Advanced Z-Isomer Pathway for Ezetimibe Manufacturing and Commercial Scale-Up
Advanced Z-Isomer Pathway for Ezetimibe Manufacturing and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and high-yielding synthetic routes for critical lipid-lowering agents like Ezetimibe. A pivotal advancement in this domain is disclosed in patent CN102112430B, which introduces a novel synthesis strategy centered around Z-isomeric intermediates. Unlike traditional methods that rely on E-isomers and suffer from cumulative yield losses and complex purification challenges, this innovative approach leverages the unique physicochemical properties of Z-5-(4-fluorophenyl)-pent-4-enoic acid derivatives. By fundamentally altering the stereochemical trajectory of the synthesis from the initial Wittig olefination step, the process achieves a surprising increase in total recovery while simplifying the isolation of key intermediates. For R&D directors and process chemists, this represents a significant opportunity to optimize the manufacturing of Ezetimibe intermediates, ensuring higher purity and reduced operational complexity in large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ezetimibe has heavily relied on E-isomeric precursors, a route extensively documented in prior art such as US-A-5856473 and related literature. These conventional pathways are plagued by inherent inefficiencies, particularly during the oxidation and reduction phases of the side chain modification. When starting from E-olefin intermediates, the oxidation step to form the corresponding ketone typically yields only between 70% and 80%, while the subsequent stereoselective reduction to the hydroxy-propyl derivative drops precipitously to approximately 42%. Furthermore, the physical properties of E-isomers often lead to oily residues or difficult-to-crystallize mixtures, necessitating extensive chromatographic purification that is impractical for multi-kilogram or ton-scale manufacturing. The accumulation of these yield losses results in a dismal overall recovery rate, often cited in combined literature methods as low as 0.07% to 0.25%, creating substantial cost burdens and supply chain vulnerabilities for API manufacturers.
The Novel Approach
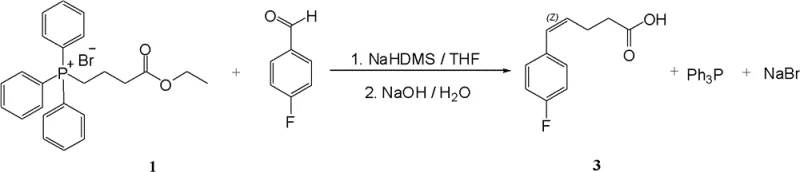
In stark contrast, the novel methodology described in CN102112430B utilizes a Z-alkene configured precursor that fundamentally transforms the efficiency of the synthetic sequence. By employing a specialized Wittig reaction, the process generates a mixture of Z and E isomers where the Z-isomer is dominant, with a Z:E ratio reaching approximately 88:12 and a total reaction yield exceeding 90%. This Z-configured intermediate exhibits superior crystallinity and solubility profiles, allowing for easy purification via recrystallization rather than column chromatography. Downstream, the Z-alkene oxidation step achieves an impressive 86% yield, and the critical chiral reduction step skyrockets to 95% yield, drastically outperforming the E-analogue route. This strategic shift not only boosts the total synthesis recovery to roughly 24.7% but also ensures a cleaner impurity profile by minimizing the formation of unwanted E-isomer byproducts that are difficult to remove in later stages.
Mechanistic Insights into Z-Selective Wittig Olefination
The cornerstone of this improved synthesis lies in the mechanistic execution of the Wittig reaction using specific phosphonium salts (Formula VI or VII). The reaction between 4-fluorobenzaldehyde and the phosphonium ylide is carefully controlled to favor the formation of the Z-alkene geometry. Typically, unstabilized ylides favor Z-alkenes, but the specific conditions outlined—utilizing bases like NaHMDS in THF at controlled temperatures—ensure high selectivity. The resulting Z-5-(4-fluorophenyl)-pent-4-enoic acid (or its ester derivative) serves as a robust building block. The mechanism avoids the thermodynamic equilibration that often leads to E-isomer dominance in other contexts. By locking in the Z-configuration early, the subsequent activation of the carboxylic acid (via oxalyl chloride) and coupling with chiral auxiliaries (such as oxazolidinones) proceeds with high fidelity. This precise stereocontrol is vital because the spatial arrangement of the double bond influences the diastereoselectivity of the subsequent aldol-type condensation with the imine component, ultimately dictating the stereochemistry of the azetidinone ring formation.
Impurity control is intrinsically built into this mechanistic design. In conventional E-routes, the proximity of the double bond to the carbonyl in the oxidation step can lead to over-oxidation or isomerization side reactions. However, the Z-configuration in this novel route appears to sterically shield the reactive centers appropriately during the Pd-catalyzed oxidation and borane reduction steps. The patent data indicates that the Z-intermediate allows for a 'cleaner' reaction profile, meaning fewer side products are generated that would otherwise co-elute with the desired API. This is particularly crucial for the final deprotection step, where residual E-isomers could lead to the formation of deprotected 4-hydroxy phenyl analogues that are structurally similar to Ezetimibe and difficult to separate. By maintaining Z-fidelity, the process ensures that the final active pharmaceutical ingredient meets rigorous regulatory standards for related substances without requiring excessive reprocessing.
