Advanced Synthesis of Gimeracil Intermediates: A Safer, High-Purity Route for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the production of critical oncology drug components, and the recent disclosure in patent CN115504904A offers a transformative approach to synthesizing Gimeracil intermediates. Gimeracil, chemically known as 5-chloro-4-hydroxy-2(1H)-pyridone, serves as a vital dihydropyrimidine dehydrogenase (DPD) inhibitor used in the oral anticancer drug S-1, enhancing the efficacy of tegafur by maintaining therapeutic concentrations of 5-fluorouracil in plasma and tumor tissues. The structural integrity and purity of its precursors are paramount for drug safety, yet traditional manufacturing methods have long been plagued by toxicity and operational hazards. This patent introduces a novel intermediate compound, designated as I-1, which serves as a superior precursor to the key molecule 3-cyano-4-methoxy-2(1H)-pyridone, fundamentally altering the synthetic landscape for this high-value pharmaceutical ingredient.
The significance of this technological breakthrough lies in its ability to bypass the severe limitations inherent in legacy synthesis routes, offering a streamlined path that aligns with modern green chemistry principles while ensuring robust supply chain continuity. By utilizing alpha-cyanoacetamide and orthoesters as starting materials, the process effectively sidesteps the use of highly toxic malononitrile and the volatile, noxious reagent DMF-DMA, which have historically posed significant risks to operator safety and environmental compliance. For R&D directors and procurement managers alike, this shift represents not merely a chemical optimization but a strategic advantage in securing a reliable, high-purity supply of complex heterocyclic intermediates essential for next-generation cancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3-cyano-4-methoxy-2(1H)-pyridone has relied heavily on a condensation reaction involving malononitrile, trimethyl orthoacetate, and N,N-dimethylformamide dimethyl acetal (DMF-DMA), a pathway extensively documented in literature such as Archiv.der.Pharmazie (1985). While chemically feasible, this conventional route suffers from profound drawbacks that compromise both operational safety and product quality, primarily due to the generation of dimethylamine gas during the cyclization step. This toxic byproduct, with a boiling point of merely 9°C, presents a severe inhalation hazard and requires sophisticated scrubbing systems to manage, while simultaneously reacting with the acetic acid solvent to form acetate salts that precipitate out of the solution. These salt impurities necessitate cumbersome and resource-intensive washing and slurring steps to achieve acceptable purity levels, leading to significant yield losses and increased wastewater treatment burdens that inflate the overall cost of goods sold.
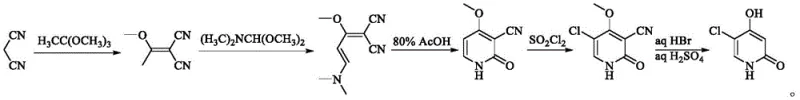
The Novel Approach
In stark contrast, the methodology outlined in patent CN115504904A introduces a paradigm shift by employing alpha-cyanoacetamide and triethyl orthoformate as the core building blocks, effectively eliminating the sources of toxicity and impurity found in the prior art. This innovative strategy utilizes triethyl orthoformate as a 'one-carbon unit' donor, which decomposes cleanly to produce ethanol during the cyclization process rather than hazardous amine gases, thereby creating a significantly safer working environment for plant personnel. Furthermore, the absence of dimethylamine prevents the formation of stubborn acetate salts, meaning the crude product requires far less downstream processing to reach high-purity specifications, directly translating to simplified unit operations and reduced solvent consumption. This streamlined approach not only enhances the environmental profile of the manufacturing process but also ensures a more consistent and reliable supply of the critical intermediate, addressing key pain points for supply chain managers concerned with regulatory compliance and production stability.
