Advanced Synthetic Route for Entecavir Intermediates: Scalability and Commercial Viability Analysis
Advanced Synthetic Route for Entecavir Intermediates: Scalability and Commercial Viability Analysis
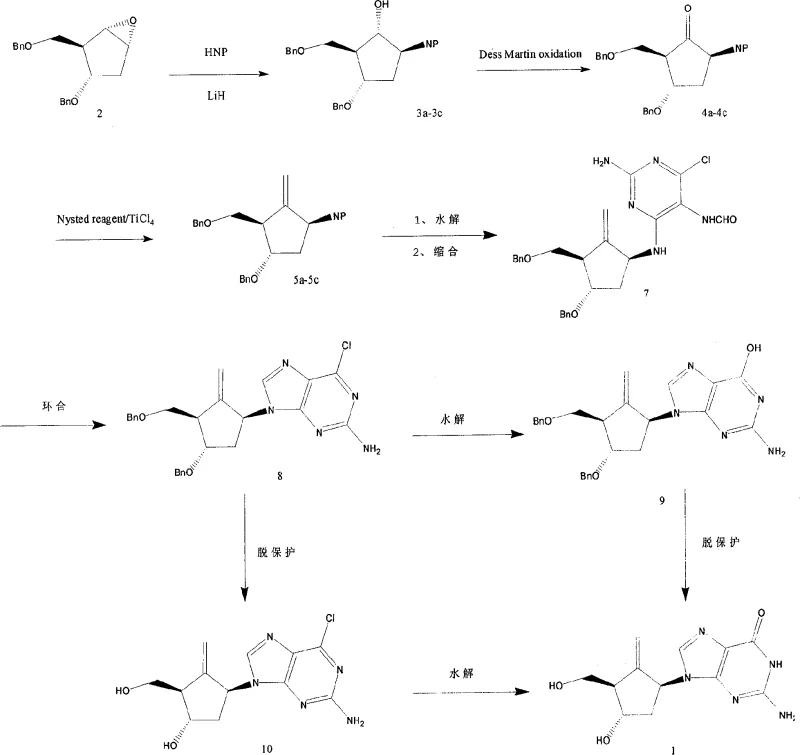
The global demand for potent antiviral agents, particularly for the treatment of chronic Hepatitis B, has necessitated the development of robust and scalable synthetic routes for key nucleoside analogs. Patent CN101148450A presents a sophisticated preparation method for 2-amino-9-[(1S,3S,4S)-4-hydroxy-3-hydroxymethyl-2-methylenecyclopentyl]-1,9-dihydro-6H-purin-6-one, widely known as Entecavir. This technical disclosure outlines a novel pathway that begins with a bicyclic ether precursor and employs a series of high-precision transformations, including Nysted methylenation and purine cyclization, to construct the complex carbocyclic nucleoside framework. For R&D directors and process chemists, this patent offers a compelling alternative to traditional methods, promising improved stereocontrol and operational simplicity. The strategic selection of reagents, such as Dess-Martin periodinane for oxidation and Titanium Tetrachloride for methylenation, underscores a commitment to maintaining high purity profiles while navigating the intricate stereochemistry required for biological efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbocyclic nucleosides like Entecavir has been plagued by challenges related to low overall yields and difficult purification steps, particularly during the formation of the exocyclic double bond and the closure of the purine ring. Conventional routes often rely on harsh reaction conditions or multiple protection-deprotection sequences that erode atom economy and generate significant chemical waste. Furthermore, earlier methodologies frequently struggled with regioselectivity during the coupling of the sugar mimic with the nucleobase, leading to complex mixtures of anomers that are costly and time-consuming to separate. These inefficiencies not only inflate the cost of goods sold (COGS) but also introduce supply chain vulnerabilities due to the reliance on specialized, low-volume reagents. The cumulative effect of these drawbacks is a manufacturing process that is difficult to scale to multi-ton quantities without compromising on the stringent purity specifications required for pharmaceutical intermediates.
The Novel Approach
In contrast, the methodology described in CN101148450A introduces a streamlined sequence that leverages the inherent strain of a bicyclo[3.1.0]hexane system to drive the formation of the cyclopentane core with high stereochemical fidelity. By initiating the synthesis with a readily available benzyloxy-substituted bicyclic ether, the process bypasses several cumbersome steps associated with building the ring system from scratch. The integration of a Nysted methylenation step allows for the precise installation of the critical methylene group under controlled低温 conditions, minimizing side reactions. Subsequently, the condensation with a pre-functionalized pyrimidine intermediate facilitates a smoother cyclization to form the purine nucleus. This approach not only enhances the overall yield but also simplifies the downstream processing, making it an attractive candidate for cost reduction in antiviral drug manufacturing. The route is designed to be robust, ensuring that the critical chiral centers are preserved throughout the synthesis, thereby delivering a high-quality intermediate suitable for final API production.
Mechanistic Insights into Nysted Methylenation and Purine Cyclization
The cornerstone of this synthetic strategy is the stereoselective methylenation of the cyclopentanone intermediate using the Nysted reagent in the presence of Titanium Tetrachloride (TiCl4). Mechanistically, the Lewis acidic TiCl4 activates the carbonyl oxygen of the ketone, facilitating the nucleophilic attack by the methylene equivalent derived from the Nysted reagent. This reaction proceeds through a concerted transition state that favors the formation of the exocyclic double bond with the desired geometry, which is essential for the subsequent biological activity of the nucleoside. The reaction is typically conducted at temperatures ranging from -78°C to 20°C, a window that balances reaction kinetics with selectivity, preventing the isomerization of the double bond or decomposition of the sensitive intermediate. This level of control is paramount for a reliable nucleoside intermediate supplier, as it ensures batch-to-batch consistency and minimizes the formation of geometric isomers that could act as difficult-to-remove impurities.
