Revolutionizing Clopidogrel Hydrogen Sulfate Production: A Safer, High-Purity Synthetic Route for Global Pharma Supply Chains
Introduction to Patent CN112341475A
The pharmaceutical industry continuously seeks robust manufacturing processes for critical cardiovascular medications, and the preparation of Clopidogrel Hydrogen Sulfate remains a focal point for process chemists worldwide. Patent CN112341475A introduces a groundbreaking synthetic methodology that addresses long-standing challenges in impurity control and operational safety. This innovation centers on a novel silyl-protection strategy that fundamentally alters the reaction kinetics of the key alkylation step. By temporarily masking the reactive amino group of (S)-o-chlorophenyl glycine methyl ester, the process effectively prevents the formation of persistent dialkylated byproducts that have plagued previous generations of synthesis routes. Furthermore, the integration of batch solvent evaporation techniques drives the reaction equilibrium forward, achieving conversion rates exceeding 96 percent while maintaining exceptional stereochemical integrity. For global supply chain leaders, this represents a pivotal shift towards more predictable, high-yield manufacturing of this essential antiplatelet agent.
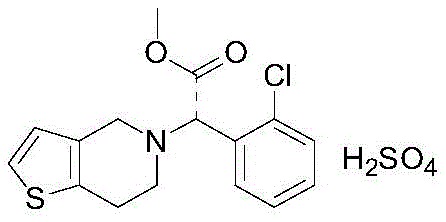
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clopidogrel has been fraught with significant technical and safety hurdles that compromise both cost-efficiency and product quality. Many established routes, such as those depicted in prior art references like Route 1, rely heavily on sodium cyanoborohydride for reductive amination steps. This reagent not only poses severe neurotoxicity risks but also generates hazardous borane gas during post-treatment, necessitating complex scrubbing systems and increasing operational expenditures. Other pathways, exemplified by Route 2 and Route 10, depend on chiral resolution strategies that inherently cap the theoretical yield at 50 percent, leading to massive material wastage and the generation of large volumes of waste containing resolving agents like camphorsulfonic or tartaric acids. Additionally, direct nucleophilic substitution without protection often results in uncontrolled dialkylation, where the secondary amine product reacts further to form difficult-to-remove impurities, drastically lowering the purity profile and complicating downstream purification efforts.
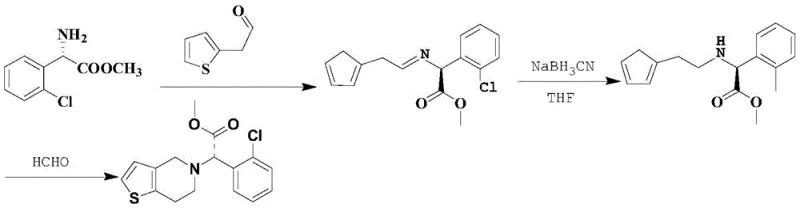
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112341475A employs a sophisticated protection-deprotection sequence that elegantly bypasses these pitfalls. The core innovation involves the initial reaction of (S)-o-chlorophenyl glycine methyl ester with 1,1,1,3,3,3-hexamethyldisilazane (HMDS) to form a trimethylsilyl-protected intermediate. This bulky silyl group acts as a temporary steric shield, preventing the nitrogen atom from undergoing a second alkylation event when exposed to 2-(2-bromoethyl)thiophene. Moreover, the process incorporates a dynamic solvent management system where acetonitrile is evaporated in batches during the reaction. This continuous removal of solvent increases the concentration of reactants, thereby accelerating the reaction rate and pushing the equilibrium towards the desired mono-alkylated product. The result is a streamlined process that eliminates the need for toxic reducing agents and avoids the yield limitations of chiral resolution, delivering a crude intermediate with purity levels suitable for direct crystallization.
