Advanced Asymmetric Hydrogenation for High-Purity Levetiracetam Intermediates and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic routes for antiepileptic agents, and patent CN1179944C presents a significant technological advancement in the production of Levetiracetam intermediates. This intellectual property details a novel method for preparing (S)-(-)-α-ethyl-2-oxo-1-pyrrolidineacetamide through the asymmetric hydrogenation of unsaturated 2-oxo-1-pyrrolidine derivatives. The core innovation lies in bypassing traditional resolution steps that inherently waste half of the produced material, instead utilizing chiral catalysis to directly generate the desired enantiomer with exceptional stereocontrol. This approach not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by reducing solvent usage and chemical waste associated with separation processes. For global procurement teams, understanding this underlying technology is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality. The structural integrity of the final active pharmaceutical ingredient depends heavily on the precision of this hydrogenation step, making the choice of manufacturing partner a critical strategic decision. 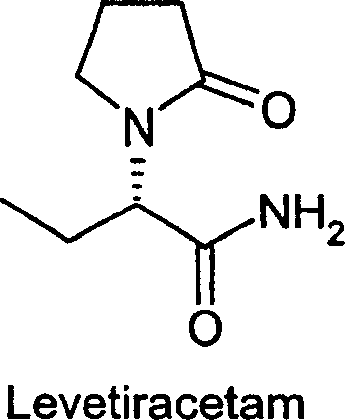
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of chiral antiepileptic compounds relied heavily on classical resolution techniques, which involve synthesizing a racemic mixture and subsequently separating the desired enantiomer from its mirror image. This traditional paradigm suffers from a fundamental theoretical yield limit of 50%, meaning that half of the synthesized material is discarded or requires costly recycling processes to be useful. Furthermore, resolution often necessitates the use of stoichiometric amounts of chiral resolving agents, which increases the raw material costs and generates substantial solid waste that must be treated and disposed of according to strict environmental regulations. The additional unit operations required for crystallization and separation extend the overall production cycle time, thereby increasing the working capital tied up in inventory and reducing the agility of the supply chain to respond to market demand fluctuations. From a quality control perspective, achieving high enantiomeric purity through resolution can be inconsistent, often requiring multiple recrystallization steps that further erode the overall process yield and economic viability. These inefficiencies create significant bottlenecks for procurement managers looking for cost reduction in API manufacturing, as the inherent waste of the resolution process translates directly into higher unit prices for the final intermediate.
The Novel Approach
In contrast, the methodology disclosed in CN1179944C leverages asymmetric catalytic hydrogenation to construct the chiral center directly during the bond-forming event, effectively circumventing the need for post-synthesis resolution. By employing specialized chiral transition metal catalysts, the reaction selectively reduces the double bond of the unsaturated precursor to form the desired (S)-enantiomer with high fidelity. This catalytic approach transforms the process from a stoichiometric operation into a highly efficient cycle where minute quantities of expensive chiral ligands can facilitate the production of large volumes of product. The elimination of the resolution step inherently doubles the potential yield from the starting materials, providing a substantial cost savings advantage that can be passed down the supply chain. Moreover, the reaction conditions described, such as moderate hydrogen pressures and ambient temperatures, are compatible with standard industrial reactor setups, facilitating easier technology transfer and scale-up. This shift represents a paradigm change in how high-purity pharmaceutical intermediates are sourced, offering a more sustainable and economically attractive route for long-term commercial partnerships.