Advanced Manufacturing Process for High-Purity Trifluoromethyl Imidazole Compounds in Pharmaceutical Supply Chains
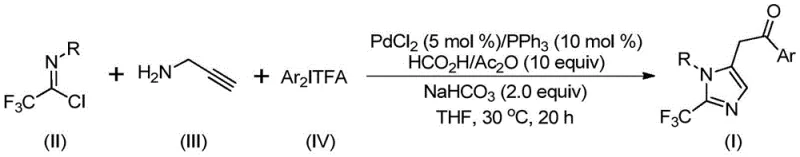
Patent CN111423381B introduces a transformative methodology for synthesizing structurally diverse 2-trifluoromethyl substituted imidazole compounds through an innovative palladium-catalyzed carbonylation process that operates under exceptionally mild conditions at only 30°C. This patented approach addresses critical limitations in traditional heterocyclic synthesis by utilizing readily accessible starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts without requiring expensive or hazardous reagents. The process demonstrates remarkable substrate tolerance across various aryl substitutions while achieving high yields up to 97% through optimized reaction kinetics that minimize side product formation. This advancement represents a significant leap forward in manufacturing efficiency for nitrogen-containing heterocycles that serve as crucial building blocks in pharmaceutical development pipelines. The methodology's compatibility with commercial scale-up protocols positions it as an ideal solution for meeting growing industry demands for high-purity trifluoromethylated intermediates in drug discovery and development while maintaining stringent quality control standards required by global regulatory agencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethylated imidazoles often rely on unstable or expensive trifluoromethyl synthons such as trifluorodiazoethane that require specialized handling procedures and generate significant safety hazards during manufacturing operations. These methods typically operate under harsh reaction conditions including elevated temperatures exceeding 80°C or highly acidic environments that promote unwanted side reactions and complicate purification processes through increased impurity profiles. The limited substrate scope of conventional approaches restricts structural diversity in final products while necessitating multiple protection-deprotection steps that substantially increase production costs and reduce overall process efficiency. Furthermore, existing methodologies frequently employ transition metal catalysts requiring complex removal protocols that introduce additional processing stages and create potential contamination risks for pharmaceutical applications where metal residues must be strictly controlled below regulatory thresholds.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that utilizes commercially available trifluoroethylimidoyl chloride as a stable precursor under mild reaction conditions at precisely controlled ambient temperatures of only 30°C. This innovative approach eliminates the need for hazardous trifluoromethyl synthons while demonstrating exceptional functional group tolerance across diverse aryl substitutions including halogenated and electron-donating moieties without requiring protective groups. The streamlined reaction sequence integrates carbon monoxide generation directly from formic acid/acetic anhydride mixtures within the same reaction vessel, significantly reducing process complexity compared to traditional multi-step syntheses that require external gas handling systems. Crucially, the methodology achieves high yields up to 97% with minimal byproduct formation through optimized catalyst loading ratios that maintain excellent selectivity while enabling straightforward purification via standard column chromatography techniques.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism proceeds through a sophisticated sequence beginning with intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate to generate a trifluoroacetamidine intermediate that undergoes spontaneous isomerization under mild thermal conditions. Subsequent palladation of propargylamine forms an alkenyl palladium species that isomerizes to an alkyl palladium intermediate before undergoing carbonylation using carbon monoxide released in situ from formic acid/acetic anhydride mixtures. The diaryl iodonium salt then participates in oxidative addition to form a tetravalent palladium complex that facilitates reductive elimination to yield the final trifluoromethyl imidazole product with precise regioselectivity at the two-position. This cascade mechanism operates through carefully balanced catalytic cycles where the palladium chloride/triphenylphosphine system maintains optimal oxidation states throughout the transformation while minimizing unwanted β-hydride elimination pathways that could lead to impurities.
Impurity control is achieved through multiple mechanistic safeguards including the inherent selectivity of the carbonylation step that prevents over-reaction pathways common in alternative methodologies. The mild reaction temperature of only 30°C suppresses thermal decomposition routes while the precise stoichiometric control of sodium bicarbonate additive prevents unwanted hydrolysis side reactions that could generate carboxylic acid impurities. Substrate design flexibility allows strategic placement of substituents that sterically hinder potential dimerization pathways while electronic effects from aryl groups stabilize key intermediates throughout the catalytic cycle. This comprehensive impurity management strategy results in exceptionally clean reaction profiles that consistently deliver products meeting pharmaceutical purity requirements without requiring additional purification stages beyond standard chromatography.