Mechanistic Insights into Silyl-Protection and Kinetic Control
The mechanistic superiority of this route lies in its precise control over nucleophilicity and reaction thermodynamics. In traditional unprotected alkylation, the primary amine reacts to form a secondary amine, which often retains sufficient nucleophilicity to attack another molecule of the alkyl halide, leading to the formation of tertiary amine impurities. As illustrated in the impurity mechanism, this dialkylation is a competitive side reaction that is difficult to suppress once initiated. However, by converting the primary amine into a trimethylsilyl derivative, the electron density on the nitrogen is significantly modulated. The silicon-nitrogen bond renders the nitrogen less nucleophilic initially, but under the basic conditions provided by ultrafine potassium carbonate, the silyl group facilitates a controlled substitution. Crucially, the steric bulk of the trimethylsilyl group physically obstructs the approach of a second alkyl halide molecule to the nitrogen center after the first substitution occurs, effectively shutting down the pathway to dialkylated impurities.
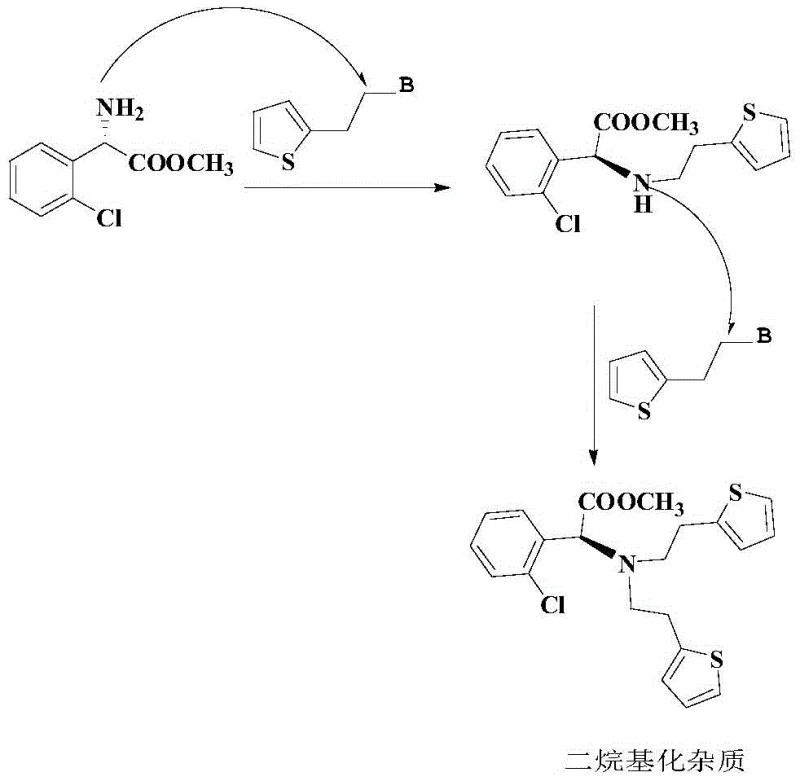
Furthermore, the choice of ultrafine potassium carbonate as the acid scavenger plays a critical role in maintaining optical purity. Standard coarse bases can lead to localized hotspots of high pH or inefficient mixing, which may promote racemization at the chiral alpha-carbon. The ultrafine powder ensures a homogeneous distribution of base throughout the reaction medium, gently neutralizing the generated hydrobromic acid without inducing epimerization. This careful balance allows the reaction to proceed at elevated temperatures of 70-80°C to ensure high conversion, while keeping the optical isomer content below 0.1 percent in the final isolated salt. The subsequent deprotection step using concentrated hydrochloric acid is seamless, as the acidic conditions simultaneously cleave the silyl protecting group and form the stable hydrochloride salt of the intermediate, simplifying the workup procedure and reducing the number of unit operations required.
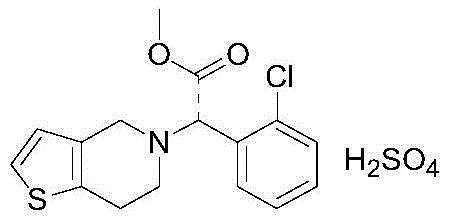
How to Synthesize Clopidogrel Hydrogen Sulfate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly regarding the batch addition of reagents and solvent management. The process begins with the silylation of the amino acid ester, followed by the critical alkylation step where 2-(2-bromoethyl)thiophene is added in multiple batches to maintain a favorable molar ratio of protected amine to alkylating agent. This kinetic control, combined with the periodic evaporation of acetonitrile, ensures that the concentration of the electrophile remains low relative to the nucleophile, further suppressing side reactions. Following the alkylation, the reaction mixture undergoes extraction and acidification to isolate the key intermediate as a hydrochloride salt, which is then subjected to a pulping refinement to remove trace optical isomers. The final step involves a Pictet-Spengler type cyclization with formaldehyde and subsequent salt formation with sulfuric acid. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Protect (S)-o-chlorophenyl glycine methyl ester using 1,1,1,3,3,3-hexamethyldisilazane (HMDS) to form the trimethylsilyl-protected intermediate.
