Advanced Emtricitabine Manufacturing: A Green Route Using L-Menthol Chiral Auxiliaries
Advanced Emtricitabine Manufacturing: A Green Route Using L-Menthol Chiral Auxiliaries
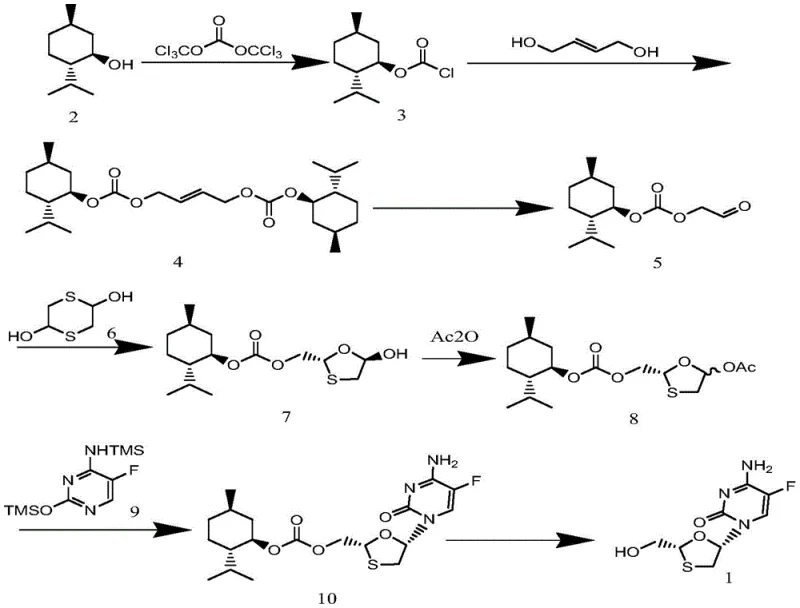
The global demand for potent antiretroviral therapies continues to drive the need for efficient, scalable, and environmentally sustainable manufacturing processes for key active pharmaceutical ingredients (APIs). Patent CN109438432B introduces a transformative preparation method for emtricitabine, a critical nucleoside reverse transcriptase inhibitor used in the treatment of HIV and Hepatitis B. This technical disclosure outlines a novel synthetic pathway that leverages L-menthol as a readily available chiral auxiliary, effectively bypassing the severe limitations of legacy production methods. By shifting away from hazardous reagents and extreme cryogenic conditions, this innovation offers a robust framework for the commercial scale-up of complex pharmaceutical intermediates. The methodology emphasizes high atom utilization and green chemistry principles, ensuring that the final product meets stringent medicinal standards while optimizing the supply chain for reliability and cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of emtricitabine has been plagued by significant technical and economic bottlenecks that hinder large-scale production efficiency. Prior art, such as the methods described in Patent AU2004200957, relies heavily on ozone oxidation of double bonds, a process that necessitates maintaining reaction temperatures as low as minus 78 degrees Celsius. Such cryogenic requirements impose a massive energy burden on manufacturing facilities and demand specialized, expensive equipment that is difficult to maintain in continuous operation. Furthermore, alternative routes disclosed in patents like CN101125872A utilize toxic oxidants and corrosive halogenating agents such as thionyl chloride. These reagents not only pose severe safety risks to personnel but also generate hazardous byproducts like hydrogen chloride, which can degrade acid-sensitive intermediates and compromise the overall yield and purity of the final API. Additionally, enzymatic methods reported in US5538975 suffer from enzyme inactivation issues and require tedious silica gel column purification, rendering them economically unviable for multi-ton production.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in CN109438432B utilizes a strategic sequence starting from inexpensive L-menthol and triphosgene. This method replaces the dangerous ozone oxidation step with a catalytic oxidation using hydrogen peroxide and tungstic acid, operating at mild temperatures between 60 and 90 degrees Celsius. This shift drastically simplifies the reactor requirements and eliminates the need for cryogenic infrastructure. The process employs a chiral auxiliary strategy where L-menthol induces stereochemistry early in the synthesis, allowing for precise control over the optical purity of the 1,3-oxathiolane ring system. By avoiding toxic halogenating agents and utilizing green solvents like dichloromethane and tert-butanol, the new route minimizes environmental impact and waste treatment costs. The result is a streamlined, seven-step synthesis that delivers high-purity emtricitabine suitable for direct pharmaceutical application, representing a significant leap forward in cost reduction in antiviral API manufacturing.
Mechanistic Insights into L-Menthol Mediated Chiral Induction
The core of this synthetic breakthrough lies in the sophisticated use of L-menthol as a chiral pool material to establish the necessary stereochemistry for the emtricitabine molecule. The process initiates with the conversion of L-menthol into L-menthyl chloroformate using triphosgene, a safer alternative to phosgene gas. This activated carbonate species then reacts with butenediol to form a bis-carbonate intermediate, effectively locking the chiral information from the menthol moiety onto the carbon chain. The subsequent oxidation step is particularly elegant; instead of cleaving the double bond destructively, the protocol uses a tungsten-based catalyst with hydrogen peroxide to selectively oxidize the alkene into a chiral aldehyde ester. This transformation preserves the stereocenter established by the menthol group while introducing the oxygen functionality required for the subsequent cyclization. The mild reaction conditions prevent racemization, ensuring that the optical integrity of the intermediate is maintained throughout the sequence.
