Advanced Stereoselective Synthesis of Rivastigmine Intermediates for Commercial Scale-up
Advanced Stereoselective Synthesis of Rivastigmine Intermediates for Commercial Scale-up
The global demand for effective treatments for neurodegenerative disorders, particularly Alzheimer's disease, continues to drive innovation in the pharmaceutical intermediate sector. Rivastigmine, a potent acetylcholinesterase and butyrylcholinesterase inhibitor, remains a cornerstone therapy for managing mild to moderate dementia. However, the commercial viability of such active pharmaceutical ingredients (APIs) is heavily dependent on the efficiency and stereoselectivity of their synthetic routes. Patent CN103073456B introduces a groundbreaking preparation method for a critical rivastigmine intermediate, specifically 3-((S)-1-((S)-1-phenylethylamino)ethyl)phenyl ethyl (methyl) carbamate. This technology addresses long-standing challenges in chiral synthesis by offering a route that is not only operationally simpler but also delivers exceptional purity and yield, positioning it as a vital asset for any reliable pharmaceutical intermediate supplier seeking to optimize their supply chain.
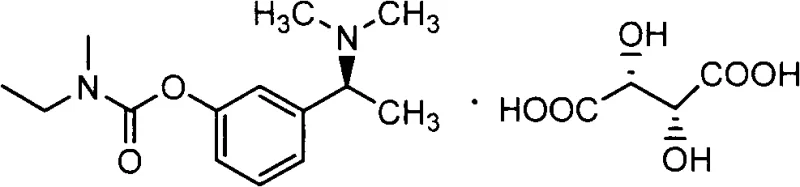
The significance of this patent lies in its ability to bypass the inefficiencies of traditional racemic resolution. By employing a highly stereoselective asymmetric synthesis strategy, the method ensures that the biologically active (S)-enantiomer is produced directly, minimizing waste and maximizing throughput. For R&D directors and process chemists, understanding the nuances of this transformation is key to implementing cost reduction in API manufacturing. The following analysis dissects the technical merits of this approach, contrasting it with conventional methods and highlighting its potential for industrial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral amines like rivastigmine has relied heavily on the resolution of racemic mixtures. As detailed in various prior art references such as German patent DE 3805744 and International Application WO 2005061446, these methods involve synthesizing the racemate and subsequently separating the enantiomers. This approach is inherently flawed from an economic and environmental standpoint because the maximum theoretical yield for the desired enantiomer is capped at 50%. Furthermore, achieving satisfactory optical purity often necessitates repeated recrystallization steps, which leads to substantial material loss and increased solvent consumption. The cumulative effect of these inefficiencies results in a high production cost for the final API, creating a bottleneck for manufacturers aiming to provide affordable medication. Additionally, the reliance on chiral transition metal catalysts in some asymmetric synthesis attempts, as seen in WO 2005058804, introduces further complications regarding catalyst cost, removal of heavy metal residues, and stringent regulatory compliance for residual metals in the final drug product.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN103073456B utilizes a direct asymmetric reductive amination strategy that fundamentally alters the production landscape. The core innovation involves the reaction of 3-acetyl phenyl ethyl (methyl) carbamate with (S)-1-phenylethylamine in the presence of titanium isopropoxide and palladium carbon under a hydrogen atmosphere. This one-pot transformation effectively installs the chiral center with high fidelity, leveraging the chirality of the inexpensive phenylethylamine auxiliary to direct the stereochemical outcome. The process operates under mild conditions, typically between 20-25°C and at a moderate hydrogen pressure of 1.5 MPa, which significantly reduces energy consumption and safety risks associated with high-pressure reactors. By achieving yields as high as 98.9% and purities exceeding 99.6% in optimized embodiments, this novel approach eliminates the yield ceiling of resolution methods and simplifies the downstream purification process, making it an ideal candidate for the commercial scale-up of complex chiral intermediates.
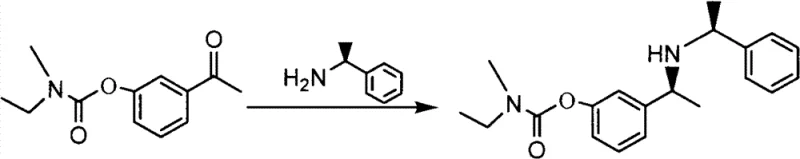
Mechanistic Insights into Titanium-Promoted Asymmetric Reductive Amination
The success of this synthetic route hinges on the synergistic interaction between the Lewis acid catalyst and the heterogeneous hydrogenation catalyst. Titanium isopropoxide serves a dual function: firstly, it activates the carbonyl group of the ketone substrate, facilitating the nucleophilic attack by the chiral amine to form an imine or iminium intermediate. Secondly, it likely coordinates with the nitrogen lone pair, rigidifying the transition state and enhancing the diastereoselectivity of the subsequent hydrogenation step. The use of 5% palladium on carbon as the hydrogenation catalyst is particularly advantageous due to its robustness and ease of removal via simple filtration. Unlike homogeneous catalysts which require complex workup procedures to meet strict ppm limits for metal residues, the heterogeneous Pd/C system allows for a clean separation, ensuring the final product meets rigorous quality standards without the need for specialized scavenging resins. This mechanistic elegance translates directly into process reliability, as the reaction demonstrates remarkable tolerance and consistency across different batches when performed in tetrahydrofuran (THF).
