Industrial Scale Production of 4,4'-Dimethoxytriphenylchloromethane via Optimized Friedel-Crafts Alkylation
Industrial Scale Production of 4,4'-Dimethoxytriphenylchloromethane via Optimized Friedel-Crafts Alkylation
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more cost-effective, and higher-purity synthetic routes. A pivotal development in this sector is documented in patent CN107056590B, which discloses a robust industrial method for preparing and purifying 4,4'-dimethoxytriphenylchloromethane (DMT-Cl). This compound serves as a critical hydroxyl protecting group in the synthesis of biological nucleosides and nucleotides, making its quality paramount for downstream oligonucleotide drug production. The patented technology shifts away from traditional, hazardous methodologies towards a streamlined Friedel-Crafts alkylation process that utilizes anisole and trichlorotoluene as primary feedstocks. By leveraging Lewis acid catalysis and a sophisticated multi-step purification protocol, this approach not only enhances safety profiles but also ensures that the final active pharmaceutical ingredient intermediates meet the rigorous purity specifications demanded by modern biopharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DMT-Cl has relied heavily on Grignard reactions involving p-methoxy magnesium bromide and 4-methoxybenzophenone, a pathway fraught with significant operational challenges and safety hazards. The use of Grignard reagents necessitates strictly anhydrous conditions, requiring expensive and highly flammable solvents such as diethyl ether or tetrahydrofuran, which pose substantial explosion risks during large-scale heating and handling. Furthermore, the sensitivity of these organometallic species to moisture complicates the reaction environment, often leading to inconsistent yields and difficult process control in an industrial setting. Previous attempts to optimize this synthesis have struggled with purification; while some methods report purities around 99.73%, they often rely on costly refined solvents like n-heptane or n-hexane without providing a clear, scalable path to the ultra-high purity (>99.9%) required for sensitive nucleotide chemistry. These legacy processes create bottlenecks in supply chains due to their inherent instability and the high cost of specialized reagents and solvents.
The Novel Approach
In stark contrast, the innovative methodology outlined in the patent data utilizes a direct Friedel-Crafts reaction between anisole and trichlorotoluene, catalyzed by aluminum trichloride, to construct the triphenylmethane backbone efficiently. This route circumvents the need for unstable Grignard reagents entirely, replacing them with stable, commodity chemicals that are readily available in the global market. The process integrates a subsequent hydrochloric acid hydrolysis and a controlled chlorination step using reagents like thionyl chloride or oxalyl chloride, which allows for precise manipulation of the reaction kinetics. Crucially, the novel approach incorporates a dual-solvent crystallization strategy using mixtures of petroleum ether and dichloromethane, which effectively removes impurities and isomeric by-products. This shift represents a fundamental improvement in process chemistry, transforming a hazardous, batch-sensitive operation into a continuous, safe, and environmentally friendly industrial procedure capable of delivering yields exceeding 80% with exceptional consistency.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts Alkylation
The core of this synthetic breakthrough lies in the electrophilic aromatic substitution mechanism facilitated by the Lewis acid catalyst, aluminum trichloride (AlCl3). In the initial stage, AlCl3 activates the trichlorotoluene, generating a highly reactive electrophilic species that attacks the electron-rich aromatic ring of anisole. The stoichiometry is carefully controlled, with a molar ratio of anisole to trichlorotoluene maintained between 2.0:1 and 2.6:1, ensuring that the reaction proceeds to the desired tri-substituted product while minimizing the formation of mono- or di-substituted intermediates. The reaction temperature is tightly regulated between 0°C and 30°C to manage the exothermic nature of the alkylation and prevent thermal degradation or polymerization side reactions. Following the alkylation, the system undergoes a hydrolysis step where dilute hydrochloric acid quenches the aluminum complexes, releasing the organic intermediate into the organic phase for extraction. This mechanistic precision is vital for maintaining the structural integrity of the molecule before the final chlorination step converts the hydroxyl precursor into the reactive chloride functionality.
Controlling the impurity profile in DMT-Cl synthesis is critical because even trace contaminants can interfere with the coupling efficiency in solid-phase oligonucleotide synthesis. The patent describes a rigorous purification mechanism where the crude product, initially possessing a purity of over 98.5%, is subjected to a specific recrystallization regime. By utilizing a mixed solvent system—typically a combination of a non-polar solvent like petroleum ether and a moderately polar solvent like dichloromethane—the process exploits differences in solubility to selectively precipitate the target DMT-Cl crystal lattice while keeping soluble impurities in the mother liquor. The ratio of solvents is optimized, with solvent C (dichloromethane) used at 1.5 to 4.5 times the mass volume and solvent B (petroleum ether) at 8 to 20 times, creating a thermodynamic environment that favors the exclusion of structurally similar by-products. This level of control ensures that individual impurities are suppressed to levels below 0.05%, resulting in a final product purity that consistently exceeds 99.9%, thereby guaranteeing reliability for high-value pharmaceutical applications.
