heavy metal contamination (beyond the easily filtered Pd/C) simplify wastewater treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also aligns with the sustainability goals of major pharmaceutical customers, adding value to the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and practical manufacturing experience. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing production lines or sourcing strategies. The answers provided reflect the specific conditions and advantages outlined in the intellectual property documentation.
Q: What are the primary advantages of this hydrogenation method over prior art?
A: Unlike previous methods requiring high-pressure vessels or tedious column chromatography, this process operates at normal atmospheric pressure (1 atm) and avoids chromatographic purification, significantly simplifying industrial scale-up and reducing solvent consumption.
Q: Which catalysts are suitable for the reduction of Formula II?
A: The patent specifies that palladium, platinum, nickel, or rhodium can be used, with 10% Palladium on Carbon (Pd/C) being the preferred catalyst due to its high activity and ease of removal via filtration.
Q: How is the purity of the final S 16257-2 hydrochloride ensured?
A: High purity is achieved through a telescoped reaction sequence that minimizes intermediate handling losses, followed by a specific recrystallization step using a mixed solvent system of methanol and ethyl acetate or dichloromethane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ivabradine Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthesis technology to the global market. Our state-of-the-art facilities are equipped to handle the specific requirements of catalytic hydrogenation and sensitive coupling reactions, ensuring that every batch of Ivabradine intermediate meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we provide a secure source for high-quality cardiovascular intermediates that support the development of life-saving medications worldwide.
We invite you to contact our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in pharmaceutical innovation.
