Advanced L-Menthol Mediated Synthesis of Lamivudine for Commercial Scale-Up
Advanced L-Menthol Mediated Synthesis of Lamivudine for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of critical antiviral agents. A significant breakthrough in this domain is detailed in patent CN109438431B, which outlines a novel preparation method for Lamivudine, a key nucleoside reverse transcriptase inhibitor used in the treatment of HIV and Hepatitis B. This technical insight report analyzes the proprietary synthesis route that leverages L-menthol as a chiral auxiliary to achieve high optical purity and yield. Unlike traditional methods that rely on expensive enzymatic resolution or complex chiral chromatography, this approach utilizes a series of chemical transformations starting from readily available raw materials. The process is characterized by mild reaction conditions, high atom utilization, and a streamlined workflow that eliminates several purification bottlenecks. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial for evaluating potential supply chain partners who can deliver high-purity pharmaceutical intermediates with consistent quality and reduced lead times.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Lamivudine has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Early methods, such as those described in WO9117159, relied heavily on enzymatic resolution, which introduces variability due to enzyme inactivation and requires stringent temperature controls that increase operational costs. Furthermore, these processes often necessitate chiral column chromatography to separate cis and trans isomers, a step that is notoriously difficult to scale beyond kilogram quantities due to high solvent consumption and low throughput. Other routes, like those disclosed in US5047407, involve long reaction sequences with expensive starting materials such as 2-bromodiethoxyethane, leading to substantial material loss during the resolution steps. The cumulative effect of these inefficiencies is a low total yield and a final product that may struggle to meet the rigorous purity specifications required for API manufacturing without extensive downstream processing. Additionally, the use of corrosive halogenating agents and expensive oxidants in older methodologies raises serious environmental compliance concerns, complicating waste disposal and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in CN109438431B presents a paradigm shift by employing L-menthol as a recoverable chiral auxiliary to drive stereoselectivity. This novel approach simplifies the synthetic route by reducing the number of unit operations and eliminating the need for enzymatic catalysts, thereby enhancing process robustness. The strategy involves the formation of a chiral carbonate intermediate which directs the subsequent cyclization and coupling reactions with high fidelity. By utilizing triphosgene and butenediol in the initial stages, the process ensures that the chiral information is installed early and maintained throughout the synthesis. The reaction conditions are notably mild, often operating between 0°C and 80°C, which reduces energy consumption and minimizes the risk of thermal degradation of sensitive intermediates. This streamlined workflow not only improves the overall yield but also significantly simplifies the purification protocol, as the desired isomers can often be isolated through crystallization rather than chromatography. For supply chain heads, this translates to a more reliable production schedule and a drastic reduction in the complexity of manufacturing logistics.
Mechanistic Insights into L-Menthol Mediated Cyclization
The core of this synthesis lies in the precise construction of the 1,3-oxathiolane ring with the correct stereochemistry, a challenge that this patent addresses through a clever oxidation and cyclization sequence. The process begins with the conversion of L-menthol into L-menthyl chloroformate using triphosgene, followed by reaction with butenediol to form a bis-carbonate. The pivotal step involves the oxidative cleavage of the double bond in the bis-carbonate using hydrogen peroxide and a tungstic acid catalyst. This oxidation generates a chiral aldehyde ester intermediate with high optical purity, setting the stage for the ring closure. The subsequent cyclization with 1,4-dithiane-2,5-diol, catalyzed by boron trifluoride, forms the oxathiolane ring structure. The steric bulk of the menthol group effectively shields one face of the molecule, ensuring that the nucleophilic attack occurs from the desired direction. This mechanistic control is critical for achieving the 5S configuration required for biological activity. The use of tungstic acid as a catalyst is particularly noteworthy, as it offers a greener alternative to heavy metal catalysts while maintaining high activity and selectivity.
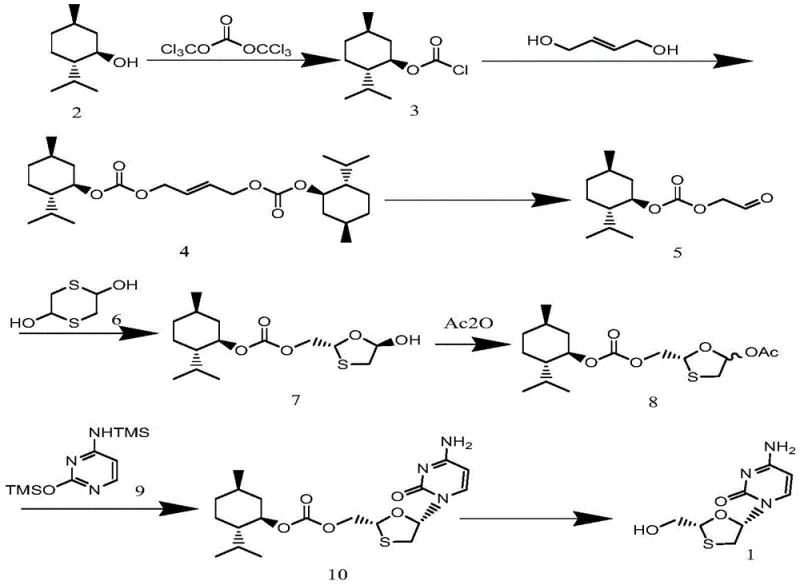
Following the ring formation, the intermediate undergoes acylation and coupling with silylated cytosine to install the nucleobase. The final step involves the removal of the chiral auxiliary under mild basic conditions using potassium carbonate in methanol. This deprotection step is gentle enough to preserve the integrity of the glycosidic bond while efficiently cleaving the carbonate linkage to release the free hydroxymethyl group of Lamivudine. The entire sequence is designed to maximize atom economy, ensuring that the majority of the starting material mass is incorporated into the final product. For R&D teams, this mechanistic clarity offers a clear path for process optimization, as each step is well-defined with specific parameters for temperature, time, and molar ratios. The ability to control impurity formation at the molecular level ensures that the final API meets stringent regulatory standards without the need for extensive reprocessing.
