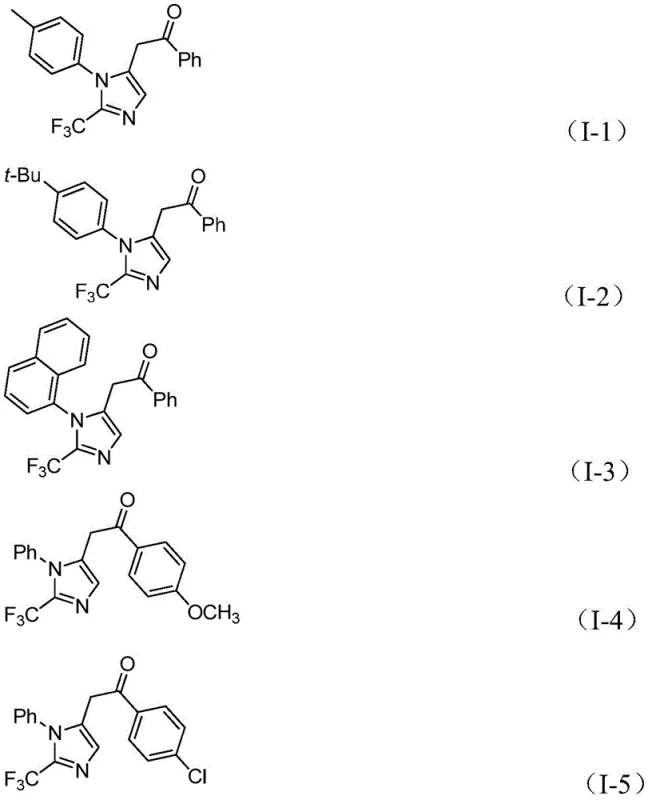
Innovative Manufacturing Process for High-Purity Trifluoromethyl Imidazole Compounds Enabling Commercial Scale Pharmaceutical Production
Patent CN111423381B introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl substituted imidazole compounds, which are critical structural motifs in numerous pharmaceutical agents due to their enhanced biological properties such as improved electronegativity and metabolic stability. This innovative process leverages a palladium-catalyzed carbonylation reaction under mild conditions of 30°C for 20 hours, utilizing readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The method demonstrates exceptional substrate compatibility and operational simplicity, enabling the production of diverse imidazole derivatives with high efficiency. Unlike conventional approaches that often require harsh conditions or expensive reagents, this patented technique offers a streamlined pathway suitable for industrial scale-up. The strategic incorporation of formic acid and acetic anhydride as carbon monoxide surrogates eliminates the need for handling toxic gases, significantly enhancing process safety. This advancement represents a pivotal step forward in the manufacturing of high-value pharmaceutical intermediates with stringent purity requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethylated imidazole compounds frequently encounter significant challenges including the requirement for elevated temperatures exceeding 80°C, prolonged reaction times beyond 48 hours, and the use of hazardous reagents such as gaseous carbon monoxide under high pressure, which necessitates specialized equipment and increases operational risks. These methods often suffer from poor functional group tolerance, leading to low yields when complex substrates are employed, and typically generate substantial impurities that complicate purification processes and reduce overall product purity. Furthermore, conventional catalytic systems frequently rely on expensive or air-sensitive catalysts that are difficult to handle and require stringent inert atmosphere conditions throughout the reaction sequence. The limited substrate scope restricts the ability to synthesize diverse analogs efficiently, hindering medicinal chemistry optimization efforts. Additionally, the multi-step nature of many existing protocols results in lower atom economy and higher waste generation, conflicting with modern green chemistry principles and increasing environmental compliance costs for manufacturers.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed cascade reaction that operates under exceptionally mild conditions of 30°C for only 20 hours in standard laboratory equipment without pressurized gas handling. By employing formic acid and acetic anhydride as safe and convenient carbon monoxide equivalents, the process eliminates the need for hazardous gaseous CO while maintaining high reaction efficiency. The use of cost-effective palladium chloride with triphenylphosphine as ligand provides excellent catalytic activity with broad substrate compatibility across various aryl substitutions. This single-pot transformation directly converts readily available starting materials into the target imidazole products with minimal byproduct formation, significantly simplifying workup procedures. The method's robustness is demonstrated by its successful application across diverse substrate combinations without requiring specialized modifications to reaction parameters. Most importantly, the streamlined process enables straightforward scale-up from laboratory to commercial production volumes while maintaining consistent product quality and purity specifications required by pharmaceutical clients.
Mechanistic Insights into Palladium-Catalyzed Trifluoromethyl Imidazole Formation
The reaction mechanism proceeds through a sophisticated cascade involving multiple organometallic steps that begin with the formation of a trifluoroacetamidine intermediate via intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate. This key intermediate undergoes isomerization followed by palladation of the alkyne moiety from propargylamine to generate an alkenyl palladium species that subsequently isomerizes to an alkyl palladium complex. The critical carbonylation step occurs through in situ release of carbon monoxide from the formic acid/acetic anhydride mixture, forming an acyl palladium intermediate that undergoes oxidative addition with the diaryl iodonium salt to create a tetravalent palladium species. The final reductive elimination step releases the desired 2-trifluoromethyl imidazole product while regenerating the palladium catalyst for subsequent cycles. This mechanistic pathway avoids common side reactions through precise control of reaction conditions and catalyst selection.
