Advanced Synthesis of Monocyclic Beta-Lactam Intermediates via Quinoxaline Coupling
Introduction to Patent CN1251836A Technology
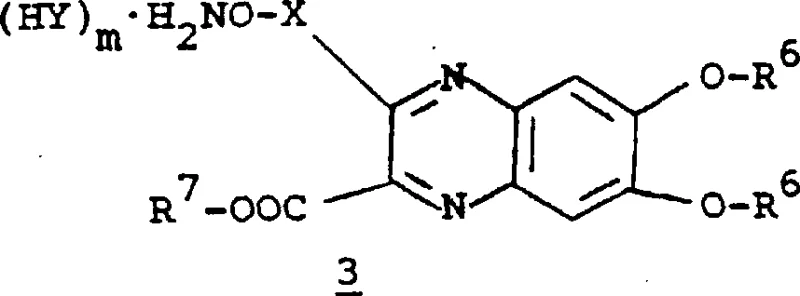
The pharmaceutical landscape for monocyclic beta-lactam antibiotics demands highly precise synthetic routes to ensure both efficacy and safety. Patent CN1251836A, a divisional application stemming from original filings in the early 1990s, presents a sophisticated methodology for preparing heteroaroyl derivatives of these critical antibiotic classes. This technology focuses on the construction of complex quinoxaline-based intermediates that serve as the acylating agents for the beta-lactam nucleus. The significance of this patent lies in its detailed exploration of protecting group strategies, specifically utilizing tert-butoxycarbonyl (BOC) and benzyl moieties to stabilize reactive hydroxylamine and phenolic groups during the synthesis. For R&D directors and process chemists, understanding these nuances is vital for replicating high-purity outcomes. The document outlines multiple pathways, including the coupling of formula 2 compounds with formula 3 intermediates, offering flexibility in manufacturing depending on the availability of starting materials. By leveraging these established protocols, manufacturers can achieve consistent quality in the production of advanced antibiotic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for beta-lactam side chains often suffer from significant drawbacks related to the instability of hydroxylamine intermediates and the difficulty in controlling regioselectivity during heterocyclic functionalization. In many conventional processes, the exposure of free hydroxyl groups or unprotected amines leads to premature oxidation or polymerization, drastically reducing overall yields and complicating purification. Furthermore, older methods frequently rely on harsh acidic or basic conditions that can degrade the sensitive beta-lactam ring if coupling occurs too early in the sequence. The lack of robust protecting groups in legacy methodologies often results in broad impurity profiles, necessitating expensive and time-consuming chromatographic separations that are not ideal for large-scale commercial operations. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher costs for the final active pharmaceutical ingredients.
The Novel Approach
The approach detailed in CN1251836A overcomes these historical hurdles by introducing a modular synthesis strategy centered on stable, protected quinoxaline derivatives. By employing protecting groups such as R6 (hydrogen or phenolic protectors like Si(t-Bu)2) and R7 (carboxyl protectors like tert-butyl), the patent ensures that reactive sites remain inert until the precise moment of coupling. This method allows for the isolation of stable intermediates, such as the N,N-diBOC-hydroxylamine derivatives, which can be stored and handled with greater safety and ease. The novel route also emphasizes pH-controlled coupling reactions, typically maintaining a pH of around 2.0 during the condensation of the quinoxaline acid with the beta-lactam amine, which significantly suppresses side reactions. This level of control translates to a more streamlined process with fewer downstream purification requirements, directly addressing the pain points of cost and efficiency faced by procurement teams.
Mechanistic Insights into Quinoxaline-Hydroxylamine Coupling
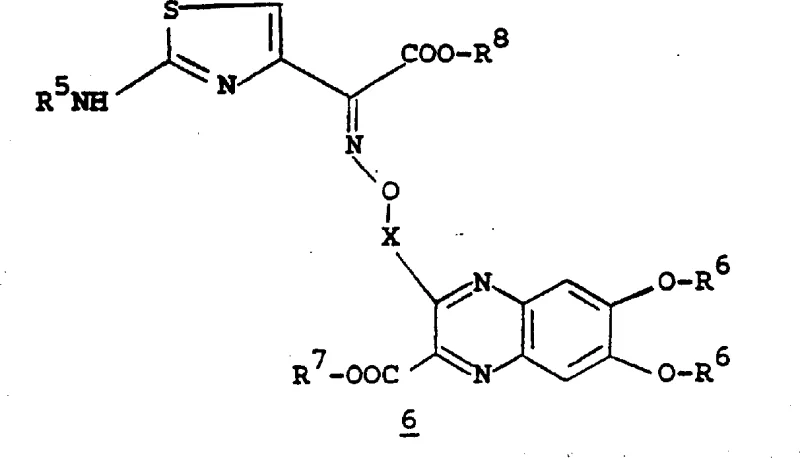
The core mechanistic advantage of this technology lies in the activation of the quinoxaline carboxylic acid and its subsequent reaction with the protected hydroxylamine functionality. As illustrated in the patent, the formation of the O-acyl hydroxylamine bond is critical for the biological activity of the final antibiotic. The process typically involves converting the quinoxaline carboxylic acid into a reactive species, often facilitated by coupling agents or through the formation of activated esters, although the patent also describes direct coupling under specific aqueous conditions. The presence of the N-oxide or specific substitution patterns on the quinoxaline ring influences the electronic density, thereby affecting the nucleophilicity of the coupling partner. Understanding these electronic effects is crucial for optimizing reaction kinetics and minimizing the formation of geometric isomers, as the patent notes that the Z-isomer generally exhibits superior antibacterial activity.