How to Synthesize Trifluoromethyl Imidazole Efficiently
This patented methodology provides an optimized pathway for manufacturing high-purity trifluoromethyl imidazole compounds through a carefully designed sequence that integrates multiple synthetic transformations into a single operational step while maintaining exceptional control over product quality attributes. The process leverages commercially available starting materials and standard laboratory equipment to achieve remarkable efficiency gains compared to conventional multi-step approaches that require specialized infrastructure. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in the patent literature; these protocols are designed to ensure consistent product quality across different production scales while accommodating variations in raw material specifications common in industrial settings. The following step-by-step guide outlines the critical operational parameters required for successful implementation of this innovative manufacturing approach.
- Prepare the reaction mixture by combining palladium chloride catalyst (5 mol%), triphenylphosphine ligand (10 mol%), sodium bicarbonate additive (2.0 equiv), trifluoroethylimidoyl chloride substrate, propargylamine, and diaryl iodonium salt in tetrahydrofuran solvent under inert atmosphere.
- Maintain precise reaction conditions at a controlled temperature of 30°C for exactly twenty hours to ensure complete conversion while preventing thermal degradation or side product formation through optimized kinetic control.
- Execute post-reaction processing including filtration through silica gel matrix followed by column chromatography purification to isolate high-purity products meeting stringent pharmaceutical quality specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This advanced manufacturing methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional heterocyclic synthesis routes through inherent process improvements that translate directly into operational efficiencies. The elimination of specialized equipment requirements and hazardous reagent handling protocols significantly reduces capital investment needs while enhancing workplace safety profiles compared to conventional approaches that necessitate dedicated infrastructure for gas handling or cryogenic operations. By utilizing cost-effective commercial raw materials available from multiple global suppliers with established quality management systems, the process inherently improves supply chain resilience while reducing dependency on single-source vendors that could create potential disruption risks during periods of market volatility or geopolitical instability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts through optimized palladium chloride usage combined with simplified purification requirements due to inherently clean reaction profiles results in substantial operational cost savings without compromising product quality standards required by regulatory authorities. The use of readily available formic acid/acetic anhydride mixtures as carbon monoxide sources avoids costly gas cylinder management systems while maintaining excellent process control throughout manufacturing operations.
- Enhanced Supply Chain Reliability: Strategic selection of globally available starting materials including commercially sourced aryl boronic acids and iodides enables robust supplier diversification strategies that mitigate single-point failure risks while ensuring consistent material availability across different geographic regions. The process's tolerance to minor raw material variations provides additional flexibility during supply chain disruptions without requiring revalidation procedures that could delay production schedules.
- Scalability and Environmental Compliance: The ambient temperature operation at only 30°C significantly reduces energy consumption compared to traditional high-temperature processes while eliminating hazardous waste streams associated with unstable reagents used in alternative methodologies. This environmentally favorable profile aligns with modern sustainability initiatives while enabling seamless scale-up from laboratory validation to commercial production volumes through straightforward process intensification strategies that maintain consistent product quality attributes.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation requirements and performance characteristics observed during extensive validation studies across multiple production scales. These insights have been compiled from practical experience applying this innovative approach to real-world manufacturing scenarios while maintaining strict adherence to regulatory quality standards required for pharmaceutical intermediates.
Q: How does this methodology overcome traditional challenges in trifluoromethylated heterocycle synthesis?
A: The patented process eliminates reliance on unstable trifluoromethyl synthons by utilizing readily available trifluoroethylimidoyl chloride precursors under mild palladium-catalyzed conditions at ambient temperature, significantly improving substrate compatibility while avoiding hazardous reagents required in conventional approaches.
Q: What specific advantages does this technology offer for pharmaceutical quality control?
A: The inherent reaction selectivity minimizes byproduct formation through precise control of carbonylation pathways and substrate design flexibility, enabling direct production of high-purity intermediates that meet stringent regulatory requirements without extensive purification steps.
Q: How does this innovation enhance supply chain resilience for procurement teams?
A: By leveraging cost-effective commercial raw materials and eliminating specialized equipment requirements through simplified process chemistry, the methodology drastically reduces lead times while improving supply reliability through robust scalability from laboratory to manufacturing scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Imidazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-billion levels. This patented methodology represents just one example of our commitment to developing innovative solutions that address complex synthetic challenges faced by global pharmaceutical manufacturers seeking reliable sources for high-value intermediates requiring exceptional purity profiles and consistent supply chain performance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements while demonstrating how our expertise can optimize your supply chain performance through scientifically validated process improvements.
