Advanced Cyclization Technology for Commercial Scale Production of Citalopram and Escitalopram Intermediates
Advanced Cyclization Technology for Commercial Scale Production of Citalopram and Escitalopram Intermediates
The pharmaceutical landscape for antidepressant medications continues to evolve, driven by the demand for higher purity active pharmaceutical ingredients (APIs) and more sustainable manufacturing processes. Patent CN101048396A introduces a significant technological breakthrough in the synthesis of Citalopram (Formula I) and its pharmacologically active S(+) enantiomer, Escitalopram (Formula II). This innovation addresses critical bottlenecks in the conventional production of these vital compounds by replacing harsh acidic conditions with a mild, highly selective cyclization protocol. By leveraging a Mitsunobu-like reaction mechanism involving azodicarboxylates and phosphines, this method ensures superior control over reaction parameters, leading to enhanced product quality and process robustness. For global procurement teams and R&D directors, understanding this shift is essential for securing a reliable citalopram supplier capable of meeting stringent regulatory standards.
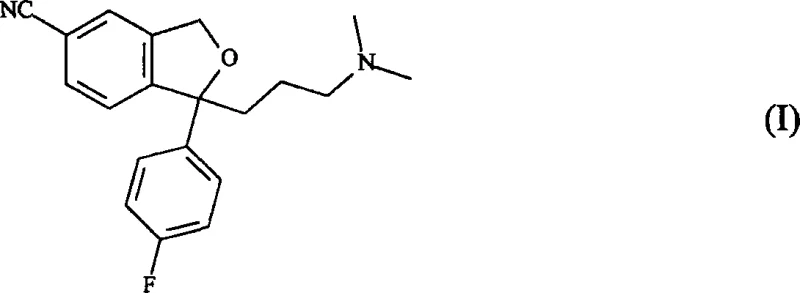
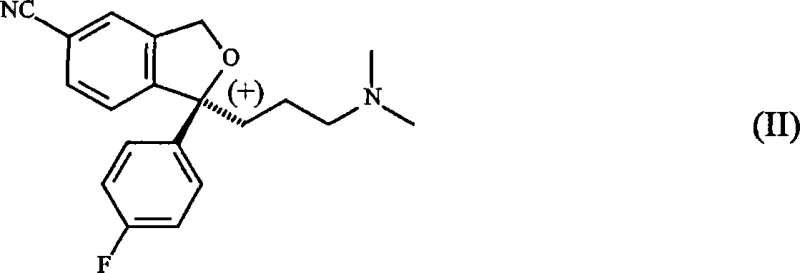
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Citalopram has relied on methodologies that pose significant challenges for large-scale manufacturing and environmental compliance. As documented in prior art such as U.S. Patent No. 4,650,884, the cyclization of the precursor diol was typically achieved using concentrated sulfuric acid (70%) at elevated temperatures around 80°C for extended periods. These aggressive conditions frequently result in substantial substrate degradation, leading to reduced reaction yields and the generation of complex impurity profiles that are difficult to purge. Furthermore, alternative approaches described in documents like EP347066 utilize methanesulfonyl chloride and triethylamine. While milder than sulfuric acid, the use of acid chlorides introduces severe handling hazards due to their instability in air and sensitivity to atmospheric moisture, creating reproducibility issues that are unacceptable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN101048396A represents a paradigm shift towards greener and more efficient chemical manufacturing. By employing a cyclization reaction mediated by an azodicarboxylate, a phosphine, and a strong base, the process operates under remarkably mild conditions, typically around 0°C. This approach eliminates the need for corrosive mineral acids or unstable acid chlorides, thereby mitigating equipment corrosion risks and simplifying waste stream management. The reaction demonstrates high reproducibility and allows for the direct conversion of precursor diols (Formula III for Citalopram and Formula IV for Escitalopram) into the target phthalan structures with high fidelity. This technological advancement not only improves the overall mass balance of the synthesis but also aligns with modern principles of cost reduction in antidepressant manufacturing by reducing the burden on downstream purification units.
Mechanistic Insights into Azodicarboxylate-Mediated Cyclization
The core of this innovative process lies in the intramolecular etherification facilitated by the activation of the hydroxyl group via a phosphonium intermediate. In the presence of a strong base such as sodium tert-butoxide, the phenolic or benzylic hydroxyl group of the precursor diol is deprotonated or activated to attack the electrophilic center generated by the interaction between the alcohol and the azodicarboxylate-phosphine complex. This mechanism proceeds with inversion of configuration at the reacting center, which is a critical factor when synthesizing the chiral Escitalopram. The use of tetrahydrofuran (THF) as the preferred solvent ensures optimal solubility of the polar intermediates and facilitates the necessary molecular collisions for the cyclization to proceed efficiently. The precise stoichiometric control, utilizing approximately 5.3 equivalents of ethyl azodicarboxylate (DEAD) and 3 equivalents of triphenylphosphine, drives the equilibrium towards the desired cyclic product while minimizing side reactions.