Impurity control is another critical aspect where this mechanism excels. The specificity of the titanium-mediated condensation minimizes the formation of side products such as over-alkylated amines or reduced alcohols, which are common pitfalls in reductive amination chemistry. The patent data indicates that maintaining the reaction temperature within the narrow window of 20-25°C is crucial; deviations to higher temperatures (e.g., 50°C) or the use of less active solvents like toluene or ethanol lead to a marked decrease in both yield and purity. This suggests that the kinetic control of the imine formation and reduction is tightly coupled to the thermal conditions, preventing the erosion of stereochemical integrity. Consequently, the resulting intermediate possesses a clean impurity profile, which is essential for the subsequent steps in the rivastigmine synthesis pathway, ultimately safeguarding the quality of the final drug substance.
How to Synthesize 3-((S)-1-((S)-1-phenylethylamino)ethyl)phenyl ethyl (methyl) carbamate Efficiently
The practical implementation of this synthesis requires precise adherence to the optimized parameters identified in the patent embodiments to ensure reproducibility and high quality. The procedure begins with the preparation of the starting material, 3-acetyl phenyl ethyl (methyl) carbamate, which can be synthesized via the reaction of m-hydroxy acetophenone with ethyl methyl carbamoyl chloride in the presence of a base, as illustrated in the preparatory examples. Once the ketone substrate is secured, the critical stereoselective step is executed in a pressurized reactor equipped with efficient agitation to ensure proper gas-liquid-solid contact. The choice of solvent is paramount, with THF demonstrating superior performance over other organic solvents in terms of reaction rate and product quality. Following the hydrogenation, the workup involves a straightforward aqueous workup and extraction protocol that avoids the use of chromatographic purification, thereby enhancing the process's suitability for large-scale manufacturing. Detailed standardized operating procedures for this synthesis are outlined below to guide process engineers in replicating these results.
- Combine 3-acetyl phenyl ethyl (methyl) carbamate and (S)-1-phenylethylamine in tetrahydrofuran solvent with titanium isopropoxide and 5% palladium carbon.
- Introduce hydrogen gas and maintain the reaction at 20-25°C under 1.5 MPa pressure for 3.5 to 4 hours.
- Filter the catalyst, extract the product with ethyl acetate, wash with brine, dry over sodium sulfate, and concentrate to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from resolution-based methods to this direct asymmetric synthesis fundamentally reshapes the cost structure of rivastigmine production. By nearly doubling the theoretical yield compared to racemic splitting, the process drastically reduces the amount of raw materials required per kilogram of final product. This efficiency gain is compounded by the elimination of multiple recrystallization cycles, which not only saves on solvent costs but also reduces the volume of hazardous waste generated, aligning with increasingly stringent environmental regulations. Furthermore, the use of commodity chemicals like titanium isopropoxide and standard palladium carbon, rather than bespoke chiral ligands or precious metal complexes, insulates the supply chain from volatility in the pricing of specialized reagents. This stability is crucial for long-term contract planning and ensures a consistent supply of high-purity rivastigmine intermediates to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the avoidance of yield-limiting separation steps. Traditional resolution methods inherently discard half of the synthesized material, whereas this asymmetric route converts the majority of the starting ketone into the desired chiral intermediate. Additionally, the simplified downstream processing, which relies on extraction and crystallization rather than column chromatography, significantly lowers operational expenditures related to labor, equipment time, and consumables. The removal of expensive chiral catalysts further contributes to a leaner cost profile, making the final API more competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized chiral reagents. This method mitigates such risks by utilizing widely available starting materials and robust catalysts that are stocked by multiple global suppliers. The operational simplicity of the reaction, which proceeds at ambient temperatures and moderate pressures, reduces the likelihood of batch failures due to equipment malfunction or operator error. This reliability ensures that production schedules can be met consistently, reducing lead times for high-purity pharmaceutical intermediates and allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden bottlenecks, but this methodology is designed with industrial application in mind. The use of heterogeneous catalysis facilitates easy catalyst recovery and recycling, minimizing metal waste. Moreover, the high purity of the crude product reduces the burden on wastewater treatment facilities by limiting the discharge of organic impurities. The process adheres to green chemistry principles by maximizing resource efficiency and minimizing the use of hazardous substances, thereby simplifying the regulatory approval process for new manufacturing sites and ensuring long-term sustainability of the production line.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the specific data points and comparative advantages presented in the documentation. These inquiries address common concerns regarding process robustness, impurity management, and the specific role of reaction parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided are derived directly from the experimental embodiments and the summary of the invention, ensuring accuracy and relevance to real-world production scenarios.
Q: What are the primary advantages of this synthesis method over traditional resolution?
A: Unlike traditional racemic splitting which suffers from a maximum theoretical yield of 50% and requires multiple recrystallizations, this asymmetric reductive amination utilizes a chiral auxiliary to directly synthesize the desired stereoisomer with yields exceeding 97% and purity above 99%.
Q: Why is tetrahydrofuran (THF) preferred as the solvent in this process?
A: Experimental data within the patent indicates that THF provides superior reaction kinetics compared to ethyl acetate, toluene, or alcohols, allowing the reaction to complete in under 4 hours at mild temperatures while maintaining high stereoselectivity and product purity.
Q: How does this method impact the cost of Rivastigmine production?
A: By eliminating the need for expensive chiral transition metal catalysts and avoiding the material losses associated with resolution processes, this method significantly lowers the raw material and processing costs for producing high-purity Rivastigmine intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of CN103073456B can be realized on an industrial scale. We understand that the production of chiral intermediates requires stringent purity specifications and rigorous QC labs to verify enantiomeric excess and chemical purity at every stage. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this hydrogenation process, guaranteeing a supply of intermediates that meet the highest international standards for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this more efficient methodology. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your downstream processes. Together, we can drive down the cost of Alzheimer's treatments while ensuring a reliable and sustainable supply of this critical medicine.