How to Synthesize Lamivudine Efficiently
Implementing this synthesis route requires a detailed understanding of the reaction parameters to ensure reproducibility and safety on a commercial scale. The patent provides specific guidance on reagent grades, solvent choices, and workup procedures that are essential for achieving the reported yields. For instance, the oxidation step specifically recommends the use of tert-butanol as a solvent to optimize the interaction between the organic substrate and the aqueous hydrogen peroxide phase. Similarly, the coupling reaction benefits from the use of hexamethyldisilazane to generate the silylated cytosine in situ, which enhances nucleophilicity and reaction rates. Operators must adhere to strict temperature controls, particularly during the exothermic formation of the chloroformate and the oxidation steps, to prevent side reactions. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- Preparation of L-menthyl chloroformate using triphosgene and subsequent reaction with butenediol to form the bis-carbonate intermediate.
- Oxidative cleavage of the double bond using hydrogen peroxide and tungstic acid to generate the chiral aldehyde ester.
- Cyclization with 1,4-dithiane-2,5-diol, followed by acylation, silylated cytosine coupling, and final deprotection to yield Lamivudine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this L-menthol mediated route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for antiviral intermediates. The primary advantage lies in the significant cost reduction in API manufacturing achieved by replacing expensive enzymatic reagents and chiral columns with cheap, commodity chemicals like L-menthol and triphosgene. The elimination of complex resolution steps reduces solvent consumption and waste disposal costs, directly impacting the bottom line. Furthermore, the robustness of the chemical steps ensures high supply chain reliability, as the process is less susceptible to the variability often seen in biocatalytic methods. The use of stable, non-hazardous reagents also simplifies logistics and storage requirements, reducing the risk of supply disruptions. For organizations seeking a reliable Lamivudine intermediate supplier, this technology represents a mature and scalable solution that can meet high-volume demand without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-cost starting materials and the high efficiency of the reaction sequence. By avoiding the need for expensive chiral resolution columns and enzymatic catalysts, the overall cost of goods sold is drastically reduced. The high yields reported in the patent examples, such as the 90.2% yield in the first step and 86% in the final deprotection, indicate a highly efficient use of raw materials. This efficiency translates into lower material costs per kilogram of final product, allowing for more competitive pricing in the global market. Additionally, the ability to recover and potentially recycle the chiral auxiliary further enhances the economic profile of the process.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for the production of life-saving medications, and this synthetic route offers superior stability compared to alternative methods. The reagents used, such as L-menthol, hydrogen peroxide, and common organic solvents, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The chemical nature of the process ensures that production can be maintained consistently regardless of seasonal or biological factors that might affect enzymatic routes. This reliability allows procurement teams to negotiate better long-term contracts and secure stable pricing. The simplified workflow also means that production lead times can be shortened, enabling faster response to market demand fluctuations and ensuring that inventory levels are maintained optimally.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the use of standard chemical engineering unit operations that do not require specialized equipment. The mild reaction conditions reduce the energy load on the manufacturing plant, contributing to lower operational costs and a smaller carbon footprint. Environmental compliance is significantly improved by the avoidance of heavy metal catalysts and the use of green oxidants like hydrogen peroxide, which decomposes into water and oxygen. This aligns with increasingly strict global environmental regulations and corporate sustainability goals. The reduced generation of hazardous waste simplifies the permitting process for new manufacturing facilities and lowers the long-term liability associated with waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. They are intended to provide clarity for stakeholders evaluating the feasibility of this route for their specific production needs. Understanding these details is essential for making informed decisions about technology transfer and vendor selection.
Q: What are the primary advantages of the L-menthol route over enzymatic methods for Lamivudine?
A: The L-menthol route avoids the high costs and stability issues associated with enzymatic resolution. It utilizes cheap, readily available chiral auxiliaries and achieves high optical purity through crystallization rather than expensive chiral chromatography, significantly simplifying the process for industrial scale-up.
Q: How does this synthesis method impact environmental compliance and waste management?
A: This method employs green oxidants like hydrogen peroxide and avoids heavy metal catalysts often found in traditional routes. The atom utilization rate is high, and the reagents are less corrosive, resulting in reduced hazardous waste generation and easier compliance with strict environmental regulations.
Q: Is the final product suitable for direct pharmaceutical formulation without further purification?
A: Yes, the patent data indicates that the final recrystallization steps yield Lamivudine with high chemical purity that meets medicinal standards. The specific removal of the chiral auxiliary under mild basic conditions ensures that impurity profiles are controlled effectively for API usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value antiviral compounds like Lamivudine. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the highest pharmaceutical standards. Our expertise in chiral synthesis and process optimization allows us to implement advanced methodologies, such as the L-menthol route, to deliver cost-effective solutions without compromising on quality. We are committed to being a partner that drives innovation and efficiency in your supply chain.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing capabilities can reduce your overall production costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume and timeline needs. Our goal is to provide you with the data and confidence required to move your projects forward successfully.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
