Optimizing Entecavir Production: A Novel Synthetic Route for High-Purity Antiviral Intermediates
Optimizing Entecavir Production: A Novel Synthetic Route for High-Purity Antiviral Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for potent antiviral agents, particularly for the treatment of chronic hepatitis B. Patent CN101838270B discloses a groundbreaking methodology for synthesizing Entecavir, a carbocyclic guanosine analogue with significant clinical efficacy. Unlike conventional approaches that rely on the direct ring-opening of epoxides with guanine derivatives—a process plagued by low yields and purification nightmares—this invention introduces a strategic sequence involving nitrogenous nucleophilic ring-opening, oxidation, and methylenation. This technical insight report analyzes the mechanistic superiority of this route, offering R&D directors and procurement specialists a clear view of how this technology translates into a reliable Entecavir intermediate supplier capability with enhanced cost structures.
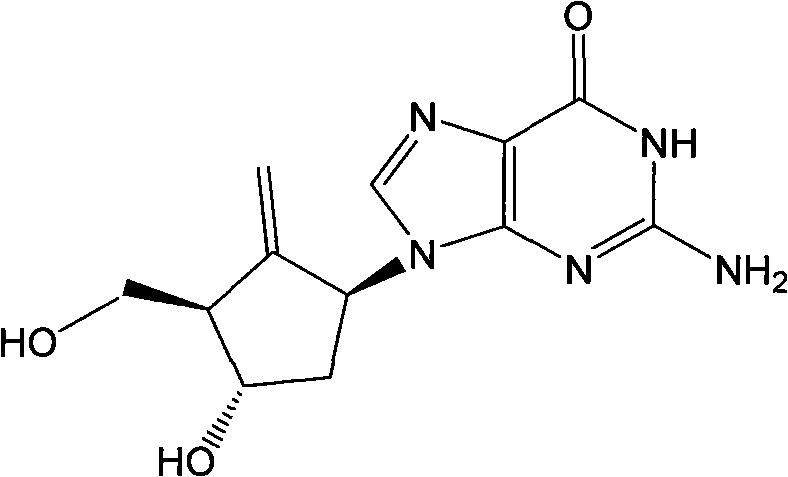
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Entecavir has been hindered by the reliance on direct nucleophilic substitution of epoxy cyclopentane compounds with protected guanine bases. As documented in prior art such as WO9809964 and JOC 1985, this traditional ring-opening reaction suffers from inherently low productivity, often reporting yields as low as 27% to 50%. Furthermore, the reaction generates a complex mixture of stereoisomers due to the lack of stereocontrol during the ring-opening event. Separating these diastereomers requires tedious and expensive repeated silica gel column chromatography, which is economically unviable for large-scale commercial production. Additionally, the amino group on the guanine ring necessitates protection strategies that are chemically unstable and difficult to remove without degrading the sensitive nucleoside structure, further complicating the downstream processing and lowering the overall purity of the final active pharmaceutical ingredient.
The Novel Approach
The methodology outlined in CN101838270B fundamentally re-engineers the synthetic logic by decoupling the formation of the carbocyclic scaffold from the attachment of the heterocyclic base. Instead of attacking the epoxide with a bulky guanine derivative, the process utilizes smaller, more reactive nitrogenous nucleophiles such as phthalimide or azides to open the epoxide ring. This strategic shift allows for precise control over the stereochemistry at the C1 position before the complex base is ever introduced. By deferring the construction of the purine system until after the carbocyclic core is fully functionalized with the exocyclic methylene group, the process eliminates the regioselectivity issues associated with 9-N versus 7-N alkylation. This results in a much cleaner reaction profile, higher overall yields, and a dramatic reduction in the need for extensive chromatographic purification, making it highly suitable for industrial scale-up.
Mechanistic Insights into Epoxide Ring-Opening and Methylenation
The core innovation lies in the sequential transformation of the epoxy cyclopentane precursor. The process initiates with the nucleophilic attack on the epoxide ring (Compound 1) by a nitrogen source, such as potassium phthalimide, under alkaline conditions in polar aprotic solvents like DMF. This step installs the nitrogen functionality with high stereochemical fidelity, yielding the trans-hydroxy intermediate (Compound 2). Following this, the secondary hydroxyl group is selectively oxidized to a ketone (Compound 3) using mild yet efficient oxidizing agents like Dess-Martin periodinane or PCC. This oxidation step is critical as it sets the stage for the subsequent methylenation. The conversion of the ketone to the exocyclic methylene group (Compound 4) is achieved using titanium-based methylenation reagents such as the Nysted or Tebbe reagent at low temperatures. This sequence ensures that the sensitive double bond is formed under controlled conditions, preserving the integrity of the protecting groups on the adjacent hydroxymethyl side chain.
