Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Drug Discovery
Introduction to Next-Generation Biheterocyclic Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component strategy for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This innovation addresses critical bottlenecks in traditional heterocycle construction by utilizing a transition metal palladium-catalyzed carbonylation cascade. The significance of this technology lies in its ability to forge multiple chemical bonds in a single operational step, thereby streamlining the synthetic route for high-value pharmaceutical intermediates. By leveraging cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, this method offers a sustainable alternative to legacy processes that often rely on hazardous reagents or multi-step sequences.
Furthermore, the structural diversity achievable through this protocol is immense, allowing for the incorporation of trifluoromethyl groups which are highly prized in medicinal chemistry for their metabolic stability and lipophilicity modulation. The reaction operates under remarkably mild conditions, typically at 30°C, which preserves sensitive functional groups that might otherwise degrade under harsh thermal regimes. For R&D directors and process chemists, this represents a pivotal shift towards more efficient library synthesis and lead optimization campaigns. The elimination of toxic carbon monoxide gas in favor of a liquid CO surrogate not only enhances laboratory safety but also simplifies the engineering requirements for scale-up, making it an attractive candidate for industrial adoption by reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has been fraught with synthetic challenges that impede rapid development cycles. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and poor regioselectivity. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles require stoichiometric oxidants and harsh conditions, leading to significant waste generation and compatibility issues with sensitive functional groups. Perhaps the most significant barrier in conventional carbonylation strategies is the reliance on gaseous carbon monoxide, a highly toxic and flammable substance that necessitates specialized high-pressure equipment and rigorous safety protocols. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for producing complex intermediates, creating friction in the supply chain for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated palladium-catalyzed tandem reaction that seamlessly integrates carbonylation and cyclization events. This novel approach employs a liquid mixture of formic acid and acetic anhydride as a safe and convenient carbon monoxide substitute, effectively bypassing the need for handling dangerous gases. The reaction proceeds through a cascade mechanism where a zero-valent palladium species inserts into a carbon-iodine bond, followed by an intramolecular Heck reaction and subsequent carbonylation. This one-pot transformation allows for the simultaneous formation of multiple bonds, drastically reducing the number of isolation steps and purification burdens. The versatility of this system is evidenced by its broad substrate scope, accommodating various electronic and steric environments on the aromatic rings without compromising yield.
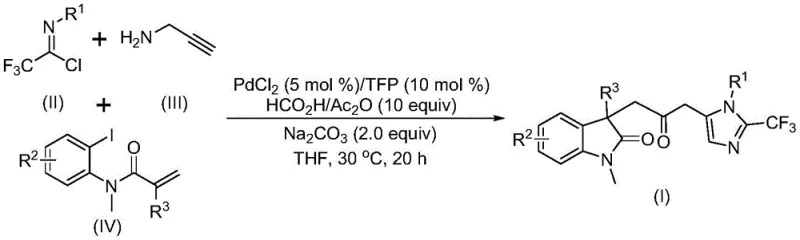
The operational simplicity of this new route cannot be overstated; reactions are conducted at a mild 30°C in common solvents like tetrahydrofuran (THF), achieving high conversion rates within 12 to 20 hours. Post-reaction processing is straightforward, involving simple filtration and silica gel chromatography, which facilitates rapid turnaround times for process development teams. By consolidating what would traditionally be a multi-step sequence into a single efficient operation, this technology offers substantial cost reduction in pharmaceutical intermediate manufacturing. It empowers chemists to access diverse chemical space with greater speed and safety, aligning perfectly with the modern demands for agile and sustainable drug discovery pipelines.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and predicting substrate compatibility. The catalytic cycle initiates with the oxidative addition of a zero-valent palladium species, generated in situ from PdCl2 and the TFP ligand, into the carbon-iodine bond of the acrylamide substrate. This generates a reactive organopalladium(II) intermediate which subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a five-membered palladacycle. This cyclization step is critical as it establishes the indolinone core structure early in the sequence. Following this, the palladium center interacts with carbon monoxide, which is released slowly and steadily from the decomposition of the formic acid/acetic anhydride mixture. This carbonylation event converts the alkyl-palladium species into an acyl-palladium intermediate, setting the stage for the final ring closure.
Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular reaction to form a trifluoroacetamidine species, which may isomerize prior to the final coupling. The acyl-palladium intermediate then activates this amidine compound, facilitating a nucleophilic attack or insertion that leads to the formation of the imidazole ring. This intricate dance of bond formations results in the final carbonyl-bridged biheterocyclic architecture. The choice of sodium carbonate as the base and TFP as the ligand is instrumental in maintaining the catalytic activity and stabilizing the palladium species throughout the cycle. This deep mechanistic understanding allows for precise tuning of reaction conditions to minimize side products and maximize the purity of the final API intermediate, ensuring that the impurity profile remains within strict regulatory limits.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to reagent quality and stoichiometry to ensure reproducible high yields. The protocol dictates a specific molar ratio of substrates, typically employing an excess of propargylamine and acrylamide relative to the trifluoroethylimidoyl chloride to drive the equilibrium forward. The catalyst loading is optimized at 5 mol% for PdCl2 and 10 mol% for the TFP ligand, balancing cost efficiency with catalytic turnover. The reaction is performed in anhydrous THF to prevent hydrolysis of the sensitive imidoyl chloride and acyl intermediates. Maintaining the temperature at 30°C is vital; while higher temperatures might accelerate the rate, they could also promote decomposition pathways or polymerization of the acrylamide component. Detailed standardized synthesis steps follow below.
- Combine palladium chloride, trifurylphosphine, sodium carbonate, and the CO substitute mixture (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction vessel.
- Stir the mixture at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. The starting materials—trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—are commodity chemicals that are commercially available in bulk quantities from multiple global vendors. This redundancy in sourcing mitigates the risk of supply disruptions that often plague projects relying on exotic or custom-synthesized building blocks. Furthermore, the elimination of gaseous carbon monoxide removes the need for specialized gas cylinders, regulators, and leak detection systems, resulting in significant capital expenditure savings and reduced operational overhead for manufacturing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the convergence of several factors. Firstly, the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps, which are typically the most labor-intensive and solvent-consuming parts of any synthesis. By reducing the number of unit operations, manufacturers can achieve substantial savings in labor, energy, and solvent disposal costs. Secondly, the use of a liquid CO surrogate avoids the high costs associated with high-pressure reactor vessels and the safety infrastructure required for toxic gas handling. Thirdly, the high yields reported in the patent examples, often exceeding 80% and reaching up to 92%, ensure that raw material utilization is maximized, minimizing waste and improving the overall mass balance of the process.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it through robustness and flexibility. The reaction conditions are mild (30°C) and utilize common solvents like THF, meaning that production can be easily transferred between different manufacturing sites without requiring specialized equipment modifications. The broad functional group tolerance means that if a specific substituent is needed for a drug candidate, the synthesis can often proceed without changing the core process parameters, allowing for rapid adaptation to changing project requirements. This flexibility ensures that production schedules remain intact even when molecular structures are optimized late in the development cycle, providing a stable foundation for long-term supply agreements.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant is often where many promising technologies fail, but this method has demonstrated successful expansion to the gram scale with consistent performance. The absence of high-pressure gases and the use of relatively non-hazardous reagents simplify the hazard analysis and operability study (HAZOP) required for scale-up. From an environmental perspective, the atom economy is superior to stepwise approaches, and the reduction in solvent usage per kilogram of product aligns with green chemistry principles. This facilitates easier compliance with increasingly stringent environmental regulations regarding waste discharge and volatile organic compound (VOC) emissions, future-proofing the manufacturing process against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing production workflows. We encourage technical teams to review these points closely to assess the alignment with their specific project goals and quality standards.
Q: What are the safety advantages of this new synthesis method?
A: This method replaces toxic carbon monoxide gas with a safe liquid mixture of formic acid and acetic anhydride, significantly reducing operational hazards and infrastructure costs.
Q: What is the substrate scope for this reaction?
A: The process demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on the aromatic rings.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly confirms successful gram-scale expansion, indicating strong potential for commercial scale-up due to mild conditions and simple workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multi-component synthesis for accelerating drug discovery programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and advanced purification capabilities to meet stringent purity specifications. Our rigorous QC labs employ comprehensive analytical methods to verify the identity and purity of every batch, guaranteeing that the carbonyl-bridged biheterocyclic intermediates we supply meet the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our team of expert process chemists is ready to conduct a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize yield and reduce overall production costs. We encourage you to contact our technical procurement team today to request specific COA data for similar compounds and to discuss route feasibility assessments. By partnering with us, you gain access to a reliable supply chain capable of delivering high-quality intermediates with the speed and flexibility required in today's competitive pharmaceutical landscape.