Following the oxidation, the chiral aldehyde ester undergoes a cyclization reaction with 1,4-dithiane-2,5-diol in the presence of a Lewis acid catalyst such as boron trifluoride. This step constructs the critical 1,3-oxathiolane ring, a structural motif essential for the biological activity of emtricitabine. The mechanism proceeds through a hemithioacetal intermediate which cyclizes to form the five-membered ring with high diastereoselectivity. Subsequent acetylation protects the hydroxyl group, preparing the molecule for the glycosylation coupling with silylated 5-fluorocytosine. The final deprotection step utilizes a weak base like potassium carbonate in methanol to cleave the menthol auxiliary, releasing the free emtricitabine and recovering the chiral helper for potential reuse. This mechanistic pathway ensures that impurities are minimized at each stage, resulting in a product with a clean impurity profile.
How to Synthesize Emtricitabine Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity, particularly during the oxidation and coupling stages. The protocol begins with the preparation of the chiral carbonate building block, followed by the critical catalytic oxidation which must be monitored to prevent over-oxidation. The cyclization step demands anhydrous conditions to ensure the formation of the oxathiolane ring without hydrolysis side reactions. Finally, the coupling with the nucleobase requires careful temperature management to achieve the correct beta-anomeric configuration. For process chemists looking to implement this technology, the detailed standardized synthesis steps are provided below to ensure reproducibility and compliance with GMP standards.
- React L-menthol with triphosgene to form L-menthyl chloroformate.
- Condense with butenediol to form the bis-carbonate intermediate.
- Perform catalytic oxidation using hydrogen peroxide and tungstic acid to generate the chiral aldehyde ester.
- Cyclize with 1,4-dithiane-2,5-diol followed by acetylation.
- Couple with silylated 5-fluorocytosine and remove the chiral auxiliary to obtain emtricitabine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages by decoupling production from volatile supply chains associated with exotic reagents. The reliance on L-menthol, a commodity chemical derived from renewable peppermint sources, ensures a stable and predictable raw material supply compared to synthetic chiral auxiliaries that may face availability constraints. Furthermore, the elimination of ozone generators and cryogenic cooling systems reduces the capital expenditure (CAPEX) required for facility setup, making it easier for contract development and manufacturing organizations (CDMOs) to adopt this technology. The simplified workflow also translates to reduced operational expenditure (OPEX) due to lower energy consumption and decreased waste disposal costs associated with hazardous halogenated byproducts. This creates a more resilient supply chain capable of meeting the growing global demand for HIV and Hepatitis B treatments without the risk of production stoppages due to equipment failure or reagent shortages.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous reagents with cost-effective alternatives like triphosgene and hydrogen peroxide drives down the direct material costs significantly. By removing the need for energy-intensive cryogenic processes, the utility costs per kilogram of product are drastically lowered. Additionally, the high atom economy of the reaction sequence means less raw material is wasted, further enhancing the overall economic efficiency of the manufacturing process. The ability to potentially recover and recycle the L-menthol auxiliary adds another layer of cost savings, making the process highly competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor fluctuations in temperature or pressure compared to ozone-based methods. This reliability ensures consistent delivery timelines for downstream pharmaceutical formulators, reducing the need for excessive safety stock inventory. The simplified purification steps also shorten the overall production cycle time, enabling faster response to market demand surges.
- Scalability and Environmental Compliance: The green chemistry profile of this route aligns perfectly with modern environmental regulations, facilitating easier permitting and compliance audits. The absence of toxic heavy metals and corrosive gases simplifies waste treatment protocols, reducing the environmental footprint of the manufacturing site. The process is inherently scalable from pilot plant to commercial tonnage without the engineering challenges associated with handling large volumes of ozone or cryogenic fluids. This scalability ensures that the technology can grow alongside market demand, providing a long-term solution for emtricitabine production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this emtricitabine synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for technical teams evaluating the transition from legacy processes to this improved methodology.
Q: Why is the L-menthol route superior to ozone oxidation methods for emtricitabine?
A: Traditional ozone oxidation requires cryogenic conditions (-78°C), demanding high energy and specialized equipment. The L-menthol route utilizes mild catalytic oxidation with hydrogen peroxide at 60-90°C, significantly reducing operational complexity and safety risks associated with ozone handling.
Q: Does this process utilize toxic halogenating agents like thionyl chloride?
A: No. Unlike prior art methods that rely on corrosive thionyl chloride which generates hazardous HCl byproducts, this patented method employs triphosgene and green oxidants. This eliminates acid-sensitive decomposition risks and improves the environmental profile of the manufacturing process.
Q: What is the expected purity profile of the final emtricitabine product?
A: The process is designed to achieve medicinal standard purity. By utilizing a robust chiral auxiliary strategy with L-menthol and optimizing crystallization steps, the method ensures high optical purity and chemical integrity suitable for direct pharmaceutical formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Emtricitabine Supplier
The technological advancements presented in CN109438432B underscore the potential for high-efficiency manufacturing of critical antiviral intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this L-menthol mediated process are executed with precision. Our commitment to quality is backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of emtricitabine intermediate meets the highest international pharmacopoeia standards. We understand the critical nature of supply continuity in the pharmaceutical sector and have optimized our operations to deliver consistent quality and volume.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio while reducing overall production costs.
