Advanced Aqueous Acylation for High-Purity Cefepime Dihydrochloride Hydrate Production
Introduction to Patent CN1042536C and Technological Breakthroughs
The pharmaceutical landscape for broad-spectrum antibiotics has long been challenged by the complexities of stereochemical control and process efficiency, particularly in the synthesis of fourth-generation cephalosporins like cefepime. Patent CN1042536C introduces a transformative aqueous acylation process that addresses these critical bottlenecks by utilizing a stable crystalline cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride. This innovation eliminates the necessity for amino and carboxyl protecting groups, which have traditionally added significant cost and complexity to manufacturing workflows. By focusing on the direct N-acylation of the cephalosporin nucleus in an aqueous environment, the technology ensures the final product is substantially free of undesirable trans-isomers and delta-2 isomers, thereby enhancing both therapeutic efficacy and regulatory compliance for global supply chains.
For procurement managers and R&D directors seeking a reliable pharmaceutical intermediates supplier, this patent represents a paradigm shift towards greener and more economical synthesis routes. The core breakthrough lies in the ability to generate the active side chain without the toxic leaving groups or extensive purification steps associated with legacy methods. This not only streamlines the production timeline but also significantly reduces the environmental footprint of the manufacturing process. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a robust solution for the commercial scale-up of complex antibiotic intermediates, ensuring consistent quality and supply continuity for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefepime and related cephalosporins has relied heavily on multi-step protection and deprotection strategies that introduce substantial inefficiencies. Conventional processes often necessitate the use of protecting groups for the 2-amino substituent on the thiazole ring and activation of the carboxylic acid moiety using reagents that can be toxic or difficult to remove, such as 2-mercaptobenzothiazole. Furthermore, prior art methods frequently result in mixtures of geometric isomers, requiring laborious chromatographic purification to separate the desired cis-(Z) isomer from the inactive or less active trans-(E) isomer. These purification steps are not only costly in terms of solvent and resin consumption but also limit the overall throughput of the manufacturing facility, creating bottlenecks that affect cost reduction in pharmaceutical intermediates manufacturing.
Additionally, the instability of certain intermediate forms, such as the zwitterionic cefepime, poses significant challenges for storage and handling, often leading to degradation before the final salt formation. The reliance on organic solvents for acylation and the generation of hazardous waste streams further complicate the regulatory approval process and increase operational expenditures. For supply chain heads, these factors translate into longer lead times and higher vulnerability to raw material fluctuations. The inability to directly crystallize the stable dihydrochloride hydrate from the reaction mixture without intermediate isolation steps adds another layer of complexity, making the conventional route less attractive for large-scale industrial applications where efficiency and safety are paramount.
The Novel Approach
In stark contrast, the novel approach detailed in CN1042536C leverages a direct aqueous acylation strategy that bypasses the need for protecting groups entirely. By employing the stable crystalline cis-isomer of the thiazolyl acid chloride hydrochloride, the process achieves high stereochemical fidelity directly during the coupling reaction. This method utilizes a simple chloride ion as the leaving group, which is non-toxic and easily managed, replacing the complex organic leaving groups of the past. The reaction is conducted in a mixed aqueous-organic solvent system, which facilitates better heat transfer and control over the exothermic acylation process, ensuring that the delicate beta-lactam ring remains intact while the side chain is attached with precision.
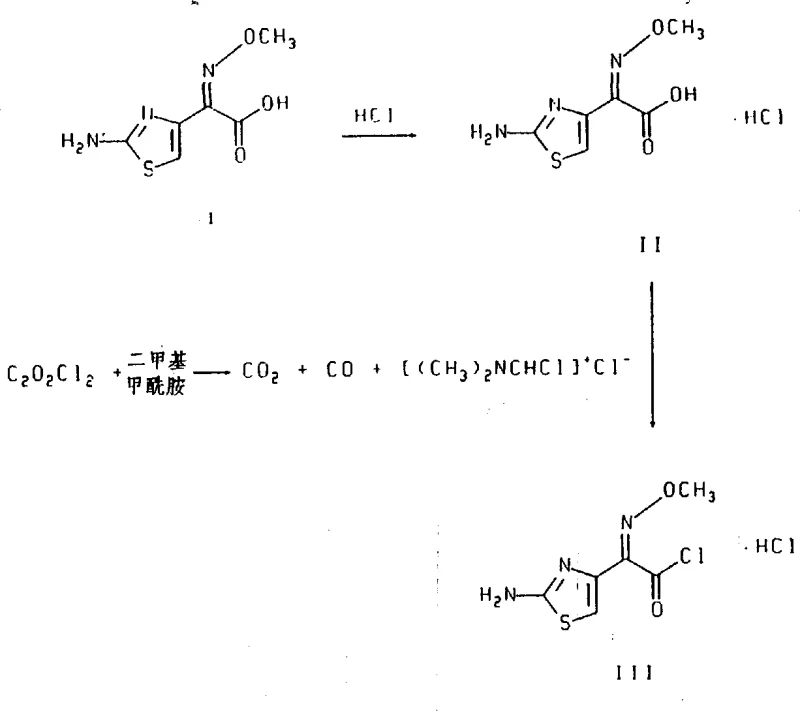
The superiority of this approach is visually represented in the reaction schemes, where the conversion from the acid to the active acid chloride is optimized to prevent isomerization. The process allows for the direct crystallization of the temperature-stable cefepime dihydrochloride hydrate from the reaction mixture, eliminating the need for isolating unstable zwitterionic forms or sulfate salts as intermediates. This streamlined workflow not only enhances the purity profile of the final API but also drastically simplifies the downstream processing requirements. For organizations aiming for high-purity pharmaceutical intermediates, this method offers a clear pathway to achieving stringent quality standards while minimizing operational overhead and waste generation.