Impurity control is inherently built into this mechanistic pathway due to the mild thermal conditions employed. Unlike the high-temperature acid-catalyzed routes that promote elimination side reactions and polymerization, this low-temperature protocol preserves the integrity of the sensitive nitrile and amine functionalities present in the molecule. For the synthesis of Escitalopram, the non-racemizing nature of the reaction conditions is paramount. The process ensures that the optical purity of the starting chiral diol (Formula IV) is retained in the final product, minimizing the formation of the pharmacologically inactive R(-) enantiomer. This high level of stereochemical control reduces the need for costly chiral resolution steps post-synthesis, directly impacting the cost of goods sold (COGS) and enhancing the economic viability of producing high-purity escitalopram for the global market.
How to Synthesize Citalopram and Escitalopram Efficiently
The operational procedure for this synthesis is designed for scalability and safety, making it ideal for contract development and manufacturing organizations (CDMOs). The process begins with the dissolution of the precursor diol in anhydrous tetrahydrofuran under an inert nitrogen atmosphere to prevent oxidation of the phosphine reagent. Reagents are added sequentially at controlled temperatures to manage the exotherm associated with the activation step. Following the reaction period, a straightforward workup involving acid quenching and pH-controlled extraction allows for the isolation of the product either as a free base or as a stable salt, such as the oxalate or hydrobromide. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for maximum yield, are outlined below.
- Dissolve the precursor diol (Formula III or IV) in tetrahydrofuran (THF) under a nitrogen atmosphere at approximately 0°C.
- Add triphenylphosphine followed by the dropwise addition of ethyl azodicarboxylate (DEAD) and sodium tert-butoxide dissolved in THF.
- Stir the mixture overnight, quench with hydrochloric acid, extract with toluene at basic pH, and isolate the product as a salt (e.g., oxalate or hydrobromide).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel cyclization technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of hazardous reagents like hot concentrated sulfuric acid and unstable acid chlorides drastically simplifies the safety protocols required for plant operation. This reduction in hazard potential lowers insurance costs and minimizes the risk of production stoppages due to safety incidents. Furthermore, the mild reaction conditions reduce the energy load required for heating and cooling cycles, contributing to a lower carbon footprint and aligning with corporate sustainability goals. The robustness of the process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers.
- Cost Reduction in Manufacturing: The transition away from corrosive acids and unstable acid chlorides results in significant capital expenditure savings regarding reactor lining and maintenance. By avoiding the degradation pathways associated with harsh acidic conditions, the process achieves higher crude yields, which reduces the volume of raw materials required per kilogram of finished product. Additionally, the simplified impurity profile lowers the consumption of solvents and adsorbents during the purification stages, leading to substantial cost savings in waste disposal and material usage without compromising product quality.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as triphenylphosphine and common azodicarboxylates, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. The stability of the reaction intermediates allows for greater flexibility in scheduling and logistics, ensuring that production timelines can be met even during periods of raw material volatility. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, enabling faster time-to-market for generic formulations.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production without the need for specialized high-pressure or high-temperature equipment. The aqueous workup generates waste streams that are easier to treat compared to those containing heavy metal catalysts or strong mineral acids. This ease of effluent treatment facilitates compliance with increasingly stringent environmental regulations, ensuring long-term operational continuity and reducing the liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclization technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical application of the method. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new cyclization process improve upon traditional sulfuric acid methods?
A: Traditional methods utilize 70% sulfuric acid at elevated temperatures (80°C), which often leads to substrate degradation and lower yields. The novel process employs mild conditions (0°C) with azodicarboxylates and phosphines, significantly reducing degradation and improving reproducibility.
Q: Is this process suitable for the production of chiral Escitalopram?
A: Yes, the process is particularly advantageous for Escitalopram (Formula II). It utilizes the chiral diol precursor (Formula IV) under non-racemizing conditions, ensuring high stereoselectivity and minimizing the formation of the inactive R(-) enantiomer.
Q: What are the key reagents required for this cyclization reaction?
A: The core reaction requires an azodicarboxylate (preferably DEAD or DIAD), a triarylphosphine (such as triphenylphosphine), and a strong base (like sodium tert-butoxide) in a solvent such as THF.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram and Escitalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific reagent requirements of this mild cyclization process safely and effectively.
We invite potential partners to engage with our technical procurement team to discuss how this innovative technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, quality, and mutual growth.
