Advanced One-Step Deprotection Strategy for High-Purity OSW-1 Manufacturing
Advanced One-Step Deprotection Strategy for High-Purity OSW-1 Manufacturing
The synthesis of complex natural products with potent biological activity often faces significant bottlenecks during the final stages of production, particularly during the removal of protecting groups. Patent CN1844138A introduces a groundbreaking methodology for synthesizing OSW-1, a steroidal saponin with exceptional anticancer properties, by utilizing a one-step deprotection strategy in an aqueous solvent under acid catalysis. This innovation specifically addresses the critical challenges associated with traditional palladium-catalyzed methods, offering a pathway to obtain the natural product OSW-1 with superior purity and safety profiles. By employing aqueous acetic acid as the primary reaction medium, this process effectively removes multiple protecting groups simultaneously without compromising the sensitive structural features of the molecule. For pharmaceutical manufacturers and research institutions, this represents a significant leap forward in process chemistry, enabling the reliable production of high-purity pharmaceutical intermediates while adhering to stringent regulatory standards regarding heavy metal contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deprotection of protected OSW-1 precursors has relied heavily on palladium-based catalytic systems, typically utilizing Pd(MeCN)2Cl2 in acetone and water mixtures. While these Lewis acid catalysts are effective at cleaving certain protecting groups, they introduce severe liabilities for commercial pharmaceutical manufacturing. The primary concern is the potential for heavy metal residue; palladium is toxic and can cause serious health issues including heart damage and organ failure, leading regulatory bodies in Europe and America to mandate strict limits, often below 20ppm, in final drug substances. Furthermore, extended reaction times in these palladium systems can lead to undesirable side reactions, such as the substitution of the 3-hydroxyl group by chloride ions, which significantly reduces the overall yield and complicates the purification process. The necessity for extensive post-reaction processing to scavenge residual palladium adds both cost and complexity, creating a substantial barrier to efficient cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to the metal-dependent legacy methods, the novel approach detailed in the patent utilizes a simple yet highly effective system comprising aqueous acetic acid. This method achieves the removal of multiple protecting groups, including silyl ethers and esters, in a single operational step without the need for any transition metal catalysts. Experimental data demonstrates that this acid-catalyzed system not only matches but often exceeds the efficiency of palladium methods, achieving yields as high as 97.4% compared to 94.7% for the Pd-catalyzed route in model studies. Crucially, the glycosidic bonds within the sugar moieties remain stable under these acidic conditions, preventing the hydrolysis that often plagues harsh deprotection protocols. This shift to a metal-free protocol fundamentally alters the economic and safety landscape of OSW-1 synthesis, providing a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Deprotection
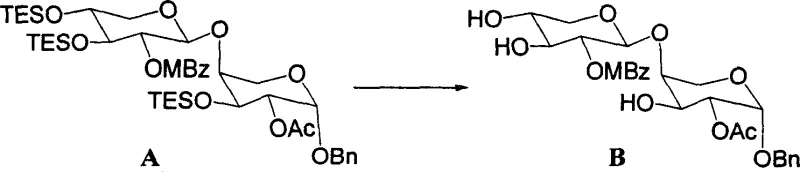
The success of this methodology lies in the precise tuning of acidity and solvent polarity to facilitate the cleavage of specific bonds while preserving others. In the presence of aqueous acetic acid, the protonation of oxygen atoms in silyl ether protecting groups (such as TBS, TES, and TBDPS) increases their electrophilicity, making them susceptible to nucleophilic attack by water molecules. This hydrolytic mechanism proceeds smoothly at moderate temperatures, typically ranging from 45°C to 85°C, ensuring that the energy barrier for deprotection is overcome without imparting excessive thermal stress on the molecule. Unlike Lewis acids which might coordinate unpredictably with various functional groups, the Brønsted acid environment provided by acetic acid offers a more uniform reaction profile. This uniformity is essential for minimizing the formation of byproducts and ensures that the reaction proceeds cleanly to the desired diol or triol products, which is critical for maintaining the rigorous purity specifications required for bioactive compounds.
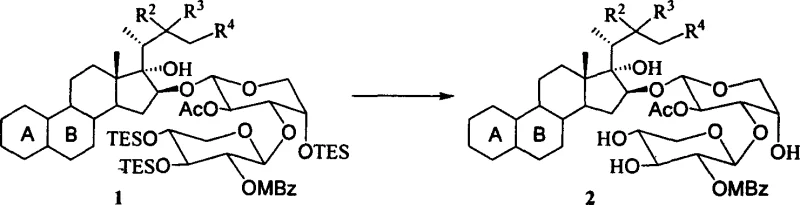
Furthermore, the impurity control mechanism inherent in this acid-catalyzed system is superior to that of halide-containing palladium salts. In conventional Pd-catalyzed reactions, the presence of chloride ions poses a constant threat of nucleophilic substitution at activated carbon centers, particularly at the C3 position of the steroid backbone. By eliminating the source of chloride ions entirely, the aqueous acetic acid method effectively shuts down this degradation pathway. This results in a cleaner crude reaction mixture, which significantly simplifies downstream purification efforts such as column chromatography. For R&D directors focused on process robustness, this means a more predictable impurity profile and a higher probability of passing stringent quality control tests on the first attempt, thereby reducing the time and resources spent on troubleshooting and reprocessing batches.
