Optimizing Clopidogrel Hydrogen Sulfate Form II Synthesis for Industrial Scale and Stability
Optimizing Clopidogrel Hydrogen Sulfate Form II Synthesis for Industrial Scale and Stability
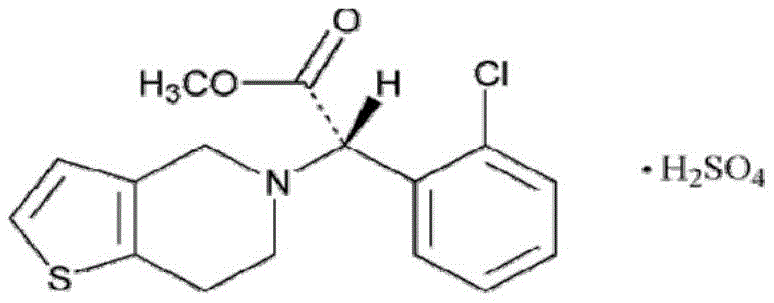
The pharmaceutical landscape for cardiovascular therapeutics demands not only efficacy but also exceptional stability and purity in active pharmaceutical ingredients. Patent CN109867684B introduces a groundbreaking preparation method for II-type clopidogrel hydrogen sulfate, addressing critical stability issues inherent in earlier generations of this antiplatelet agent. Unlike conventional methods that often yield products susceptible to oxidation and discoloration during storage, this novel approach ensures the production of a highly stable crystalline form that strictly adheres to pharmacopoeia standards. The innovation lies in a streamlined synthetic pathway that begins with R-o-chloromandelic acid and employs a unique solvent management strategy to maximize efficiency. By integrating chiral resolution directly into the process flow, manufacturers can achieve superior enantiomeric purity while minimizing the environmental footprint associated with solvent waste. This technical advancement represents a significant leap forward for reliable pharmaceutical intermediates supplier networks seeking to secure high-quality supply chains for generic drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of clopidogrel hydrogen sulfate has been plagued by complex multi-solvent systems and stability challenges that complicate large-scale manufacturing. Traditional routes often involve switching between different organic solvents for esterification, sulfonylation, and substitution steps, necessitating energy-intensive concentration and drying processes between each stage. This fragmentation not only inflates production costs through excessive solvent consumption but also increases the risk of product degradation due to prolonged exposure to heat and air during solvent removal. Furthermore, many legacy methods struggle to control impurity profiles effectively, leading to final products that may discolor over time or fail to meet stringent regulatory specifications for optical purity. The reliance on toxic solvents and the generation of substantial waste liquid further exacerbate the environmental compliance burden for chemical manufacturers, making these older processes less viable in a modern, sustainability-focused industrial context.
The Novel Approach
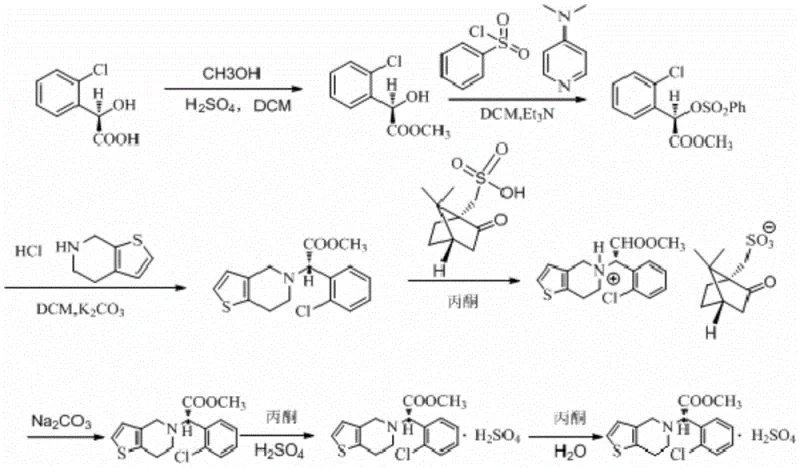
The methodology disclosed in the patent revolutionizes this workflow by implementing a continuous single-solvent strategy for the initial synthesis stages. By utilizing dichloromethane as the exclusive reaction medium for the first three steps—esterification, sulfonylation, and nucleophilic substitution—the process eliminates the need for intermediate solvent exchanges. The reaction mixture from the esterification of R-o-chloromandelic acid is directly utilized in the subsequent sulfonylation with benzenesulfonyl chloride, and the resulting solution is immediately subjected to substitution with tetrahydrothienopyridine hydrochloride. This seamless integration drastically reduces processing time and solvent loss, allowing for a solvent recovery rate exceeding 75 percent. The result is a robust, scalable process that delivers high-purity clopidogrel free base with minimal operational complexity, setting a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Chiral Resolution and Salt Formation
The core of this synthesis relies on precise stereochemical control to ensure the biological activity of the final API. The process initiates with the acid-catalyzed esterification of R-o-chloromandelic acid, where concentrated sulfuric acid facilitates the formation of the methyl ester intermediate under mild thermal conditions ranging from 40 to 60°C. Following this, the introduction of benzenesulfonyl chloride in the presence of an organic base like triethylamine and a catalyst such as DMAP activates the benzylic position for nucleophilic attack. This activation is critical for the subsequent displacement reaction with the thienopyridine moiety. The mechanistic elegance continues in the chiral resolution step, where the racemic clopidogrel free base is treated with L-camphorsulfonic acid. This diastereomeric salt formation selectively precipitates the desired S-enantiomer, leveraging the differences in solubility between the diastereomers in an acetone and ethyl acetate mixture. This resolution step is pivotal for achieving the high optical purity required for therapeutic efficacy, effectively filtering out the inactive R-isomer before the final salt formation.
