Advanced Somaglutide Synthesis: Overcoming Racemization via Side-Chain First Coupling Strategy
Introduction to Advanced Somaglutide Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing processes for complex peptide therapeutics, particularly for long-acting GLP-1 analogues like Somaglutide. A recent technical disclosure, identified by patent number CN112250755A, introduces a transformative preparation method that addresses critical bottlenecks in solid-phase peptide synthesis (SPPS). This innovation specifically targets the structural complexity of Somaglutide, which features a modified Lysine26 side chain connected to a fatty diacid moiety via a PEG linker. The traditional approach of assembling the main peptide backbone before attaching this bulky side chain has historically resulted in significant challenges, including low yields and high levels of stereochemical impurities. 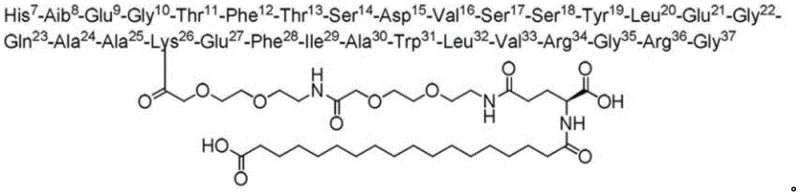 The new methodology reverses this sequence by coupling the side chain components onto the Lysine residue earlier in the synthesis cycle. This strategic adjustment effectively mitigates the risk of racemization, particularly at the Histidine7 position, which is highly susceptible to epimerization under acidic deprotection conditions. For procurement and supply chain leaders, understanding this shift is vital, as it represents a move towards more reliable somaglutide supplier capabilities, ensuring consistent quality and reduced production variability in the manufacturing of high-purity peptide APIs.
The new methodology reverses this sequence by coupling the side chain components onto the Lysine residue earlier in the synthesis cycle. This strategic adjustment effectively mitigates the risk of racemization, particularly at the Histidine7 position, which is highly susceptible to epimerization under acidic deprotection conditions. For procurement and supply chain leaders, understanding this shift is vital, as it represents a move towards more reliable somaglutide supplier capabilities, ensuring consistent quality and reduced production variability in the manufacturing of high-purity peptide APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Somaglutide followed a linear progression where the entire amino acid backbone was assembled first, followed by the attachment of the complex side chain to the Lysine26 residue. This conventional route, illustrated in comparative studies within the patent data, suffers from inherent chemical inefficiencies. As the peptide chain grows longer and more sterically hindered, the coupling efficiency for the bulky side chain decreases dramatically. Furthermore, the deprotection steps required to expose the Lysine epsilon-amino group for side-chain attachment often involve repeated exposure to acidic conditions.  These acidic cycles are detrimental to the chiral integrity of adjacent residues, most notably Histidine. Data from comparative examples indicates that this traditional method can result in His racemic impurity levels as high as 20.67%, necessitating extensive and costly downstream purification to meet pharmacopeial standards. For a reliable agrochemical intermediate supplier or pharma partner, such high impurity loads translate directly into increased solvent consumption, lower overall recovery, and extended production timelines, creating significant friction in the supply chain.
These acidic cycles are detrimental to the chiral integrity of adjacent residues, most notably Histidine. Data from comparative examples indicates that this traditional method can result in His racemic impurity levels as high as 20.67%, necessitating extensive and costly downstream purification to meet pharmacopeial standards. For a reliable agrochemical intermediate supplier or pharma partner, such high impurity loads translate directly into increased solvent consumption, lower overall recovery, and extended production timelines, creating significant friction in the supply chain.
The Novel Approach
The novel approach detailed in the patent fundamentally restructures the synthesis timeline to optimize chemical outcomes. By coupling the side chain components—specifically Fmoc-AEEA-OH, Fmoc-Glu-OtBu, and the octadecanedioic acid derivative—immediately after the 26th amino acid is attached, the process avoids the steric congestion of a fully formed 37-mer peptide. This "side-chain first" strategy ensures that the bulky fatty acid modification is installed while the growing chain is still relatively short and accessible. Consequently, the subsequent coupling of the remaining N-terminal amino acids (from position 25 down to 7) proceeds with higher efficiency and fidelity. This method effectively isolates the sensitive Histidine residue from the harsh conditions associated with side-chain deprotection, thereby preserving its optical purity. For stakeholders focused on cost reduction in peptide manufacturing, this approach offers a pathway to significantly simplified purification protocols and enhanced batch consistency.
Mechanistic Insights into Solid-Phase Peptide Synthesis Optimization
The core mechanistic advantage of this preparation method lies in the precise management of protecting group orthogonality and reaction kinetics. The process utilizes a resin, such as CTC or Wang resin, with a controlled substitution degree of 0.45 to 0.75 mmol/g to prevent aggregation. The synthesis initiates with the loading of Fmoc-Gly-OH, followed by the sequential addition of amino acids up to Lysine26, which is protected with an orthogonal group like Mtt or Alloc. This orthogonal protection allows for the selective removal of the side-chain protecting group without disturbing the main chain Fmoc groups. The coupling of the side chain employs potent reagents like HATU/DIEA or DIC/HOBt to drive the reaction to completion despite the increasing steric bulk. By installing the AEEA-PEG-Glu-fatty acid moiety at this stage, the chemical environment for the subsequent elongation steps is stabilized.