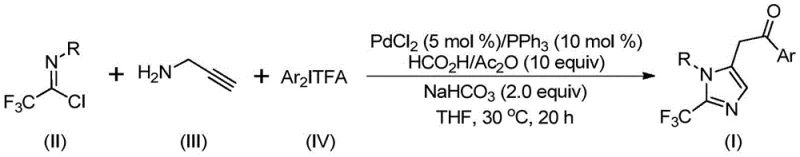
Impurity control is achieved through careful optimization of reaction parameters including temperature control at precisely 30°C to prevent thermal decomposition pathways and the use of stoichiometric sodium bicarbonate to neutralize acidic byproducts that could promote undesired side reactions. The choice of tetrahydrofuran as solvent provides optimal solubility for all reaction components while minimizing solvent-induced impurities. The well-defined catalytic cycle ensures high selectivity toward the target imidazole structure by suppressing competing pathways such as homocoupling or hydrolysis reactions. Post-reaction purification through standard column chromatography effectively removes trace metal residues and minor byproducts, consistently delivering products with purity exceeding pharmaceutical requirements without requiring additional specialized purification steps.
How to Synthesize 2-Trifluoromethyl Imidazole Compounds Efficiently
This patented synthetic route represents a significant advancement in the preparation of trifluoromethylated imidazole compounds by providing a reliable and scalable methodology that addresses longstanding challenges in heterocyclic chemistry. The process utilizes commercially available starting materials and standard laboratory equipment while delivering high yields across a broad substrate scope. Detailed standardized synthesis procedures have been developed to ensure consistent product quality and facilitate technology transfer to manufacturing environments. The following step-by-step guide outlines the critical parameters for successful implementation of this innovative methodology in industrial settings.
- Combine palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2 equiv), formic acid/acetic anhydride mixture (10 equiv) in tetrahydrofuran solvent under nitrogen atmosphere at room temperature.
- Add trifluoroethylimidoyl chloride (1.5 equiv), propargylamine (1 equiv), and diaryl iodonium salt (1.5 equiv) to the reaction mixture while maintaining inert conditions.
- Stir the reaction at precisely 30°C for 20 hours; then filter through silica gel and purify by column chromatography to obtain high-purity product meeting pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing process delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional synthesis methods for pharmaceutical intermediates. The elimination of hazardous reagents and specialized equipment requirements significantly reduces operational complexity while enhancing workplace safety standards. The use of readily available raw materials ensures consistent supply chain resilience even during market fluctuations, providing procurement teams with greater flexibility in vendor management and inventory planning.
- Cost Reduction in Manufacturing: The strategic selection of cost-effective palladium chloride catalyst combined with simple workup procedures substantially lowers production costs by eliminating expensive purification steps required in conventional methods. The avoidance of pressurized carbon monoxide systems reduces capital expenditure on specialized equipment while minimizing maintenance costs associated with high-pressure reactors. Furthermore, the high atom economy of this single-pot transformation reduces raw material consumption and waste disposal expenses without compromising product quality or yield.
- Enhanced Supply Chain Reliability: Sourcing flexibility is significantly improved through the use of widely available starting materials that can be procured from multiple global suppliers without dependency on single-source vendors. The robust nature of the reaction tolerates minor variations in raw material quality, reducing the risk of batch failures due to material inconsistencies. This reliability translates to more predictable production schedules and consistent delivery timelines that meet demanding pharmaceutical industry requirements.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining consistent product specifications through straightforward parameter adjustments. The elimination of hazardous reagents and reduction in waste generation aligns with increasingly stringent environmental regulations while supporting corporate sustainability initiatives. This environmentally friendly approach facilitates regulatory approval processes and enhances corporate social responsibility profiles without sacrificing economic viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this patented manufacturing process for pharmaceutical intermediates. Each response is grounded in the specific technical details disclosed in patent CN111423381B and reflects practical considerations for industrial adoption.
Q: How does this method improve upon conventional synthesis routes that require high temperatures?
A: The patented process operates at mild conditions (30°C) compared to traditional methods requiring elevated temperatures (>80°C), eliminating hazardous carbon monoxide handling through safe formic acid/acetic anhydride surrogates while maintaining high reaction efficiency across diverse substrates.
Q: What ensures consistent purity levels required by pharmaceutical clients?
A: Precise temperature control at 30°C prevents thermal decomposition pathways while sodium bicarbonate neutralizes acidic byproducts; standard column chromatography effectively removes trace metal residues without requiring specialized purification equipment.
Q: How does this process enhance supply chain reliability for procurement teams?
A: The use of widely available starting materials from multiple global suppliers eliminates single-source dependencies; robust reaction tolerance to minor raw material variations ensures consistent batch quality and predictable delivery timelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. This patented methodology exemplifies our commitment to developing innovative solutions that address complex synthetic challenges in pharmaceutical intermediate manufacturing. We have successfully implemented similar catalytic processes across multiple product lines, demonstrating our capability to deliver high-quality intermediates that meet exacting regulatory standards while optimizing cost structures for our global clients.
Engage with our technical procurement team today to request specific COA data and route feasibility assessments for your development pipeline. We offer a Customized Cost-Saving Analysis that identifies potential efficiency improvements in your current supply chain while ensuring seamless integration with your manufacturing requirements.