Advanced Asymmetric Hydrogenation Route for High-Purity Clopidogrel Hydrogen Sulfate Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of critical cardiovascular medications, and the synthesis of Clopidogrel Hydrogen Sulfate remains a focal point of process chemistry innovation. Patent CN109574984B introduces a transformative methodology that leverages asymmetric hydrogenation to construct the chiral center of this vital antiplatelet agent with exceptional precision. This technical disclosure outlines a concise synthetic strategy that begins with the condensation of 2-thiophene ethylamine and a specific carbonyl acetamide derivative, followed by a highly enantioselective reduction step. The resulting chiral secondary amine is then subjected to cyclization and salt formation to yield the final active pharmaceutical ingredient. By shifting away from traditional resolution techniques, this approach addresses long-standing inefficiencies in yield and waste generation, positioning itself as a superior alternative for modern API manufacturing.
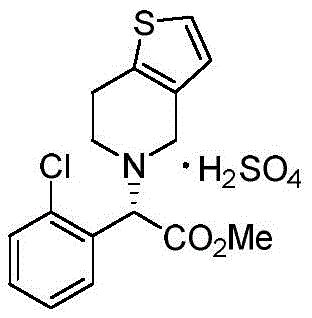
Historically, the manufacturing landscape for Clopidogrel has been dominated by processes that suffer from inherent thermodynamic and economic limitations, particularly those relying on the resolution of racemic mixtures. Conventional Method 1, for instance, involves the synthesis of racemic clopidogrel free base followed by chiral resolution, a process that theoretically discards half of the produced material, leading to substantial raw material wastage and increased disposal costs. Furthermore, alternative routes such as Method 3 often necessitate the use of hazardous reagents like sodium cyanide and require high-pressure hydrolysis conditions that demand specialized, expensive equipment and rigorous safety protocols. These legacy methods not only inflate the cost of goods sold but also introduce significant supply chain vulnerabilities due to the handling of toxic substances and the complexity of purification steps required to meet stringent regulatory purity standards.
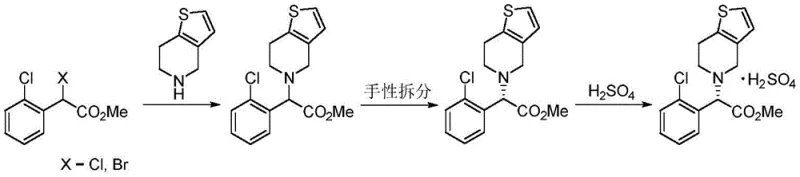
In stark contrast to these cumbersome legacy protocols, the novel approach detailed in the patent utilizes a streamlined sequence of classical organic reactions that are both mild and highly efficient. The core innovation lies in the construction of the chiral amine intermediate via asymmetric hydrogenation of an imine precursor, a step that bypasses the need for resolution entirely. This new route ensures that the stereochemistry is established early in the synthesis with high fidelity, allowing subsequent steps such as the Pictet-Spengler type cyclization with formaldehyde to proceed with minimal risk of racemization. The overall process is characterized by its operational simplicity, utilizing common solvents like alcohols and aromatic hydrocarbons, and avoiding extreme temperatures or pressures, which collectively contribute to a more sustainable and cost-effective manufacturing profile suitable for global supply chains.
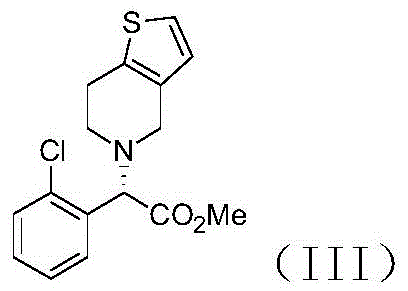
Mechanistic Insights into Iridium and Rhodium Catalyzed Asymmetric Hydrogenation
The heart of this synthetic advancement is the asymmetric hydrogenation step, where an achiral imine substrate is converted into a chiral secondary amine with remarkable enantioselectivity. This transformation is mediated by a catalyst system formed in situ from metal salts such as [Ir(COD)Cl]2 or [Rh(COD)Cl]2 and bulky chiral diphosphine ligands like (R)-BINAP or (R)-f-amphox. The mechanism involves the coordination of the imine nitrogen to the metal center, followed by the migratory insertion of hydride species generated from molecular hydrogen. The steric environment created by the chiral ligand dictates the facial selectivity of the hydrogen addition, ensuring that the (S)-enantiomer is produced predominantly. The patent highlights that this catalytic system is robust enough to achieve conversion rates exceeding 99% and enantiomeric excess values ranging from 97% to 99%, even when the catalyst loading is reduced to as low as one ten-thousandth of the substrate molar amount.