Impurity control is meticulously managed through temperature regulation and specific washing protocols throughout the synthesis. During the sulfonylation phase, maintaining the reaction temperature between -15 and 5°C suppresses side reactions that could lead to racemization or over-sulfonylation. The subsequent washing steps, including acid washes to remove excess base and water washes to eliminate inorganic salts, ensure that the reaction matrix remains clean prior to the critical substitution step. In the final stages, the conversion of the S-clopidogrel free base to the hydrogen sulfate salt is performed under an inert nitrogen atmosphere to prevent oxidative degradation. The use of activated carbon decolorization prior to crystallization further refines the product, removing trace colored impurities that could compromise the visual quality of the API. This comprehensive approach to impurity management ensures that the final high-purity pharmaceutical intermediates meet the rigorous demands of global regulatory bodies.
How to Synthesize Clopidogrel Hydrogen Sulfate Efficiently
Implementing this synthesis requires strict adherence to the optimized molar ratios and thermal profiles defined in the patent to maximize yield and purity. The process is designed to be telescoped where possible, particularly in the early stages, to leverage the benefits of the single-solvent system. Operators must carefully monitor the pH adjustments during the workup phases, specifically using sodium bicarbonate to neutralize acidic byproducts without inducing hydrolysis of the ester linkage. The resolution step demands precise control over the acetone to ethyl acetate ratio, as this dictates the crystallization efficiency of the camphorsulfonate salt. For a detailed breakdown of the specific operational parameters, reagent quantities, and safety precautions required for execution, please refer to the standardized protocol below.
- Esterify R-o-chloromandelic acid with methanol in dichloromethane using sulfuric acid catalyst at 40-60°C.
- React the resulting ester with benzenesulfonyl chloride and organic base at -15 to 5°C without changing solvents.
- Perform nucleophilic substitution with tetrahydrothienopyridine hydrochloride and potassium carbonate at 30-50°C.
- Resolve the racemic mixture using L-camphorsulfonic acid in acetone/ethyl acetate to isolate the S-enantiomer.
- Form the hydrogen sulfate salt using concentrated sulfuric acid in acetone under inert atmosphere.
- Recrystallize the crude product from an acetone-water mixture to obtain the final high-purity Form II crystal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this preparation method offers tangible logistical and financial benefits that extend beyond simple yield metrics. The consolidation of three major reaction steps into a single solvent system fundamentally alters the cost structure of production by reducing the volume of raw materials required and simplifying the waste management infrastructure. This simplification translates directly into enhanced supply chain reliability, as the dependency on multiple specialized solvents is minimized, reducing the risk of shortages or price volatility affecting the production schedule. Furthermore, the high stability of the resulting Form II crystal reduces the need for specialized storage conditions or rapid turnover, allowing for more flexible inventory management and longer shelf life for the finished API.
- Cost Reduction in Manufacturing: The elimination of intermediate concentration and solvent exchange steps significantly lowers energy consumption and labor hours associated with distillation and drying processes. By recovering and recycling over 75 percent of the dichloromethane solvent, the facility reduces its raw material expenditure and waste disposal fees, leading to substantial overall cost savings. The streamlined workflow also increases equipment throughput, allowing the same reactor volume to produce more batches over a given period, thereby optimizing capital asset utilization without requiring additional infrastructure investment.
- Enhanced Supply Chain Reliability: The use of readily available starting materials like R-o-chloromandelic acid and common reagents such as benzenesulfonyl chloride ensures a stable supply of inputs that are not subject to the geopolitical or logistical constraints often associated with exotic catalysts. The robustness of the process against minor variations in conditions means that production can be scaled across different manufacturing sites with consistent results, mitigating the risk of batch failures that could disrupt the supply of critical cardiovascular medication to the market.
- Scalability and Environmental Compliance: The reduction in total solvent usage and the high recovery rate align perfectly with green chemistry principles, making it easier for manufacturers to meet increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The process generates less hazardous waste liquid, simplifying the effluent treatment requirements and lowering the environmental compliance burden. This eco-friendly profile not only reduces operational risk but also enhances the corporate sustainability credentials of the manufacturing entity, which is increasingly important for partnerships with major global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's value proposition. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production.
Q: Why is Clopidogrel Hydrogen Sulfate Form II preferred over other forms?
A: Form II offers superior stability compared to traditional forms which are prone to oxidation and discoloration (turning yellow or red) during storage. This method ensures the product meets strict pharmacopoeia clarity requirements and maintains high purity over long-term storage.
Q: How does this preparation method reduce manufacturing costs?
A: The process utilizes a single solvent system (dichloromethane) for the first three critical reaction steps. By carrying the reaction solution directly to the next step without intermediate concentration or solvent exchange, the method significantly reduces solvent consumption, energy usage for distillation, and waste liquid generation.
Q: What is the achievable purity level using this synthetic route?
A: The optimized route, including specific chiral resolution and recrystallization steps, achieves a crude product purity exceeding 99.9% and a final refined product purity of up to 99.98%, with negligible isomer impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Hydrogen Sulfate Supplier
The technical sophistication of this preparation method underscores the importance of partnering with a manufacturer that possesses deep expertise in complex organic synthesis and chiral resolution. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling the precise temperature controls and inert atmospheres required for this synthesis, ensuring that every batch meets stringent purity specifications. With rigorous QC labs dedicated to monitoring optical rotation, residual solvents, and related substances, we guarantee that our clopidogrel hydrogen sulfate form II consistently exceeds the quality benchmarks set by international pharmacopoeias.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this more efficient manufacturing process for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
