Advanced One-Pot Synthesis for 1-Alkylpyrrolidine Carbapenem Antibacterial Agents
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex antibiotic structures, particularly within the carbapenem class known for their broad-spectrum activity. Patent CN1486316A introduces a significant technological breakthrough in the production of carbapenem-type antibacterial agents possessing a 1-alkylpyrrolidine structure. This patent details a novel manufacturing method that streamlines the assembly of these complex molecules through a so-called one-pot synthesis approach, contrasting sharply with traditional multi-stage protocols. By utilizing a specific synthetic intermediate, 5-alkyl-2-thia-5-azabicyclo[2.2.1]heptan-3-one, the disclosed process achieves a continuous combination of three partial structures in a single reaction stage. This innovation addresses critical pain points in antibiotic manufacturing, including yield optimization, impurity control, and the feasibility of large-scale commercial production, making it a highly relevant technology for modern pharmaceutical supply chains seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbapenem antibacterial agents with 1-alkylpyrrolidine structures has been fraught with inefficiencies and scalability challenges. Prior art, such as the method described in JP-A-11-71277, relies on combining the three partial structures of the carbapenem molecule in distinct, sequential stages. This stepwise approach inherently increases the number of unit operations, leading to higher cumulative losses in yield and extended production timelines. Furthermore, literature references such as Heterocycles, Vol. 41, p. 147 (1995), describe methods where the nitrogen atom of the pyrrolidine ring is protected with a carbonyl group, specifically a p-nitrobenzyloxycarbonyl group. While this protection prevents side reactions by weakening nucleophilicity, it necessitates additional deprotection steps and limits the reactivity of the intermediate. Additionally, these conventional methods have historically been demonstrated only on extremely small scales, such as 315 mg with a 69% yield, raising serious doubts about their applicability to industrial manufacturing. The reliance on difficult-to-remove condensing agents like N,N'-dicyclohexylcarbodiimide (DCC) in intramolecular cyclization steps further complicates post-treatment and purification, rendering these older routes economically unviable for cost reduction in carbapenem manufacturing.
The Novel Approach
In stark contrast to the fragmented nature of conventional synthesis, the method disclosed in CN1486316A employs a unified one-pot strategy that significantly enhances process efficiency. The core of this innovation lies in the direct reaction of compound (1), a 5-alkyl-2-thia-5-azabicyclo[2.2.1]heptan-3-one derivative, with an amine compound (2) and a carbapenem precursor (3) within the same reaction vessel. Unlike prior methods that require nitrogen protection to manage nucleophilicity, this novel approach utilizes starting materials where the nitrogen atom retains its nucleophilic character, allowing for direct coupling without the burden of installing and removing protecting groups on the pyrrolidine nitrogen. This simplification not only reduces the number of chemical steps but also minimizes the generation of waste associated with protection-deprotection sequences. The process is designed to be robust, utilizing common bases like diisopropylethylamine or sodium bicarbonate and solvents such as dimethylformamide or dimethyl sulfoxide. By enabling the continuous combination of structural fragments, this method offers a pathway to high-purity carbapenem intermediates that is far more amenable to the commercial scale-up of complex antibiotic precursors required by global health markets.