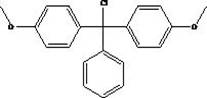
How to Synthesize 4,4'-Dimethoxytriphenylchloromethane Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and solvent management to replicate the high yields and purity reported in the patent examples. The process begins with the charging of trichlorotoluene and anisole into a reactor, followed by the controlled addition of the aluminum trichloride catalyst under inert conditions to initiate the Friedel-Crafts alkylation. Once the reaction mixture is formed, it undergoes a quenching and extraction sequence to isolate the organic intermediate, which is then concentrated to remove the extraction solvent. The subsequent chlorination step involves the addition of a chlorinating agent under reflux, followed by a cooling crystallization to obtain the crude solid. Finally, the crude material is recrystallized using the optimized binary solvent system to achieve the final specification. For laboratory and pilot plant teams looking to adopt this methodology, the detailed standardized operating procedures are essential for ensuring reproducibility and safety.
- Conduct Friedel-Crafts alkylation of anisole and trichlorotoluene using aluminum trichloride catalyst at 0-30°C.
- Perform hydrochloric acid hydrolysis and solvent extraction to isolate the intermediate organic phase.
- Execute chlorination with thionyl chloride followed by dual-solvent recrystallization to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented industrial method offers profound strategic advantages that extend beyond simple chemical yield improvements. By eliminating the reliance on Grignard reagents and volatile ethers, the process significantly reduces the complexity of raw material sourcing and storage, as anisole and trichlorotoluene are stable, high-volume commodity chemicals with robust global supply chains. This stability translates directly into enhanced supply chain reliability, reducing the risk of production stoppages caused by the scarcity or transportation hazards associated with specialized organometallic reagents. Furthermore, the ability to recycle solvents such as dichloromethane and petroleum ether throughout the extraction and crystallization phases creates a closed-loop system that minimizes waste disposal costs and environmental compliance burdens. These factors combine to create a manufacturing process that is not only safer and more environmentally sustainable but also inherently more cost-efficient, allowing for competitive pricing structures in the highly demanding nucleoside intermediate market.
- Cost Reduction in Manufacturing: The elimination of expensive, moisture-sensitive Grignard reagents and the replacement of high-cost refined solvents like n-heptane with standard industrial solvents leads to a drastic simplification of the bill of materials. The process design facilitates the recovery and reuse of solvents, which substantially lowers the variable costs associated with solvent consumption and waste treatment. Additionally, the avoidance of complex drying protocols required for anhydrous Grignard reactions reduces energy consumption and equipment downtime, contributing to overall operational expenditure savings without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for pharmaceutical supply chains, and this method leverages widely available starting materials that are produced at scale by multiple global suppliers. The robustness of the Friedel-Crafts chemistry against minor fluctuations in environmental moisture, compared to the extreme sensitivity of Grignard routes, ensures consistent batch-to-batch performance and reduces the likelihood of failed runs. This reliability allows for more accurate production planning and inventory management, ensuring that critical intermediates for nucleoside drug manufacturing are available exactly when needed, thereby supporting uninterrupted downstream synthesis operations.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from liter-scale laboratory experiments to 500-liter industrial reactors without loss of yield or purity, proving its viability for commercial tonnage production. The use of recyclable solvents and the avoidance of heavy metal catalysts or hazardous pyrophoric reagents align with modern green chemistry principles, simplifying the permitting process and reducing the environmental footprint of the manufacturing facility. This scalability ensures that the supply can grow in tandem with market demand for oligonucleotide therapeutics, providing a secure foundation for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DMT-Cl synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is crucial for R&D teams assessing process transfer and procurement teams evaluating vendor capabilities.
Q: Why is the Friedel-Crafts route preferred over Grignard reagents for DMT-Cl production?
A: The Friedel-Crafts route eliminates the safety risks associated with diethyl ether and the high moisture sensitivity of Grignard reagents, significantly lowering operational costs and improving industrial safety.
Q: What purity levels can be achieved with this purification method?
A: Through optimized solvent crystallization using petroleum ether and dichloromethane mixtures, the process consistently achieves finished product purity exceeding 99.9%, meeting stringent nucleoside synthesis standards.
Q: Is the solvent system recyclable in this industrial process?
A: Yes, the process design allows for the recovery and reuse of extraction and crystallization solvents, which drastically reduces waste generation and raw material consumption costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dimethoxytriphenylchloromethane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 4,4'-dimethoxytriphenylchloromethane meets the exacting standards required for nucleoside and nucleotide synthesis. Our infrastructure is designed to handle the specific solvent systems and reaction conditions outlined in advanced patents like CN107056590B, guaranteeing a supply of high-purity intermediates that support your drug development timelines.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates how switching to this optimized synthesis route can impact your overall manufacturing economics. We encourage potential partners to contact us directly to obtain specific COA data from our recent production batches and to discuss route feasibility assessments tailored to your project's unique volume and purity requirements. Let us be your partner in delivering high-quality chemical solutions that drive innovation in the life sciences industry.