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise interaction between the substrate and the chiral catalyst complex, typically based on rhodium or ruthenium centers coordinated with bidentate phosphine ligands. The patent extensively details the screening of various ligands such as DUPHOS, BINAP, and DIPAMP, revealing that the steric and electronic properties of these ligands dictate the facial selectivity of the hydrogen addition. For instance, the use of (S,S)-Et-DUPHOS ligands in conjunction with rhodium precursors has demonstrated superior performance in terms of both conversion rates and enantiomeric excess compared to other systems. The mechanism involves the coordination of the olefinic double bond of the precursor to the metal center, followed by the migratory insertion of hydride species generated from molecular hydrogen. This sequence is highly sensitive to the solvent environment, with non-coordinating solvents like dichloromethane often providing better results than protic solvents which might compete for coordination sites on the catalyst. Understanding these mechanistic nuances is vital for R&D directors evaluating the robustness of the process, as it ensures that the impurity profile remains tightly controlled throughout the production campaign. 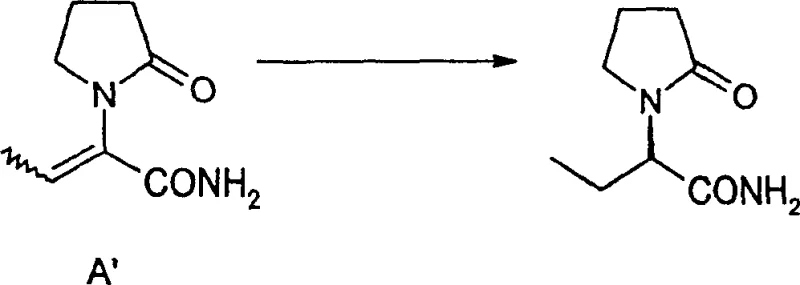
Controlling the impurity profile is equally critical, particularly regarding the removal of residual heavy metals from the final product to meet stringent pharmacopoeial standards. The patent describes purification protocols involving recrystallization from solvents like acetone or isopropanol, which effectively reduce metal residues to levels as low as 23 ppm. This level of purity is achieved without compromising the enantiomeric integrity of the molecule, ensuring that the final intermediate is suitable for direct use in subsequent drug substance manufacturing steps. The ability to consistently achieve enantiomeric excess values greater than 97% minimizes the risk of chiral impurities that could affect the safety and efficacy of the final medication. Such rigorous control over the chemical landscape demonstrates a deep understanding of process chemistry, providing confidence to regulatory affairs teams during the filing and approval stages of new drug applications. The detailed optimization of reaction parameters, including catalyst loading and hydrogen pressure, further underscores the maturity of this technology for industrial application.
How to Synthesize Levetiracetam Efficiently
The synthesis begins with the preparation of the unsaturated precursor through the condensation of alpha-ketocarboxylic acid derivatives with pyrrolidone, followed by the critical asymmetric hydrogenation step. Detailed standardized synthetic steps see the guide below. This structured approach ensures reproducibility and safety across different manufacturing sites, allowing for consistent quality regardless of production volume. The integration of these steps into a cohesive workflow is essential for maintaining the efficiency gains promised by the novel catalytic method.
- Prepare the unsaturated 2-oxo-1-pyrrolidine derivative precursor (Formula A) by condensing alpha-ketocarboxylic acid derivatives with pyrrolidone under reflux conditions.
- Conduct asymmetric catalytic hydrogenation using chiral Rh or Ru catalysts (e.g., DUPHOS, BINAP) in solvents like DCM or EtOH under controlled hydrogen pressure.
- Purify the resulting crude product through recrystallization from solvents such as acetone or isopropanol to achieve high enantiomeric excess and remove metal residues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology offers transformative benefits that extend beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the manufacturing process, which eliminates the need for resolution agents and the associated waste disposal costs. This streamlining leads to significant cost savings in manufacturing, as the raw material efficiency is nearly doubled compared to traditional racemic synthesis and resolution pathways. Furthermore, the reduced number of processing steps shortens the overall production lead time, enabling suppliers to respond more rapidly to urgent orders and market fluctuations. The use of commercially available solvents and standard hydrogenation equipment means that the barrier to entry for scaling this process is low, ensuring a stable and continuous supply of materials. These factors collectively enhance supply chain reliability, reducing the risk of shortages that can disrupt downstream drug production schedules. 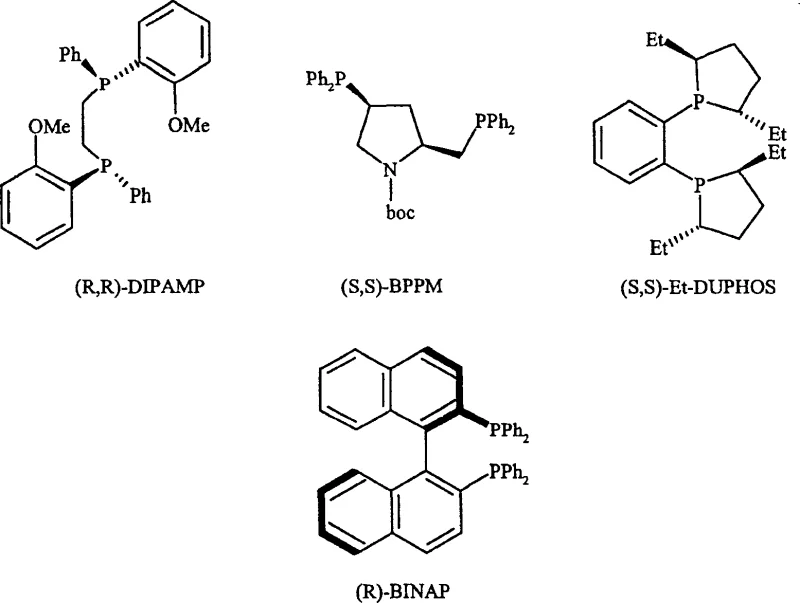
