Advanced Aqueous Oxa-Michael Addition for Efficient Ester Manufacturing and Commercial Scale-Up
Advanced Aqueous Oxa-Michael Addition for Efficient Ester Manufacturing and Commercial Scale-Up
The chemical industry is constantly seeking more efficient and environmentally benign pathways to construct essential molecular scaffolds, particularly ester linkages which are ubiquitous in pharmaceuticals, agrochemicals, and functional materials. Patent CN111187160A introduces a transformative methodology for the synthesis of ester compounds that deviates significantly from classical approaches by leveraging an oxa-Michael addition strategy in an aqueous medium. This innovation utilizes readily available organic carboxylic acids and α,β-unsaturated ketones as starting materials, mediated by a simple sodium carbonate aqueous solution. By shifting the paradigm from traditional acid-catalyzed condensation to a base-mediated conjugate addition, this technology offers a robust platform for generating complex ester architectures with high atom economy. For R&D directors and process chemists, this represents a critical opportunity to streamline synthetic routes, while supply chain managers will recognize the potential for reduced raw material costs and simplified logistics associated with avoiding hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial and laboratory synthesis of ester compounds has relied heavily on Fischer esterification or reactions involving activated acid derivatives such as acid chlorides or anhydrides. These conventional pathways often impose severe operational constraints, including the necessity for strong mineral acids like sulfuric acid which pose significant corrosion risks to reactor vessels and require specialized metallurgy. Furthermore, these reactions frequently demand elevated temperatures, sometimes exceeding 160°C, to drive the equilibrium forward, leading to high energy consumption and potential thermal degradation of sensitive functional groups. The requirement to synthesize specific alcohol precursors adds additional steps to the supply chain, increasing both the lead time and the cumulative cost of goods sold. Additionally, the use of stoichiometric coupling reagents or dehydrating agents generates substantial chemical waste, complicating downstream purification and wastewater treatment protocols, which is increasingly problematic under modern environmental regulations.
The Novel Approach
In stark contrast, the method disclosed in CN111187160A utilizes a mild, base-catalyzed oxa-Michael addition that operates effectively in water, a solvent that is non-toxic, non-flammable, and universally available. This approach eliminates the need for pre-activating the carboxylic acid or preparing specific alcohol partners, as it directly couples the acid with an α,β-unsaturated ketone. The reaction conditions are remarkably gentle, often proceeding at room temperature or with mild heating up to 40°C, which preserves the integrity of thermally labile substrates and reduces energy overheads. The use of sodium carbonate as a cheap and safe base further enhances the economic viability of the process, removing the dependency on expensive transition metal catalysts or hazardous strong acids. This simplicity translates directly into a more streamlined workflow, where the reaction can be monitored easily and worked up using standard extraction techniques, thereby accelerating the timeline from bench-scale discovery to pilot production.
Mechanistic Insights into Aqueous Oxa-Michael Addition
The core of this synthetic breakthrough lies in the activation of the carboxylic acid nucleophile within an aqueous basic environment. Upon dissolution in the sodium carbonate solution, the organic carboxylic acid is deprotonated to form a carboxylate anion, which serves as a potent oxygen-centered nucleophile. This anion then undergoes a conjugate addition to the β-carbon of the α,β-unsaturated ketone electrophile. Unlike traditional Michael additions that often require anhydrous conditions to prevent hydrolysis or side reactions, this system leverages the unique properties of water to stabilize the ionic intermediates while maintaining high reactivity. The reaction proceeds through a concerted or stepwise mechanism where the carbon-oxygen bond is formed regioselectively at the beta-position, followed by protonation to yield the final saturated ester product containing a ketone functionality. This mechanistic pathway ensures high atom utilization, as all atoms from the starting materials are incorporated into the final product without the generation of stoichiometric byproducts like salts or water that typically plague condensation reactions.
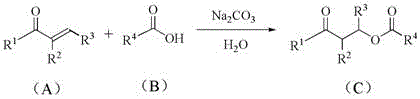
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed routes. The absence of strong acids prevents common side reactions such as dehydration, polymerization of the unsaturated ketone, or rearrangement of sensitive skeletal structures. The mild basicity of sodium carbonate is sufficient to drive the nucleophilic attack but not strong enough to induce aldol condensations or other base-sensitive degradations that might occur with stronger bases like sodium hydride or alkoxides. Furthermore, the aqueous medium acts as a heat sink, preventing localized hot spots that could lead to thermal runaway or the formation of tars. The result is a cleaner reaction profile with fewer side products, which simplifies the purification process and leads to higher isolated yields of the target ester. This level of selectivity is crucial for pharmaceutical applications where strict limits on genotoxic impurities and residual solvents must be maintained throughout the manufacturing lifecycle.
How to Synthesize Ester Compounds Efficiently
Implementing this synthesis protocol requires minimal specialized equipment, making it highly accessible for both laboratory research and industrial scale-up. The process begins with the preparation of a sodium carbonate aqueous solution with a concentration ranging from 0.1 to 1 M, which serves as both the solvent and the base source. The α,β-unsaturated ketone and the organic carboxylic acid are then added sequentially to this mixture, typically at room temperature, though slight heating to 40°C can be employed for less reactive substrates. The reaction progress is conveniently monitored via thin-layer chromatography (TLC) until the starting materials are fully consumed, indicating the completion of the oxa-Michael addition. Following the reaction, standard workup procedures involving extraction with ethyl acetate, washing with brine, and drying over anhydrous sodium sulfate are sufficient to isolate the crude product, which can then be purified by column chromatography if high purity is required for downstream applications.