Controlling impurities in this catalytic cycle is critical for meeting pharmaceutical grade specifications, and the described method offers inherent advantages in this regard. The high turnover number (TON) of up to 100,000 implies that the concentration of residual heavy metals in the final product can be kept extremely low, simplifying the downstream purification process. Additionally, because the reaction proceeds under mild conditions (20-30°C) in alcohol solvents, the formation of thermal degradation byproducts or solvent-derived impurities is minimized. The subsequent cyclization step uses paraformaldehyde and acid, which are standard reagents that generate manageable byproducts, further ensuring that the impurity profile of the crude clopidogrel free base is clean. This high level of chemical purity reduces the burden on crystallization and washing steps, ultimately leading to a final sulfate salt that consistently meets rigorous quality control benchmarks without the need for complex chromatographic separations.
How to Synthesize Clopidogrel Hydrogen Sulfate Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the catalyst and the control of reaction parameters during the hydrogenation phase. The process begins with the condensation of the amine and keto-amide in an aromatic solvent with water removal, followed by the critical asymmetric reduction in an alcohol medium under hydrogen pressure. Detailed operational guidelines regarding stoichiometry, temperature ramps, and workup procedures are essential for reproducing the high yields and selectivity reported in the patent literature. For process chemists looking to adopt this technology, understanding the nuances of catalyst activation and the specific order of reagent addition is paramount to success.
- Condense 2-thiophene ethylamine with N-benzyl-2-(2-chlorophenyl)-2-carbonyl acetamide in an aromatic solvent to form the imine intermediate.
- Perform asymmetric hydrogenation on the imine using a chiral metal catalyst (Ir or Rh based) in alcohol solvent to obtain the chiral secondary amine with high ee value.
- Execute acid-catalyzed cyclization with formaldehyde followed by sulfuric acid salt formation to yield the final Clopidogrel Hydrogen Sulfate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this asymmetric hydrogenation route offers profound advantages in terms of cost structure and supply security. By eliminating the chiral resolution step, manufacturers effectively double the theoretical yield of the chiral intermediate from the same amount of starting materials, which translates directly into substantial raw material savings. This improvement in atom economy means that less feedstock is required to produce the same volume of finished API, insulating the supply chain from volatility in the pricing of key precursors like thiophene ethylamine and chlorophenyl derivatives. Furthermore, the avoidance of toxic reagents such as sodium cyanide removes the logistical and regulatory burdens associated with hazardous material transport and storage, streamlining the overall procurement process.
- Cost Reduction in Manufacturing: The elimination of the resolution step fundamentally alters the cost equation by removing the waste of the unwanted enantiomer, which traditionally accounts for a significant portion of material costs in racemic syntheses. Additionally, the high efficiency of the catalyst system allows for ultra-low metal loading, reducing the expense associated with precious metal recovery and waste treatment. The use of common, recyclable solvents like methanol and toluene further contributes to lower operating expenditures compared to processes requiring exotic or difficult-to-recycle solvent systems.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of specialized chiral pool reagents. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or equipment limitations, leading to more predictable production schedules and consistent on-time delivery performance. This reliability is crucial for maintaining continuous inventory levels for downstream formulation partners who depend on steady API supplies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard stainless steel reactors and avoiding extreme cryogenic or high-temperature conditions that limit batch sizes. The absence of highly toxic reagents simplifies wastewater treatment and废气 handling, ensuring compliance with increasingly stringent environmental regulations without the need for capital-intensive abatement technologies. This green chemistry profile enhances the long-term sustainability of the manufacturing site and reduces the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on catalyst selection, reaction conditions, and product quality. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: What are the key advantages of the asymmetric hydrogenation route over traditional resolution methods?
A: The asymmetric hydrogenation route eliminates the theoretical 50% yield loss inherent in chiral resolution processes, significantly improving atom economy and reducing raw material costs while achieving high enantiomeric excess (>99% ee).
Q: What catalyst systems are suitable for this synthesis?
A: The patent specifies the use of Iridium or Rhodium metal salts complexed with chiral ligands such as (R)-BINAP or (R)-f-amphox, which allow for extremely low catalyst loading with high turnover numbers.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes classical organic reactions under mild conditions (20-30°C, moderate hydrogen pressure) and avoids toxic reagents like sodium cyanide, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Hydrogen Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes for high-demand cardiovascular therapeutics like Clopidogrel Hydrogen Sulfate. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of API intermediate meets the highest global pharmacopoeia standards, providing our partners with absolute confidence in product quality.
We invite potential collaborators to engage with our technical procurement team to discuss how this advanced asymmetric hydrogenation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-value chemical solutions that drive your business forward.