- Cost Reduction in Manufacturing: The elimination of resolution steps removes the inherent 50% yield loss associated with separating racemic mixtures, effectively doubling the output from the same amount of starting materials. Additionally, the catalytic nature of the reaction means that expensive chiral ligands are used in small quantities and can potentially be recovered, further lowering the variable cost per kilogram. The reduction in solvent consumption and waste treatment requirements also contributes to a lower overall environmental compliance cost, making the process economically superior. These efficiencies allow for a more competitive pricing structure without sacrificing margin, providing tangible value to the purchasing department.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route, the number of potential failure points in the manufacturing process is significantly reduced, leading to higher batch success rates. The reliance on standard industrial equipment and readily available reagents ensures that production is not bottlenecked by specialized or scarce resources. This robustness translates into more predictable delivery schedules and the ability to maintain safety stock levels with greater confidence. For supply chain planners, this reliability is crucial for managing inventory levels and ensuring uninterrupted production of the final pharmaceutical product.
- Scalability and Environmental Compliance: The process is designed to scale seamlessly from laboratory bench-top experiments to multi-ton commercial production without requiring fundamental changes to the chemistry. The reduced generation of chemical waste and the avoidance of stoichiometric chiral auxiliaries align with increasingly strict global environmental regulations. This compliance reduces the regulatory burden on the manufacturer and mitigates the risk of production shutdowns due to environmental violations. Consequently, partners can rely on a sustainable supply source that is future-proofed against tightening ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What enantiomeric excess can be achieved with this hydrogenation method?
A: According to patent CN1179944C, the asymmetric hydrogenation process using optimized chiral catalysts like (S,S)-Et-DUPHOS can consistently achieve enantiomeric excess (e.e.) values exceeding 97%, with some conditions reaching up to 99% e.e., ensuring high optical purity for pharmaceutical applications.
Q: Which catalysts are most effective for this specific transformation?
A: The patent highlights rhodium-based complexes with chiral diphosphine ligands such as DUPHOS, BPPM, and BINAP as highly effective. Specifically, Rh-DUPHOS complexes in non-coordinating solvents like dichloromethane have shown superior selectivity and conversion rates compared to other systems.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the methodology is designed for industrial scalability. The use of standard hydrogenation equipment, recoverable catalysts, and robust reaction conditions allows for seamless transition from laboratory gram-scale experiments to multi-ton annual commercial production without compromising quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced technologies like the one described in CN1179944C to deliver superior pharmaceutical intermediates to our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Levetiracetam intermediate meets the highest international standards. Our commitment to technical excellence allows us to optimize these catalytic processes for maximum efficiency and cost-effectiveness, providing you with a competitive edge in the marketplace. Partnering with us means gaining access to a wealth of chemical expertise and a robust manufacturing infrastructure dedicated to quality.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your project goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized supply chain solutions. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to handle your most challenging synthesis projects. Let us collaborate to drive innovation and efficiency in your pharmaceutical manufacturing operations.