Mechanistic Insights into Orthoester-Mediated Cyclization
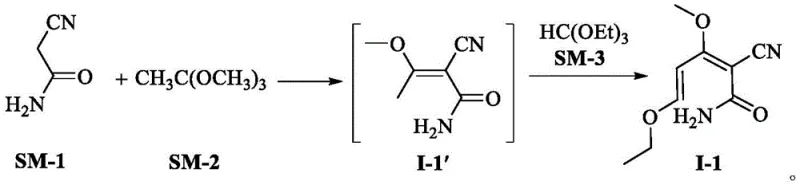
The core of this technological advancement resides in the unique reactivity of alpha-cyanoacetamide when subjected to sequential condensation with orthoesters under controlled thermal conditions, a mechanism that facilitates the construction of the pyridone ring with exceptional regioselectivity. The process begins with the reaction of alpha-cyanoacetamide (SM-1) and trimethyl orthoacetate (SM-2) at moderate temperatures ranging from 65°C to 70°C, forming an initial condensed species that is subsequently reacted with triethyl orthoformate (SM-3) at slightly elevated temperatures of 80°C to 85°C. This two-stage thermal protocol drives the formation of the novel intermediate I-1, a stable precursor that possesses the necessary electronic configuration to undergo smooth cyclization without the side reactions typical of malononitrile-based routes. The use of orthoesters as electrophilic partners allows for the precise introduction of methoxy and ethoxy groups which are later manipulated to close the ring, demonstrating a sophisticated control over the molecular architecture that is crucial for minimizing impurity profiles.
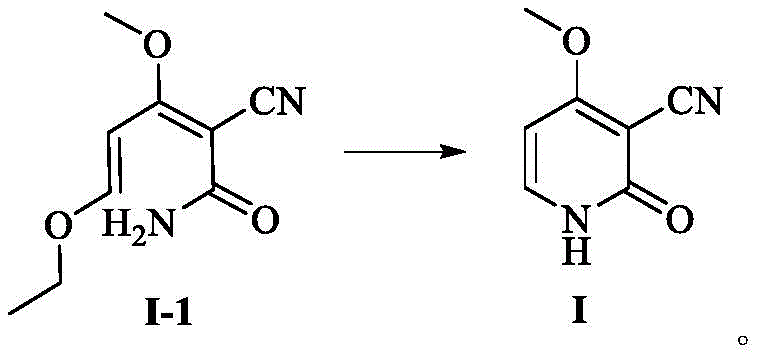
Following the formation of intermediate I-1, the subsequent cyclization to the target 3-cyano-4-methoxy-2(1H)-pyridone is achieved through a base-mediated intramolecular condensation that leverages the nucleophilicity of the amide nitrogen. By dissolving intermediate I-1 in an alcoholic solvent such as ethanol and adjusting the pH to a highly alkaline range of 12 to 14 using inorganic bases like sodium hydroxide, the molecule undergoes a conformational change that promotes ring closure at temperatures between 60°C and 85°C. This mechanistic pathway is particularly advantageous because the byproduct of this cyclization is simply ethanol, a benign solvent that does not interfere with the crystallization of the final product, unlike the salt-forming byproducts of the DMF-DMA route. The final acidification step to pH 6-7 triggers the precipitation of the product as light yellow needle-like crystals, a physical form that indicates high crystallinity and purity, confirming that the mechanistic design successfully avoids the entrapment of impurities within the crystal lattice.
How to Synthesize 3-cyano-4-methoxy-2(1H)-pyridone Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, heating, and pH adjustment that is highly amenable to standard reactor configurations found in most fine chemical facilities. Operators begin by combining the starting materials at room temperature and gradually ramping the heat to facilitate the condensation reactions, monitoring the progress to ensure complete conversion before proceeding to the concentration step. Once the intermediate I-1 is isolated, it is redissolved in a preferred alcohol solvent, and the pH is carefully modulated using aqueous base solutions to initiate the ring-closing reaction, followed by cooling and acidification to harvest the solid product. Detailed standardized operating procedures for scaling this specific pathway from laboratory benchtop to commercial production volumes are provided in the structured guide below, ensuring reproducibility and safety at every stage of the manufacturing process.
- Condense alpha-cyanoacetamide with trimethyl orthoacetate at 65-70°C, then react with triethyl orthoformate at 80-85°C to form Intermediate I-1.
- Dissolve Intermediate I-1 in an alcohol solvent (e.g., ethanol) and adjust pH to 12-14 using an inorganic base like sodium hydroxide.