Impurity control is meticulously managed through the strategic use of protecting groups that are orthogonal to each other. For instance, the use of BOC groups on the hydroxylamine nitrogen prevents self-condensation, while benzyl or isopropylidene groups on the phenolic oxygens prevent unwanted etherification or oxidation. The deprotection steps are equally critical; the patent describes hydrolysis using concentrated hydrochloric acid at elevated temperatures (e.g., 80-85°C) to remove acetonide protections, followed by careful pH adjustment to precipitate the zwitterionic final products. This sequence ensures that the final intermediate is obtained in a highly pure crystalline form, suitable for direct use in the final antibiotic assembly without extensive recrystallization. Such mechanistic precision reduces the risk of batch-to-batch variability, a key concern for regulatory compliance.
How to Synthesize Quinoxaline Beta-Lactam Intermediates Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and reagent quality. The patent provides a comprehensive roadmap, starting from simple nitro-catechol precursors and building up to the complex quinoxaline system. Key to success is the maintenance of anhydrous conditions during the initial protection steps and the precise control of stoichiometry during the coupling phases. Operators must be vigilant regarding temperature gradients, especially during exothermic additions of reagents like thionyl chloride or during hydrogenation steps involving palladium catalysts. The detailed examples in the patent, such as Example 20, offer a preferred sequence that balances yield and purity, serving as a benchmark for process development teams aiming to scale this chemistry. Adhering to these standardized protocols ensures that the structural integrity of the beta-lactam mimic is preserved throughout the synthesis.
- Preparation of protected quinoxaline precursors via nitration, reduction, and condensation with dicarbonyl compounds.
- Functionalization of the quinoxaline methyl group to introduce a leaving group or hydroxylamine moiety.
- Coupling the quinoxaline intermediate with the beta-lactam side chain under controlled pH conditions followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN1251836A offers tangible benefits in terms of operational stability and cost efficiency. The ability to isolate stable, protected intermediates means that production can be decoupled into distinct stages, allowing for better inventory management and reduced risk of total batch loss due to a single step failure. This modularity enhances supply chain resilience, ensuring that critical raw materials can be sourced and stockpiled without the immediate pressure of degradation. Furthermore, the improved selectivity of the coupling reactions reduces the consumption of expensive solvents and chromatography media, leading to substantial cost savings in waste disposal and material usage. By minimizing the number of purification cycles required to meet stringent pharmaceutical specifications, manufacturers can significantly shorten the overall production cycle time.
- Cost Reduction in Manufacturing: The implementation of robust protecting group strategies eliminates the need for complex and costly purification steps often associated with unstable hydroxylamine chemistry. By preventing side reactions and polymerization, the process yield is maximized, reducing the amount of starting material required per kilogram of final product. This efficiency directly lowers the cost of goods sold (COGS) and improves the margin profile for the final antibiotic. Additionally, the use of common reagents and standard unit operations like filtration and crystallization makes the process economically viable for large-scale production without requiring specialized equipment.
- Enhanced Supply Chain Reliability: The stability of the protected intermediates allows for longer storage periods and easier transportation, mitigating the risks associated with just-in-time delivery of unstable compounds. This reliability is crucial for maintaining continuous production schedules for life-saving antibiotics. The patent's emphasis on aqueous workups and standard organic solvents ensures that the supply chain is not dependent on exotic or hard-to-source reagents, further securing the continuity of supply. Manufacturers can confidently plan production runs knowing that the intermediate quality remains consistent over time.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial manufacturing scales. The reduction in hazardous waste generation, achieved through higher selectivity and fewer purification steps, aligns with modern environmental, health, and safety (EHS) standards. This compliance reduces the regulatory burden and potential liabilities associated with chemical manufacturing. The process facilitates the commercial scale-up of complex pharmaceutical intermediates while adhering to strict environmental guidelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of these quinoxaline derivatives. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining the high purity standards required for pharmaceutical applications.
Q: What are the key challenges in synthesizing quinoxaline beta-lactam intermediates?
A: The primary challenges involve managing the stability of hydroxylamine functionalities and achieving high regioselectivity during the coupling of complex heterocyclic systems without degrading the sensitive beta-lactam ring.
Q: How does the patented method improve impurity profiles?
A: By utilizing specific protecting groups like tert-butoxycarbonyl (BOC) and benzyl groups, the method minimizes side reactions during the coupling phase, leading to a cleaner crude product that requires less intensive purification.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the patent describes conditions such as aqueous workups and standard chromatography that are adaptable to larger scales, provided that strict pH control and temperature management are maintained during the coupling steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry described in patents like CN1251836A is executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoxaline derivatives meets the exacting standards of our international partners. Our commitment to quality assurance means that you can rely on us for consistent supply, reducing the risk of production delays in your downstream API manufacturing.
We invite you to collaborate with us to optimize your supply chain for beta-lactam intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable source of advanced pharmaceutical intermediates that combine technical excellence with commercial viability, ensuring your projects move forward without interruption.