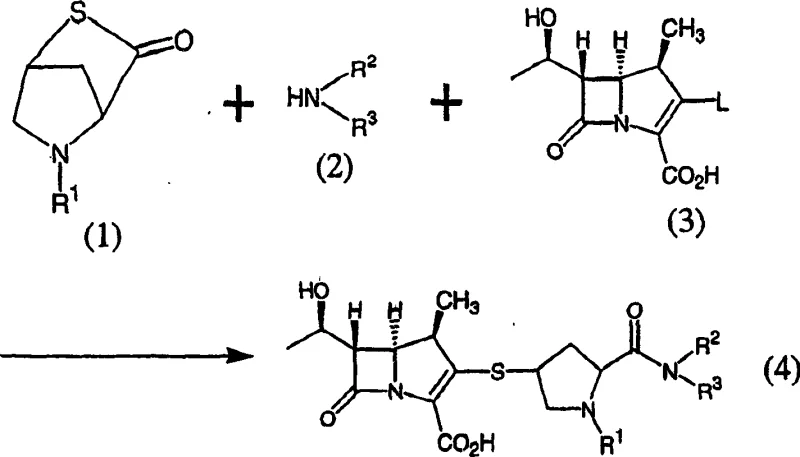
Mechanistic Insights into Intramolecular Cyclization and Coupling
The chemical elegance of this patent lies in the dual innovation of both the final coupling step (Method A) and the synthesis of the key bicyclic intermediate (Method B). Method A involves the nucleophilic attack of the amine component on the activated carbapenem structure, facilitated by the unique reactivity of the 5-alkyl-2-thia-5-azabicyclo[2.2.1]heptan-3-one scaffold. The reaction proceeds in the presence of a base, which neutralizes acidic byproducts and drives the equilibrium towards the formation of the amide bond linking the pyrrolidine side chain to the carbapenem core. The choice of solvent is critical here; polar aprotic solvents like DMF or DMSO are preferred to ensure solubility of the diverse reactants while maintaining the stability of the sensitive beta-lactam ring. The reaction temperature is carefully controlled between 10°C to 50°C to balance reaction kinetics with the prevention of thermal degradation, ensuring that the stereochemical integrity of the chiral centers is preserved throughout the transformation.
Equally important is Method B, which details the preparation of the essential intermediate compound (1). This step involves the intramolecular dehydration condensation of cis-2-carboxy-4-mercapto-1-alkylpyrrolidine (compound 5) in the presence of an acid anhydride. The mechanism proceeds through the activation of the carboxylic acid moiety by the anhydride, followed by nucleophilic attack from the proximal mercapto group to form the thia-azabicyclic ring system. A key advantage of this mechanistic pathway is the avoidance of difficult post-treatment reagents; by using acetic anhydride instead of carbodiimides, the byproduct is simply acetic acid, which is easily removed during workup. This significantly simplifies the purification process and reduces the risk of contaminating the final API with persistent organic impurities. The stereochemistry is rigorously maintained, with the (2S, 4S) configuration being preferred, ensuring that the resulting intermediate possesses the correct spatial arrangement for subsequent biological activity.
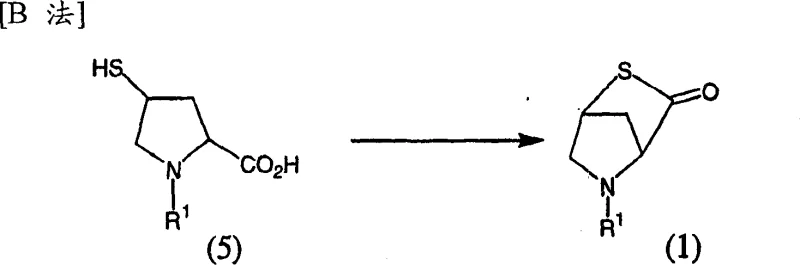
How to Synthesize 5-Alkyl-2-thia-5-azabicyclo[2.2.1]heptan-3-one Efficiently
The synthesis of the key bicyclic intermediate is a critical determinant of overall process success. The patent outlines a clear protocol starting from cis-2-carboxy-4-mercapto-1-alkylpyrrolidine derivatives, which can themselves be derived from readily available hydroxyproline sources. The process leverages standard organic transformations such as esterification, alkylation, sulfonylation, and thioester displacement to build the necessary functionality before the final cyclization event. This modular approach allows for flexibility in sourcing raw materials and optimizing each sub-step for maximum yield. The detailed standardized synthesis steps see the guide below provide a roadmap for replicating this high-efficiency route in a GMP-compliant environment.
- Prepare cis-2-carboxy-4-mercapto-1-alkylpyrrolidine or its salt as the starting material.