Impurity control is rigorously addressed through this reordered sequence. In conventional synthesis, the final deprotection and cleavage steps often induce racemization at the N-terminal Histidine due to prolonged exposure to trifluoroacetic acid (TFA) and scavengers. By completing the side-chain coupling earlier, the number of acidic treatment cycles the Histidine residue undergoes is minimized. Experimental data from the patent demonstrates that this method reduces the His racemic impurity from over 20% in comparative examples to approximately 5.88% in the crude product, ultimately yielding a final purity of 99.8% after HPLC purification. This level of control is critical for producing high-purity OLED material grade peptides or pharmaceutical intermediates where trace impurities can compromise biological activity and safety profiles.
How to Synthesize Somaglutide Efficiently
The synthesis of Somaglutide via this optimized route requires precise adherence to the stepwise coupling and deprotection protocols outlined in the technical disclosure. The process begins with resin swelling and loading, followed by iterative cycles of Fmoc deprotection and amino acid coupling. A critical juncture occurs at the Lysine26 position, where the orthogonal protecting group is selectively removed to facilitate the attachment of the lipidated side chain. Once the side chain is secured, the synthesis resumes towards the N-terminus. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and reaction times, are essential for replicating the high yields and purity reported in the patent examples.
- Load Fmoc-Gly-OH onto initial resin (CTC or Wang) and cap unreacted sites to form Fmoc-Gly-resin.
- Sequentially couple amino acids from position 36 down to 26, stopping at the Lysine residue with orthogonal protecting groups.
- Couple the side chain components (AEEA, Glu, and octadecanedioic acid derivative) onto the Lys26 residue while still on the resin.
- Remove the Lys main chain protecting group and continue coupling the remaining N-terminal amino acids (25 to 7).
- Cleave the full peptide from the resin using a TFA-based cocktail and purify via preparative HPLC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the substantial cost savings derived from improved process efficiency. By significantly reducing the formation of difficult-to-remove stereoisomers, the manufacturing process eliminates the need for excessive recycling of fractions or the use of large volumes of specialized chromatography media. This streamlining of the purification phase directly correlates to reduced solvent waste and lower operational expenditures. Furthermore, the simplified operational flow reduces the risk of batch failure, ensuring a more predictable supply of critical peptide intermediates for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required to remove high levels of racemic impurities leads to a drastic simplification of the downstream processing workflow. By avoiding the accumulation of stereochemical errors, manufacturers can reduce the consumption of expensive HPLC columns and organic solvents, resulting in substantial cost savings per kilogram of active ingredient produced. This efficiency makes the commercial scale-up of complex peptide APIs more economically viable.
- Enhanced Supply Chain Reliability: The robustness of the "side-chain first" method enhances the reliability of the supply chain by minimizing batch-to-batch variability. Traditional methods prone to high impurity loads often result in inconsistent yields, causing delays in delivery schedules. This optimized protocol ensures a stable and continuous production output, allowing partners to maintain leaner inventory levels while securing a steady flow of high-quality materials for their drug development pipelines.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial production, utilizing standard SPPS equipment and commercially available reagents. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, facilitating easier regulatory approval for manufacturing sites. This scalability ensures that the transition from laboratory-scale development to multi-ton commercial production can be achieved with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Somaglutide preparation method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential manufacturing partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production facilities.
Q: Why does the side-chain first coupling method reduce racemization?
A: Conventional methods couple the bulky fatty acid side chain last, which requires harsh deprotection conditions near the sensitive Histidine residue, leading to racemization. By coupling the side chain earlier (at Lys26) before completing the N-terminal sequence, the Histidine residue is exposed to fewer acidic cycles, significantly preserving its chiral integrity.
Q: What type of resin is recommended for this synthesis?
A: The patent specifies the use of CTC resin (2-chlorotrityl chloride resin) or Wang resin with a substitution degree preferably between 0.45 to 0.75 mmol/g. These resins provide the necessary stability and loading capacity for the long peptide chain assembly.
Q: How is the final purity of Somaglutide ensured?
A: High purity is achieved through a combination of optimized coupling reagents (such as HATU/DIEA or DIC/HOBt) to minimize deletion sequences, and a final purification step using semi-preparative reverse-phase liquid chromatography (RP-HPLC) with a C18 column.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somaglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis strategies to meet the growing global demand for GLP-1 analogues. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel side-chain first coupling method are fully realized in a GMP environment. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Somaglutide adheres to the highest international standards for safety and efficacy.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains. By leveraging our technical expertise, you can achieve significant efficiencies in your peptide manufacturing processes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
