Revolutionizing DMT-Cl Production: Advanced Catalytic Routes for Commercial Scale-Up
The pharmaceutical industry constantly demands higher purity standards for critical intermediates, particularly those used in the synthesis of complex nucleoside analogues. Patent CN113816837A introduces a groundbreaking synthetic methodology for 4,4'-dimethoxytriphenylchloromethane (DMT-Cl), a vital hydroxy protective agent widely utilized in oligonucleotide and nucleotide chemistry. This innovative approach addresses long-standing inefficiencies in traditional manufacturing by employing a unique dual-catalyst system in the initial alkylation step and a solvent-free high-pressure chlorination strategy in the second step. By fundamentally rethinking the reaction engineering, this technology not only enhances the chemical yield but also drastically simplifies the downstream processing requirements. For global procurement leaders and R&D directors, this patent represents a significant leap forward in securing a stable, high-quality supply chain for essential fine chemicals. The method effectively mitigates the risks associated with hazardous reagents while delivering a product with exceptional purity profiles suitable for sensitive biological applications.
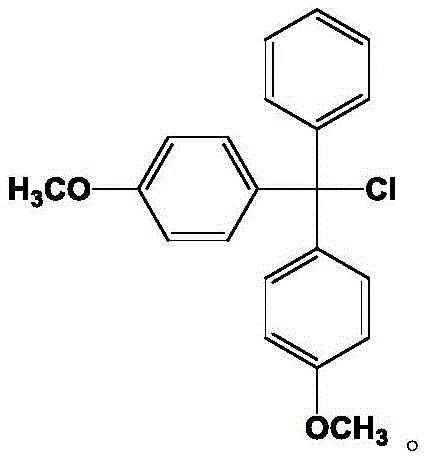
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4,4'-dimethoxytriphenylchloromethane has relied heavily on Grignard reactions involving p-methoxy magnesium bromide and 4-methoxy benzophenone. This traditional pathway is fraught with significant operational hazards and economic inefficiencies that hinder large-scale production. The requirement for anhydrous conditions is extremely stringent, as even trace moisture can quench the Grignard reagent, leading to catastrophic yield losses and inconsistent batch quality. Furthermore, the solvents typically employed, such as diethyl ether or tetrahydrofuran, present severe safety challenges; diethyl ether carries a high risk of explosion due to peroxide formation and low flash points, while tetrahydrofuran incurs substantial procurement costs. The subsequent chlorination steps often involve aggressive reagents like thionyl chloride or acetyl chloride, which generate corrosive byproducts and complicate waste management. These factors collectively result in a process that is difficult to scale, expensive to maintain, and prone to generating impurities that are challenging to remove to the levels required for pharmaceutical grade materials.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated Friedel-Crafts alkylation strategy that bypasses the need for unstable organometallic reagents entirely. The process initiates with the reaction of 4,4'-dimethoxydiphenylmethanol directly with benzene, cleverly employing benzene as both the reactant and the reaction medium. This dual function significantly reduces the volume of auxiliary solvents required, thereby lowering the environmental footprint and operational complexity. The introduction of a specific catalytic system comprising trifluoroacetic acid and a cuprous salt facilitates the dehydration and alkylation under mild thermal conditions, achieving conversion rates that far exceed traditional acid-catalyzed methods. The second stage involves a high-pressure chlorination using carbon tetrachloride, which acts simultaneously as the chlorinating agent and the solvent. This elegant design eliminates the need for additional catalysts in the final step and ensures a clean reaction profile with minimal side products, resulting in a final product purity that consistently exceeds 99.9 percent.
Mechanistic Insights into Cu-Catalyzed Friedel-Crafts Alkylation
The core innovation of this synthesis lies in the synergistic catalytic effect observed in the first step, where trifluoroacetic acid works in concert with cuprous salts such as CuI or CuBr. Mechanistically, the trifluoroacetic acid protonates the hydroxyl group of the diphenylmethanol substrate, facilitating the departure of water and generating a stabilized carbocation intermediate. The presence of the cuprous salt is believed to coordinate with the aromatic system or the leaving group, lowering the activation energy for the electrophilic aromatic substitution with benzene. This cooperative catalysis prevents the formation of polymeric byproducts and ensures high regioselectivity, which is critical for maintaining the structural integrity of the triphenylmethane backbone. The reaction proceeds efficiently at temperatures between 50°C and 60°C, avoiding the thermal degradation often seen in harsher acidic conditions. This controlled environment allows for the precise formation of the 4,4'-dimethoxytriphenylmethane intermediate with yields reaching up to 98 percent, providing a robust foundation for the subsequent chlorination step.
Following the formation of the triphenylmethane intermediate, the process transitions to a high-pressure chlorination mechanism that is equally ingenious in its simplicity and effectiveness. By heating the intermediate with carbon tetrachloride in a sealed high-pressure reactor, the system generates reactive chlorine species in situ without the need for external radical initiators or Lewis acid catalysts. The elevated pressure, typically maintained between 3 MPa and 7 MPa, forces the equilibrium towards the formation of the chlorinated product while suppressing the volatilization of the solvent. This high-energy environment promotes a clean substitution of the benzylic hydrogen with a chlorine atom, driven by the thermodynamic stability of the resulting trityl chloride bond. The absence of metal catalysts in this stage means there is no risk of heavy metal contamination in the final API intermediate, a crucial factor for regulatory compliance. The result is a highly pure crystalline product that requires only simple recrystallization to meet the most stringent pharmaceutical specifications.