- Heat the mixture to 60-85°C for cyclization, then cool and acidify to pH 6-7 to precipitate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers substantial strategic benefits that extend far beyond simple chemical yield improvements, fundamentally reshaping the cost structure and risk profile of Gimeracil intermediate manufacturing. By eliminating the need for hazardous reagents like DMF-DMA and toxic starting materials like malononitrile, the process significantly reduces the regulatory burden and insurance costs associated with handling dangerous chemicals, while simultaneously simplifying the waste management protocols required for compliant disposal. The removal of the salt-removal washing steps translates directly into reduced water usage and shorter cycle times, allowing for higher throughput in existing reactor trains without the need for capital-intensive equipment upgrades. Furthermore, the reliance on commercially abundant and stable orthoesters ensures a robust supply of raw materials that is less susceptible to the market volatility often seen with specialized nitriles, thereby enhancing the overall resilience of the supply chain against disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic malononitrile in favor of alpha-cyanoacetamide, combined with the removal of complex salt-removal purification steps, leads to a drastic simplification of the production workflow that inherently lowers operational expenditures. Without the need for extensive washing, filtration, and drying cycles to remove acetate salts, the consumption of solvents and utilities is significantly curtailed, resulting in a leaner and more cost-effective manufacturing process. Additionally, the higher purity of the crude product reduces the loss of material during purification, maximizing the yield of saleable product per batch and improving the overall economic efficiency of the plant.
- Enhanced Supply Chain Reliability: The use of stable, non-volatile starting materials such as alpha-cyanoacetamide and orthoesters mitigates the risks associated with the storage and transport of hazardous chemicals, ensuring a smoother and more predictable logistics operation. Since the process does not generate toxic gases that require specialized scrubbing infrastructure, it can be implemented in a wider range of manufacturing facilities, diversifying the potential supplier base and reducing dependency on single-source vendors with niche capabilities. This flexibility is crucial for maintaining continuous supply to downstream API manufacturers, preventing bottlenecks that could delay the production of life-saving oncology medications.
- Scalability and Environmental Compliance: The generation of ethanol as the primary byproduct aligns perfectly with green chemistry initiatives, drastically reducing the environmental footprint of the manufacturing process and simplifying the permitting process for facility expansion. The absence of noxious amine emissions means that the process is inherently safer for scale-up, removing a major barrier to increasing production capacity to meet growing global demand for Gimeracil. This environmental compatibility not only future-proofs the supply chain against tightening regulations but also enhances the corporate sustainability profile of the manufacturing partner, a key factor for multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity on its advantages over traditional methods and its suitability for large-scale production. These insights are derived directly from the experimental data and technical disclosures within patent CN115504904A, offering a factual basis for decision-making regarding technology adoption and supplier qualification. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this new intermediate into their existing supply networks.
Q: Why is the new synthesis route for Gimeracil intermediates considered safer than conventional methods?
A: The new route replaces toxic malononitrile with alpha-cyanoacetamide and eliminates DMF-DMA, thereby preventing the release of hazardous dimethylamine gas and avoiding the formation of difficult-to-remove acetate salts.
Q: What are the purity levels achievable with this novel intermediate synthesis method?
A: Experimental data from the patent indicates that the target compound 3-cyano-4-methoxy-2(1H)-pyridone can be achieved with HPLC purity exceeding 99%, significantly reducing the need for extensive downstream purification.
Q: How does this process impact environmental compliance in pharmaceutical manufacturing?
A: By generating ethanol as the primary byproduct instead of toxic amine gases and reducing the need for water washing to remove salts, the process drastically lowers the environmental footprint and wastewater treatment burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gimeracil Intermediate Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this innovative patent technology into a robust, commercial-scale reality for global pharmaceutical clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-cyano-4-methoxy-2(1H)-pyridone meets the exacting standards required for oncology drug applications.
We invite forward-thinking procurement leaders to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements, along with access to specific COA data and route feasibility assessments tailored to your project timelines. Partnering with us ensures not only access to high-quality intermediates but also a collaborative relationship focused on continuous improvement and long-term supply security.