Mechanistic Insights into Stereochemical Control and Aqueous Acylation
The mechanistic success of this process hinges on the precise control of the chlorination step used to activate the thiazolyl acetic acid derivative. The patent specifies the use of oxalyl chloride in combination with dimethylformamide (DMF) to generate the Vilsmeier-Haack reagent in situ, which acts as the chlorinating agent. Crucially, the molar ratio of DMF to oxalyl chloride must be carefully maintained, with a slight excess of DMF preferred to ensure complete conversion without promoting isomerization. The reaction temperature is kept strictly below -10°C, preferably between -15°C and -40°C, to kinetically favor the formation of the cis-isomer and suppress the thermodynamic drift towards the trans-configuration. This low-temperature regime is essential for preserving the geometric integrity of the methoxyimino double bond, which is critical for the biological activity of the final antibiotic.
Furthermore, the aqueous acylation step itself is engineered to maintain a specific pH range, typically between 5.0 and 7.5, using organic bases like triethylamine or N-methylmorpholine. This pH control is vital for neutralizing the hydrochloric acid generated during the coupling without causing hydrolysis of the acid chloride or degradation of the cephalosporin nucleus. The use of water-miscible organic solvents such as acetone or acetonitrile in conjunction with water creates a homogeneous reaction medium that supports rapid mixing and efficient heat dissipation. The result is a reaction profile that yields the desired product with minimal byproduct formation, specifically avoiding the formation of the delta-2 isomer which can arise from double bond migration in the cephalosporin ring under harsh conditions.
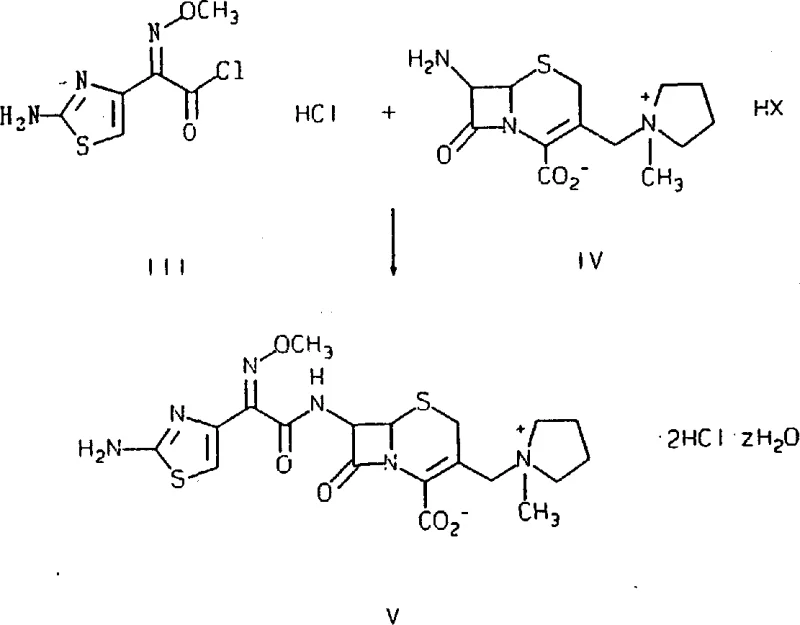
The structural distinction between the desired cis-isomer and the unwanted trans-isomer is profound, affecting both the physical properties and the biological potency of the molecule. As illustrated in the comparative structural analysis, the spatial arrangement of the methoxyimino group relative to the thiazole ring dictates the molecule's ability to bind to penicillin-binding proteins. The patent provides detailed NMR data confirming that the process yields a product with characteristic chemical shifts distinct from the trans-isomer, serving as a robust quality control metric. This level of mechanistic understanding allows manufacturers to implement rigorous in-process controls, ensuring that every batch meets the strict specifications required for clinical use. For R&D teams, this transparency in reaction dynamics facilitates easier technology transfer and scale-up from pilot plant to commercial production facilities.