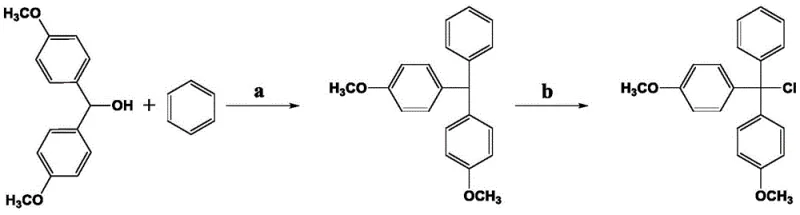
How to Synthesize 4,4'-Dimethoxytriphenylchloromethane Efficiently
The synthesis protocol outlined in the patent offers a streamlined pathway that is readily adaptable for pilot and commercial scale operations. The process begins by dispersing the starting alcohol and cuprous salt powder in benzene, followed by the controlled addition of trifluoroacetic acid under stirring. Once the reaction reaches completion, indicated by the disappearance of the starting material, the mixture is cooled and filtered to remove the catalyst residues. The filtrate is then subjected to a standard aqueous workup involving extraction and washing to isolate the intermediate triphenylmethane. For the final transformation, the intermediate is charged into a high-pressure vessel with carbon tetrachloride and heated to approximately 220°C. Detailed standardized operating procedures regarding exact stoichiometric ratios, pressure ramping rates, and specific recrystallization solvents are critical for reproducibility.
- React 4,4'-dimethoxydiphenylmethanol with benzene using trifluoroacetic acid and cuprous salt catalyst at 50-60°C.
- Isolate the intermediate 4,4'-dimethoxytriphenylmethane via filtration and recrystallization.
- Chlorinate the intermediate in carbon tetrachloride within a high-pressure reactor at 210-250°C to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond mere chemical efficiency. The elimination of Grignard reagents removes a major bottleneck related to raw material stability and storage, as organometallics require specialized handling and have limited shelf lives. By shifting to stable, commodity-grade chemicals like benzene and carbon tetrachloride, the supply chain becomes significantly more resilient against market fluctuations and logistical disruptions. Furthermore, the simplification of the workup procedures—specifically the ability to use one reactant as a solvent—drastically reduces the volume of waste solvent that needs to be treated or disposed of. This reduction in waste generation translates directly into lower operational expenditures related to environmental compliance and waste management fees. The high purity achieved directly from the reactor minimizes the need for resource-intensive purification steps like column chromatography, further driving down the cost of goods sold.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dual role of benzene and carbon tetrachloride as both reagents and solvents. This design feature inherently lowers the total mass of materials required per kilogram of product, reducing raw material procurement costs substantially. Additionally, the high conversion rates mean that less starting material is wasted, maximizing the atom economy of the process. The removal of expensive and hazardous reagents like thionyl chloride also contributes to a safer and more cost-effective operating environment, reducing the need for specialized corrosion-resistant equipment.
- Enhanced Supply Chain Reliability: Sourcing stable precursors is a critical concern for maintaining continuous production schedules. Unlike moisture-sensitive Grignard reagents which can degrade during transport or storage, the raw materials for this process are robust and widely available from multiple global suppliers. This diversity in sourcing options mitigates the risk of single-supplier dependency and ensures that production timelines are not compromised by raw material shortages. The simplified process flow also reduces the likelihood of batch failures due to operator error or environmental factors, leading to more predictable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The transition from batch processes involving hazardous ethers to a contained high-pressure system significantly improves the safety profile of the manufacturing facility. High-pressure reactors are standard equipment in modern chemical plants, making the scale-up from laboratory to multi-ton production straightforward and low-risk. Moreover, the reduced generation of acidic waste gases and the elimination of heavy metal catalysts in the final step simplify the effluent treatment process. This alignment with green chemistry principles facilitates easier permitting and regulatory approval, ensuring long-term operational sustainability in increasingly regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is the new catalytic method superior to traditional Grignard routes?
A: The new method eliminates the safety hazards associated with Grignard reagents, such as explosion risks with diethyl ether and extreme moisture sensitivity, while offering higher yields and easier purification.
Q: What represents the primary cost driver in this synthesis?
A: By utilizing benzene as both a reactant and solvent in the first step, the process significantly reduces raw material consumption and waste disposal costs compared to traditional methods requiring excess solvents.
Q: How does this process ensure high purity for nucleoside synthesis?
A: The specific combination of trifluoroacetic acid and cuprous salts minimizes side reactions, and the subsequent high-pressure chlorination step achieves over 99.9% purity without complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dimethoxytriphenylchloromethane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 4,4'-dimethoxytriphenylchloromethane that meets stringent purity specifications, leveraging our state-of-the-art rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement the advanced catalytic technologies described in recent patents allows us to offer a product that is not only chemically superior but also economically competitive.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to our manufactured grade. We encourage you to contact us directly to obtain specific COA data and comprehensive route feasibility assessments tailored to your project needs. Let us collaborate to secure a reliable, high-quality supply of this critical nucleoside protecting group for your future developments.