How to Synthesize OSW-1 Efficiently
The implementation of this deprotection strategy is straightforward and relies on standard laboratory equipment, making it highly accessible for process development teams. The general procedure involves dissolving the fully protected precursor, which typically contains silyl and ester groups on both the aglycone and the sugar moieties, in a mixture of acetic acid and water. The reaction is then heated to a specific temperature, monitored by TLC until the starting material is completely consumed. Following the reaction, the solvent is removed under reduced pressure, and the residue is purified to yield the final product. This streamlined workflow eliminates the need for specialized catalyst handling or complex quenching procedures associated with metal catalysts. For detailed standardized synthetic steps and specific condition optimizations for various analogues, please refer to the guide below.
- Dissolve the fully protected OSW-1 precursor (containing silyl and ester protecting groups) in a mixture of aqueous organic solvent, specifically recommending acetic acid and water.
- Heat the reaction mixture to a temperature range between 45°C and 85°C, maintaining stirring until TLC analysis confirms the complete disappearance of the starting material.
- Remove the solvent under reduced pressure and purify the resulting crude product via flash column chromatography to isolate the final high-purity OSW-1 or its analogues.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition to this acid-catalyzed deprotection method offers profound advantages that extend beyond mere chemical efficiency. The elimination of palladium catalysts removes a significant cost driver, as precious metal prices are volatile and the materials themselves are expensive. Moreover, the reliance on commodity chemicals like acetic acid and water ensures a stable and secure supply chain, free from the geopolitical or mining constraints that often affect rare earth or precious metal catalysts. This stability is crucial for long-term production planning and helps in reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands without the risk of raw material shortages.
- Cost Reduction in Manufacturing: The removal of expensive palladium catalysts directly lowers the bill of materials for each batch. Additionally, the simplified workup process eliminates the need for costly metal scavenging resins or extensive washing protocols designed to meet heavy metal limits. This reduction in processing steps translates to lower labor costs, reduced solvent consumption, and decreased waste disposal fees, collectively driving down the overall cost of goods sold. The qualitative improvement in process efficiency allows for better resource allocation, ensuring that capital is not tied up in unnecessary purification infrastructure.
- Enhanced Supply Chain Reliability: By utilizing reagents that are globally available and produced at massive scales, the risk of supply disruption is minimized. Acetic acid and water are fundamental industrial chemicals with robust logistics networks, unlike specialized organometallic complexes which may have limited suppliers. This reliability ensures continuous production schedules and prevents delays that could arise from waiting for catalyst shipments. For supply chain heads, this means a more resilient manufacturing operation capable of maintaining consistent output levels even during periods of global supply chain volatility.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies environmental compliance and waste management significantly. Disposing of palladium-contaminated waste requires specialized treatment and incurs high regulatory costs. In contrast, the aqueous acidic waste generated by this new method is much easier to neutralize and treat, aligning with green chemistry principles. This environmental friendliness facilitates easier permitting for larger scale facilities and supports corporate sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates more feasible and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel deprotection technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this method for their own production lines or research programs.
Q: Why is the aqueous acetic acid method superior to palladium-catalyzed deprotection for OSW-1?
A: The aqueous acetic acid method eliminates the risk of toxic palladium residue, which is strictly regulated in pharmaceuticals (often <20ppm). Additionally, it avoids side reactions such as chloride substitution at the 3-hydroxyl position that can occur with Pd(MeCN)2Cl2 systems, thereby improving overall yield and purity.
Q: Does this deprotection method affect the stability of the glycosidic bond?
A: No, the optimized conditions using aqueous acetic acid are mild enough to preserve the integrity of the glycosidic bond while effectively cleaving silyl ethers (such as TBS, TES) and ester groups. This selectivity is crucial for maintaining the biological activity of the final saponin product.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses inexpensive, readily available reagents like acetic acid and water, avoiding costly transition metal catalysts. The simplified workup procedure, which does not require complex heavy metal scavenging steps, makes it highly scalable and environmentally compliant for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable OSW-1 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this acid-catalyzed deprotection technology in the production of high-value anticancer intermediates like OSW-1. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities at ppm levels. We are committed to leveraging advanced synthetic methodologies to deliver products that meet the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this metal-free process. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and reliable chemical foundations available today.