Impurity control is intrinsically built into this mechanistic pathway. In traditional routes, the nucleophilic ambiguity of the guanine base leads to N7-alkylated byproducts that are structurally similar to the desired N9-product, making them notoriously difficult to separate. In contrast, the new route utilizes a phthalimide or azide group which acts as a masked amine. Since this nitrogen source is mono-functional and sterically defined, it attacks the epoxide exclusively at the intended position, preventing the formation of regioisomers. Furthermore, the intermediate ketone and alkene species (Compounds 3 and 4) possess distinct physical properties that allow for easy crystallization or simple extraction, avoiding the need for the loaded resin chromatography required in silane-based precursor methods. This inherent purity advantage ensures that the final coupling with the pyrimidine fragment proceeds with minimal side reactions, safeguarding the quality of the final Entecavir API.
How to Synthesize Entecavir Intermediates Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and minimize waste. The patented process details a robust protocol starting from readily available epoxy cyclopentane derivatives. The key to success lies in the selection of the nitrogenous nucleophile and the optimization of the methylenation step. Detailed operational guidelines regarding solvent choices, temperature gradients, and workup procedures are essential for reproducibility. For a comprehensive breakdown of the standardized synthetic steps, including specific molar ratios and quenching protocols, please refer to the technical guide below.
- Perform nucleophilic ring-opening of the epoxy cyclopentane compound using a nitrogenous reagent like phthalimide to form the hydroxy-intermediate.
- Oxidize the resulting hydroxyl group to a ketone using reagents such as Dess-Martin periodinane, followed by methylenation to introduce the exocyclic double bond.
- Couple the amino-cyclopentane intermediate with a chlorinated pyrimidine derivative, followed by cyclization and final deprotection to yield Entecavir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification train. By eliminating the need for repeated column chromatography to separate stereoisomers, the process significantly reduces solvent consumption and processing time. This directly translates to cost reduction in API manufacturing, as the most expensive unit operations in fine chemical synthesis are often the purification stages. Moreover, the avoidance of unstable protecting groups on the guanine base reduces the risk of batch failures due to decomposition, thereby enhancing supply chain reliability and ensuring consistent delivery schedules for downstream formulation partners.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents associated with traditional guanine protection strategies leads to substantial cost savings. By utilizing stable phthalimide or azide precursors, the process avoids the complex deprotection steps that often require harsh acidic or basic conditions, which can degrade the product and lower yields. The use of common solvents like ethanol and DMF, coupled with the ability to isolate intermediates via crystallization rather than chromatography, drastically lowers the operational expenditure per kilogram of produced intermediate, making the final Entecavir API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway ensures a more stable supply of critical antiviral intermediates. Traditional methods are prone to variability due to the sensitivity of the guanine ring-opening step, which can lead to inconsistent batch quality and delayed shipments. In contrast, the stepwise construction of the carbocyclic core described in this patent offers high reproducibility. The intermediates generated are chemically stable and can be stored or transported with greater ease, reducing the risk of supply disruptions caused by material degradation. This reliability is crucial for maintaining continuous production lines for life-saving hepatitis B medications.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is superior for commercial scale-up. The avoidance of heavy metal catalysts in the coupling steps (or their minimization) and the use of milder oxidants like Dess-Martin reduce the toxic load of the waste stream. The process is designed to be scalable from pilot plant to multi-ton production without the engineering bottlenecks associated with handling large volumes of silica gel or specialized resins. This aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility, which is increasingly important for compliance with international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthetic strategy. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for process chemistry and quality control.
Q: Why is the traditional guanine ring-opening method considered inefficient for Entecavir production?
A: Traditional methods suffer from low ring-opening yields (often below 50%), difficult separation of stereoisomers requiring repeated column chromatography, and complex protection/deprotection steps for the guanine amino group, which destabilizes the product.
Q: How does the novel epoxide ring-opening route improve impurity profiles?
A: By constructing the base attachment point via a specific nitrogenous nucleophile on the carbocyclic ring first, this route avoids the regioselectivity issues (9-N vs 7-N attack) inherent in direct guanine alkylation, significantly reducing diastereomeric impurities.
Q: What are the scalability advantages of using Dess-Martin oxidation in this pathway?
A: Dess-Martin oxidation offers mild reaction conditions and high transformation efficiency compared to harsh oxidants like chromic acid, facilitating easier handling and safer scale-up for commercial manufacturing without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of high-purity intermediates in the production of antiviral therapeutics. Our technical team has extensively analyzed the pathway described in CN101838270B and possesses the expertise to execute this complex chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Entecavir intermediate meets the highest pharmacopeial standards required for global regulatory submissions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the economic and operational efficiency of your Entecavir production program.