Following the construction of the carbocyclic scaffold, the formation of the purine ring via cyclization represents another critical mechanistic juncture. The condensation of the aminocyclopentyl intermediate with 2-amino-4-chloro-5-nitropyrimidinone sets the stage for ring closure. The subsequent cyclization, promoted by triethyl orthoformate and acid catalysis, involves the displacement of the nitro group and the formation of the imidazole ring of the purine system. This intramolecular cyclization is driven by the thermodynamic stability of the aromatic purine nucleus. The use of triethyl orthoformate serves a dual purpose: it acts as a dehydrating agent to drive the equilibrium forward and provides the one-carbon unit necessary for the imidazole ring closure. Understanding these mechanistic nuances allows process engineers to optimize reaction parameters, such as acid concentration and temperature, to maximize conversion and minimize the generation of des-chloro or open-ring byproducts, thereby ensuring high-purity Entecavir intermediate output.
How to Synthesize Entecavir Intermediate Efficiently
The execution of this synthesis requires careful attention to reaction conditions and reagent quality to achieve the reported high yields. The process begins with the ring-opening of the bicyclic starting material using an imide nucleophile in the presence of Lithium Hydride, followed by oxidation and methylenation to establish the core cyclopentene structure. The subsequent coupling with the pyrimidine base and cyclization steps must be tightly controlled to prevent degradation of the sensitive purine ring. For detailed operational parameters, including specific solvent volumes, stoichiometric ratios, and workup procedures, operators should refer to the standardized protocols derived from the patent examples. The following guide summarizes the critical phases of the synthesis, providing a roadmap for laboratory and pilot-scale implementation.
- Perform ring-opening of the bicyclo[3.1.0]hexane starting material with an imide group using Lithium Hydride (LiH) at elevated temperatures (100-150°C).
- Oxidize the resulting alcohol to a ketone using Dess-Martin periodinane, followed by methylenation with Nysted reagent and TiCl4 to introduce the exocyclic double bond.
- Condense the cyclopentyl amine with 2-amino-4-chloro-5-nitropyrimidinone, followed by cyclization with triethyl orthoformate and final deprotection using Boron Trichloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The reliance on readily available starting materials, such as the benzyloxy-substituted bicyclic ether, mitigates the risk of raw material shortages that often plague complex pharmaceutical syntheses. Furthermore, the elimination of exotic or highly toxic reagents in favor of more common industrial chemicals like Dess-Martin periodinane and Titanium Tetrachloride simplifies the sourcing process and reduces regulatory burdens associated with hazardous material handling. This streamlining of the supply chain translates directly into enhanced reliability and predictability for production schedules, allowing manufacturers to respond more agilely to market demands for antiviral therapies.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by improving the overall yield and reducing the number of purification steps required. By avoiding the formation of complex mixtures of isomers, the need for expensive chromatographic separations is minimized, leading to substantial savings in both solvent consumption and processing time. Additionally, the high efficiency of the Nysted methylenation step reduces the amount of starting material needed per kilogram of final product, directly lowering the variable cost of production. These efficiencies compound over large-scale campaigns, resulting in a more competitive cost structure for the final API.
- Enhanced Supply Chain Reliability: The use of robust and well-understood chemical transformations ensures that the manufacturing process is less susceptible to variability and scale-up issues. The starting materials are commodity chemicals or simple derivatives thereof, which are less prone to supply disruptions compared to highly specialized custom synthons. This stability allows for better long-term planning and inventory management, ensuring a continuous flow of intermediates to downstream API manufacturers. Consequently, partners can rely on a steady supply of high-purity pharmaceutical intermediates without the fear of unexpected production halts.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are amenable to large-scale reactor operations. The avoidance of extreme pressures or temperatures reduces the capital expenditure required for specialized equipment. Moreover, the improved atom economy and reduced waste generation align with modern green chemistry principles, simplifying waste treatment and disposal. This environmental compliance is increasingly critical for maintaining operating licenses and meeting the sustainability goals of global pharmaceutical clients, facilitating the commercial scale-up of complex nucleoside analogs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Entecavir intermediates as described in the patent literature. These insights are derived from a detailed analysis of the reaction mechanisms and process parameters to assist decision-makers in evaluating the feasibility of this route for their specific manufacturing needs. Understanding these nuances is essential for optimizing the technology transfer process and ensuring successful commercialization.
Q: What are the critical advantages of using the Nysted reagent in this synthesis?
A: The use of Nysted reagent combined with Titanium Tetrachloride allows for the highly stereoselective formation of the exocyclic methylene group at the C2 position of the cyclopentane ring under mild conditions (-78°C to 20°C), which is crucial for the biological activity of the final Entecavir molecule.
Q: How does this patent address the yield issues of previous methods?
A: By utilizing a specific bicyclo[3.1.0]hexane starting material and optimizing the condensation and cyclization steps, the process achieves significantly higher overall yields compared to prior art, specifically improving the efficiency of the purine ring formation and subsequent deprotection stages.
Q: Is the starting material for this route commercially viable?
A: Yes, the patent highlights that the starting material, [1S-(1α,2β,3α,5α)]-3-(benzyloxy)-2-[(benzyloxy)methyl]-6-oxabicyclo[3.1.0]hexane, is readily available, which simplifies the supply chain and reduces the risk of raw material bottlenecks during commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the rapidly evolving landscape of antiviral drug development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify identity and assay. Our dedication to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply chains for critical medications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