How to Synthesize Ezetimibe Intermediates Efficiently
The synthesis of high-purity Ezetimibe intermediates via this Z-selective route involves a streamlined sequence of well-defined chemical transformations suitable for industrial scale-up. The process begins with the preparation of the phosphonium salt followed by the key Wittig olefination to establish the carbon backbone with the correct Z-stereochemistry. Subsequent steps involve amide coupling, imine condensation, and cyclization to form the beta-lactam core, followed by side-chain functionalization. The detailed standardized synthetic steps, including specific reagent stoichiometry, temperature profiles, and workup procedures for each stage, are outlined in the guide below to assist process development teams in replicating this high-yield pathway.
- Perform a Wittig reaction between 4-fluorobenzaldehyde and a specific phosphonium salt (Formula VI or VII) to generate the Z-alkene configured acid or ester intermediate with high Z/E selectivity.
- Activate the Z-acid intermediate and couple it with a chiral auxiliary (such as an oxazolidinone) to form the amide, followed by condensation with an imine to establish the azetidinone ring structure.
- Execute the final cyclization, oxidation, and stereoselective reduction steps, concluding with catalytic hydrogenation to remove protecting groups and yield high-purity Ezetimibe.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Z-isomer synthesis route offers compelling economic and logistical advantages over legacy methods. The primary driver of value is the drastic simplification of the purification train. Because the Z-intermediates possess favorable crystallization properties, the reliance on expensive and time-consuming preparative chromatography is significantly reduced or eliminated. This translates directly into lower solvent consumption, reduced waste disposal costs, and shorter batch cycle times. Furthermore, the substantial improvement in overall yield means that less raw material is required to produce the same amount of final API, effectively lowering the cost of goods sold (COGS) without compromising on quality. The robustness of the reaction conditions also implies a more reliable supply chain, as the process is less sensitive to minor variations in operating parameters that might cause batch failures in more finicky E-isomer routes.
- Cost Reduction in Manufacturing: The elimination of complex separation processes and the reduction in raw material waste due to higher yields create a leaner manufacturing cost structure. By avoiding the low-yield reduction steps associated with E-isomers, the process maximizes the throughput of existing reactor capacity, allowing for significant operational expenditure savings. Additionally, the use of commercially available starting materials like 4-fluorobenzaldehyde ensures that input costs remain stable and predictable, shielding the supply chain from volatility associated with exotic or custom-synthesized reagents.
- Enhanced Supply Chain Reliability: The high selectivity of the Wittig reaction and the crystallinity of the intermediates ensure consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability is critical for maintaining continuous supply to downstream formulation partners. The process is designed to be scalable from kilogram to multi-ton quantities without losing efficiency, meaning that suppliers can confidently commit to long-term volume agreements. The simplified workflow also reduces the dependency on specialized equipment, making it easier to qualify multiple manufacturing sites for redundancy.
- Scalability and Environmental Compliance: The reduction in solvent usage and waste generation aligns with modern green chemistry principles and stringent environmental regulations. Fewer purification steps mean a smaller environmental footprint, which is increasingly important for pharmaceutical companies aiming to meet sustainability goals. The process avoids the use of hazardous reagents where possible and utilizes standard workup procedures that are easily managed in existing wastewater treatment facilities. This environmental compliance facilitates faster regulatory approvals and reduces the administrative burden associated with environmental reporting and auditing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Z-isomer synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: Why is the Z-isomer pathway preferred over the traditional E-isomer route for Ezetimibe?
A: The Z-isomer pathway significantly improves total synthesis yield (up to 24.7% vs <1% in some prior art combinations) and offers superior physical properties for purification, avoiding the complex mixtures often associated with E-isomer oxidation and reduction steps.
Q: What represents the key technological breakthrough in this synthesis method?
A: The core breakthrough is the use of a modified Wittig reaction that selectively produces the Z-alkene configuration (Z:E ratio approx. 88:12) directly, eliminating the need for difficult isomer separation and enabling higher yields in downstream cyclization and reduction steps.
Q: How does this process impact the impurity profile of the final API?
A: By strictly controlling the Z-configuration throughout the synthesis, the formation of E-isomer impurities and their corresponding deprotected analogues is minimized, ensuring a cleaner final product that meets stringent pharmaceutical purity specifications without extensive chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of pharmaceutical intermediates. Our team of expert process chemists has thoroughly analyzed the Z-isomer pathway described in CN102112430B and possesses the technical capability to implement this advanced methodology immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Ezetimibe intermediate we deliver adheres to the highest global standards.
We invite you to collaborate with us to leverage this high-yield technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this Z-selective route can optimize your budget. Please contact us today to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our intermediates. Let us be your partner in driving down costs while elevating the quality of your lipid-lowering medication portfolio.