- React the starting material with an acid anhydride, preferably acetic anhydride, in a solvent such as acetic acid.
- Maintain reaction temperature between 30°C to 80°C for 1.5 to 5.0 hours to effect intramolecular dehydration condensation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from multi-step protected syntheses to this direct one-pot methodology represents a substantial opportunity for value creation. The elimination of nitrogen protecting groups removes entire categories of reagents and the associated waste disposal costs, directly impacting the cost of goods sold. Furthermore, the use of common, non-proprietary reagents like acetic anhydride and sodium bicarbonate ensures that the supply chain is not vulnerable to bottlenecks associated with specialized catalysts or exotic coupling agents. The robustness of the reaction conditions, operating at moderate temperatures and pressures, translates to lower energy consumption and reduced safety risks during manufacturing. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands for essential antibiotics without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in synthetic steps and the simplification of purification workflows. By avoiding the use of expensive and difficult-to-remove condensing agents like DCC, the process eliminates the need for complex chromatographic separations often required to purge urea byproducts. Additionally, the removal of nitrogen protection-deprotection sequences reduces the consumption of protecting group reagents and the solvents required for their handling. This streamlining leads to significant cost savings in raw material procurement and waste treatment, making the production of high-purity carbapenem intermediates more economically sustainable in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical antibiotic intermediates is paramount for public health security. This manufacturing method enhances reliability by utilizing starting materials that are chemically stable and commercially available in bulk quantities. The scalability of the process is evidenced by the patent examples, which demonstrate successful execution on scales ranging from hundreds of milligrams to over 400 grams, with yields consistently exceeding 80% in optimized examples. This proven scalability indicates that the technology can be readily transferred to multi-kilogram and ton-scale production facilities without the unforeseen engineering challenges often associated with scaling up delicate beta-lactam chemistry, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers distinct advantages. The substitution of hazardous condensing agents with benign acid anhydrides reduces the toxicity profile of the waste stream, facilitating easier compliance with increasingly stringent environmental regulations. The solvents employed, such as acetic acid, DMF, and DMSO, are well-understood within the industry and have established recovery and recycling protocols. The high atom economy of the cyclization step, where water is the primary byproduct of the ring closure, aligns with green chemistry principles. This environmental compatibility not only lowers regulatory hurdles but also positions manufacturers as responsible stewards of chemical safety, a key consideration for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and specifications provided in CN1486316A, offering clarity on the practical aspects of adopting this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement teams assessing the quality standards of potential suppliers.
Q: What is the primary advantage of the one-pot synthesis method described in CN1486316A?
A: The primary advantage is the ability to combine three partial structures of the carbapenem antibacterial agent in a single stage continuously, eliminating the need for stepwise combination and reducing processing time compared to conventional methods like those in JP-A-11-71277.
Q: How does this method improve scalability compared to prior art?
A: Unlike prior methods limited to small scales (e.g., 315 mg), this process utilizes robust reagents like acid anhydrides and avoids difficult post-treatment condensing agents like DCC, enabling production scales demonstrated up to 400g and beyond in the provided examples.
Q: What specific intermediate is critical for this synthesis route?
A: The critical intermediate is 5-alkyl-2-thia-5-azabicyclo[2.2.1]heptan-3-one (Compound 1), which is synthesized via intramolecular cyclization of cis-2-carboxy-4-mercapto-1-alkylpyrrolidine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
The technological advancements detailed in CN1486316A underscore the complexity and sophistication required to produce modern antibiotic intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this one-pot synthesis can be realized at the volumes required by the global market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbapenem intermediate meets the highest international standards for safety and efficacy, providing our partners with absolute confidence in the quality of their supply chain.
We invite pharmaceutical companies and contract manufacturers to explore the potential of this advanced synthesis route for their product portfolios. By leveraging our capabilities, you can achieve significant operational efficiencies and secure a stable supply of critical materials. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this technology can drive value and reliability for your organization.