- React the protected intermediate with 2-(2-bromoethyl)thiophene using ultrafine potassium carbonate, employing batch addition and solvent evaporation to minimize dialkylated impurities.
- Deprotect the amine with hydrochloric acid, followed by cyclization with formaldehyde and salt formation with sulfuric acid to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages by mitigating regulatory risks and optimizing raw material utilization. The elimination of highly toxic reagents such as sodium cyanide and sodium cyanoborohydride removes the need for specialized handling protocols and expensive waste treatment facilities associated with cyanide and borane disposal. This transition to safer chemistry not only lowers the environmental compliance burden but also ensures uninterrupted production schedules by avoiding the supply chain volatility often associated with controlled hazardous substances. Additionally, the high conversion rates and superior selectivity of the silyl-protection route mean that less raw material is wasted on byproduct formation, directly translating to improved cost efficiency per kilogram of active pharmaceutical ingredient produced.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by abandoning chiral resolution steps that inherently discard half of the starting material. By utilizing a chiral pool starting material and preserving its stereochemistry throughout the synthesis through mild basic conditions, the overall yield is drastically improved compared to resolution-based routes. Furthermore, the simplified workup procedure, which combines deprotection and salt formation in a single acidic treatment, reduces solvent consumption and processing time, leading to lower utility costs and higher throughput in existing reactor trains without the need for major capital investment.
- Enhanced Supply Chain Reliability: Reliance on exotic or strictly regulated reagents poses a constant threat to supply continuity. This method utilizes commodity chemicals like HMDS, potassium carbonate, and common organic solvents, which are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction against impurity formation means that quality control failures are less frequent, ensuring a consistent flow of high-purity intermediates to downstream formulation partners and minimizing the risk of batch rejection.
- Scalability and Environmental Compliance: The technology is inherently designed for scale-up, utilizing standard heterogeneous reaction conditions that translate well from pilot plant to commercial manufacturing. The reduction in hazardous waste generation, specifically the absence of heavy metal catalysts or toxic cyanide streams, aligns perfectly with modern green chemistry initiatives and stringent environmental regulations. This eco-friendly profile not only simplifies the permitting process for new manufacturing sites but also enhances the corporate sustainability metrics of pharmaceutical companies adopting this supply chain, appealing to increasingly environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy technologies in terms of safety, purity, and efficiency. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does this new method control dialkylated impurities compared to traditional routes?
A: The method utilizes a trimethylsilyl protective group on the amino function, which sterically hinders secondary alkylation. Combined with batch addition of the alkylating agent, this reduces dialkylated impurity content to less than 0.5%, significantly lower than conventional nucleophilic substitution methods.
Q: Does this synthesis route involve toxic reagents like sodium cyanide or sodium borohydride?
A: No. Unlike several prior art routes (such as Route 1 and Route 9) that rely on neurotoxic sodium cyanoborohydride or highly toxic sodium cyanide, this patented process uses safer reagents like HMDS and ultrafine potassium carbonate, eliminating significant safety hazards and waste disposal costs.
Q: What is the optical purity advantage of using ultrafine potassium carbonate in this process?
A: The use of ultrafine potassium carbonate powder enhances the acid-binding efficiency during the alkylation step. This improved kinetics helps suppress the racemization of the chiral center, resulting in an optical isomer content of less than 0.1% in the final purified intermediate, ensuring high enantiomeric excess for the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Hydrogen Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented silyl-protection method are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities and optical isomers at ppm levels, guaranteeing that every batch of Clopidogrel Hydrogen Sulfate meets the highest global pharmacopoeia standards. Our commitment to quality assurance ensures that the high purity and low impurity profiles described in the patent are consistently delivered to our clients.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this safer, higher-yield route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot batches and to discuss route feasibility assessments tailored to your project timelines and regulatory needs.