- Prepare an aqueous sodium carbonate solution (0.1 to 1M concentration) in a reaction vessel at room temperature or slightly elevated temperatures up to 40°C.
- Sequentially add the alpha,beta-unsaturated ketone and the organic carboxylic acid to the reaction mixture and stir until TLC analysis confirms complete consumption of starting materials.
- Extract the reaction mixture with ethyl acetate, wash with water and brine, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous oxa-Michael addition technology presents a compelling value proposition centered on cost efficiency and operational resilience. The primary drivers of cost reduction stem from the elimination of expensive catalysts and the simplification of the raw material portfolio. By utilizing commodity chemicals like sodium carbonate and water instead of precious metal catalysts or specialized coupling reagents, the direct material costs are significantly lowered. Moreover, the ability to use a wide range of commercially available carboxylic acids and enones without derivatization reduces the number of SKUs that need to be managed and stored, streamlining inventory control. The mild reaction conditions also translate to lower utility costs, as there is no need for energy-intensive heating or cryogenic cooling systems, contributing to a leaner manufacturing budget and improved margin potential for high-volume ester intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound due to the substitution of high-cost reagents with inexpensive, bulk-available commodities. Traditional esterification often relies on thionyl chloride or carbodiimides which are costly and generate hazardous waste, whereas this method uses sodium carbonate which is fractionally cheaper and safer to handle. The removal of transition metal catalysts eliminates the need for expensive scavenging resins or complex filtration steps required to meet strict ppm limits for metal residues in pharmaceutical ingredients. Additionally, the high atom economy means that less raw material is wasted as byproduct, maximizing the yield per kilogram of input and reducing the overall cost of goods sold. These factors combine to create a manufacturing process that is inherently more cost-effective and less susceptible to price volatility in the specialty chemical market.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced by the reliance on robust, non-hazardous starting materials that are widely sourced globally. Strong acids and anhydrides often face stricter transportation regulations and storage requirements, which can lead to delays and increased logistics costs; in contrast, sodium carbonate and simple ketones are classified as low-risk materials. The simplicity of the reaction setup also means that production can be easily transferred between different manufacturing sites or contract organizations without the need for specialized corrosion-resistant reactors. This flexibility ensures continuity of supply even in the face of regional disruptions or equipment maintenance schedules. Furthermore, the shorter reaction times and simplified workup reduce the occupancy time of production vessels, increasing throughput capacity and allowing for faster response to fluctuating market demands.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the use of water as the primary solvent, which mitigates the fire and explosion risks associated with large volumes of flammable organic solvents. The aqueous waste stream is easier to treat and neutralize compared to acidic or organic-heavy waste, facilitating compliance with increasingly stringent environmental discharge regulations. The absence of heavy metals and halogenated solvents aligns perfectly with green chemistry initiatives and corporate sustainability goals, reducing the regulatory burden and potential liability associated with hazardous waste disposal. This environmental compatibility not only future-proofs the manufacturing process against tightening regulations but also enhances the brand reputation of the end-product as being sustainably produced, a factor that is becoming increasingly important to downstream customers and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this ester synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of what process teams can expect during technology transfer. Understanding these nuances is essential for risk assessment and for designing robust control strategies that ensure consistent product quality across different batches and scales of production.
Q: What are the primary advantages of this oxa-Michael addition method over traditional Fischer esterification?
A: Unlike traditional Fischer esterification which often requires strong mineral acids, high temperatures, and the preparation of alcohol precursors, this method utilizes mild aqueous sodium carbonate conditions at near-room temperature. It directly couples carboxylic acids with unsaturated ketones, eliminating the need for harsh acidic environments and simplifying the precursor supply chain.
Q: What is the substrate scope for the carboxylic acid component in this synthesis?
A: The method demonstrates excellent adaptability, accommodating aliphatic organic carboxylic acids, various substituted aryl carboxylic acids, and even amino acid derivatives. The substituents on aromatic rings can include alkyl, alkoxy, halogen, or nitro groups, allowing for significant structural diversity in the final ester products.
Q: How does the use of water as a solvent impact the environmental profile of the process?
A: Using water as the reaction medium significantly reduces the reliance on volatile organic solvents during the reaction phase, aligning with green chemistry principles. This aqueous approach minimizes hazardous waste generation and lowers the overall environmental footprint compared to processes requiring large volumes of organic solvents or corrosive acid catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ester Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic methodologies like the aqueous oxa-Michael addition to maintain a competitive edge in the global fine chemicals market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity ester intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether your project requires custom synthesis of complex building blocks or the optimization of existing routes for cost and sustainability, our CDMO capabilities are designed to support your specific technical requirements with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis platform can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your specific volume needs. We encourage you to contact us today to obtain specific COA data for our available ester catalog and to initiate route feasibility assessments for your proprietary targets. Let us partner with you to drive innovation, reduce costs, and secure your supply of critical chemical intermediates for the future.