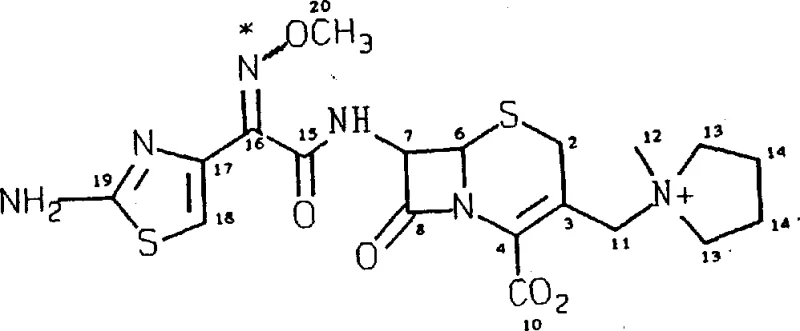
How to Synthesize Cefepime Dihydrochloride Hydrate Efficiently
The synthesis of cefepime dihydrochloride hydrate via this patented route involves a sequence of highly controlled chemical transformations designed to maximize yield and purity while minimizing waste. The process begins with the preparation of the key intermediate, the cis-2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride, which serves as the acylating agent. This intermediate is then reacted with the cephalosporin nucleus, 7-amino-3-[(1-methyl-1-pyrrolidinyl)methyl]ceph-3-ene-4-carboxylate, in a carefully buffered aqueous-organic solvent system. The detailed standardized synthesis steps for implementing this efficient route are outlined in the guide below, providing a clear roadmap for technical teams to follow.
- Preparation of the stable crystalline cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride using oxalyl chloride and DMF at low temperatures.
- N-acylation of 7-amino-3-[(1-methyl-1-pyrrolidinyl)methyl]ceph-3-ene-4-carboxylate in a mixed aqueous organic solvent at controlled pH.
- Crystallization of the final cefepime dihydrochloride hydrate by acidification and solvent adjustment to ensure high purity and stability.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aqueous acylation technology offers compelling advantages that directly impact the bottom line and supply chain resilience. By eliminating the need for protecting groups and the associated deprotection steps, the process significantly reduces the number of unit operations required, leading to lower labor costs and reduced equipment occupancy time. The removal of chromatographic purification steps further contributes to cost reduction in pharmaceutical intermediates manufacturing by saving on expensive resins and large volumes of organic solvents. Additionally, the use of non-toxic reagents and the generation of simpler waste streams align with increasingly stringent environmental regulations, reducing the cost of waste disposal and mitigating regulatory risk for the manufacturing site.
Supply chain reliability is enhanced by the stability of the key intermediate salt, which can be produced, isolated, and stored for extended periods without degradation. This decouples the production of the side chain from the final coupling step, allowing for better inventory management and flexibility in scheduling. The ability to produce the final API in a stable crystalline hydrate form directly from the reaction mixture ensures consistent product quality and simplifies packaging and distribution logistics. For supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates, this process offers a faster time-to-market for new batches and a more robust response to fluctuating demand patterns without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of protecting group chemistry and chromatographic purification drastically lowers raw material consumption and processing time. By using simple chloride leaving groups instead of complex toxic alternatives, the process reduces the cost of reagents and the expense associated with hazardous waste treatment. The streamlined workflow means fewer reactor turns and less energy consumption per kilogram of product, resulting in substantial operational savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The stability of the crystalline intermediate allows for strategic stockpiling, ensuring that production of the final API is not held up by upstream synthesis delays. The robustness of the aqueous acylation process against minor variations in reaction conditions leads to higher batch success rates and consistent output. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting contractual obligations without interruption.
- Scalability and Environmental Compliance: The use of aqueous solvent systems improves heat transfer and safety profiles, making the process inherently safer and easier to scale from pilot to commercial volumes. The reduction in organic solvent usage and the absence of heavy metal catalysts or toxic leaving groups simplify effluent treatment and ensure compliance with green chemistry principles. This environmental advantage future-proofs the manufacturing asset against tightening global regulations on pharmaceutical emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN1042536C, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical procurement teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does this process control stereochemistry compared to conventional methods?
A: The process utilizes a specific Vilsmeier-type chlorination with controlled DMF ratios and low temperatures (-10°C to -40°C) to maintain the Z-configuration, avoiding the formation of unwanted trans-isomers common in prior art.
Q: What are the stability advantages of the intermediate salt?
A: The cis-isomer acid chloride hydrochloride is a stable crystalline salt that can be isolated and stored, unlike unstable zwitterionic forms, allowing for flexible production scheduling and reduced waste.
Q: Does this method require chromatographic purification?
A: No, the aqueous acylation method yields the product substantially free of trans and delta-2 isomers directly from the reaction mixture, eliminating the need for expensive and time-consuming column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our expertise in process development allows us to leverage innovations like the aqueous acylation method described in CN1042536C to deliver high-quality cefepime intermediates with exceptional purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials regardless of volume requirements. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the highest international standards for antibiotic intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this protected-group-free methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your production goals, ensuring a seamless transition to a more efficient and sustainable manufacturing process.
