Advanced Combretastatin Intermediate Synthesis: Scalable Commercial Manufacturing Solutions
Advanced Combretastatin Intermediate Synthesis: Scalable Commercial Manufacturing Solutions
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value anticancer agents, and the technology disclosed in patent CN1291970C represents a significant leap forward in the manufacturing of combretastatin derivatives. This specific intellectual property details novel methods for preparing combretastatins, which are potent tubulin-binding agents, by optimizing the critical Wittig condensation and subsequent reduction steps. For R&D directors and procurement specialists evaluating supply chain resilience, the shift from traditional zinc-mediated reductions to iron-based protocols offers a compelling value proposition regarding yield enhancement and impurity control. The patent explicitly outlines pathways that not only improve the chemical efficiency of producing the stilbene backbone but also address the stringent purity requirements necessary for downstream API synthesis. By leveraging these improved methodologies, manufacturers can achieve a more consistent supply of high-purity pharmaceutical intermediates while mitigating the risks associated with hazardous reagents. This report analyzes the technical depth of CN1291970C to demonstrate its viability for commercial scale-up and its potential to reduce overall manufacturing costs through process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
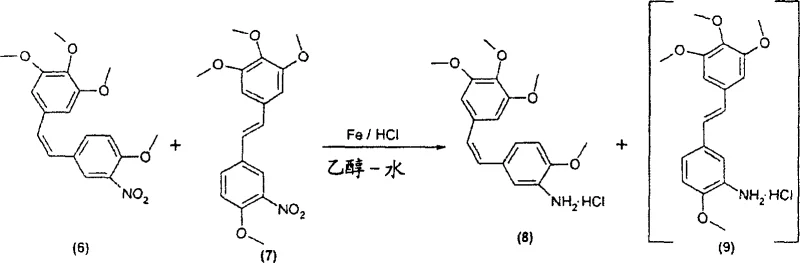
Historically, the synthesis of combretastatin analogues has relied heavily on reduction methods utilizing zinc powder in acetic acid, a process fraught with significant inefficiencies and safety concerns that hinder large-scale production. According to the background art cited in the patent, such as US 5,525,632, the reduction of nitro derivatives using zinc often results in incomplete reactions and the formation of substantial amounts of unwanted azo compounds due to the coupling of amino and nitroso intermediates. Furthermore, the yield of the desired pure Z-isomer in these conventional zinc-mediated processes is notoriously low, typically ranging between 21% and 33% based on the aldehyde starting material, which creates a massive bottleneck for commercial viability. The use of zinc also introduces challenges in waste management and metal removal, requiring extensive downstream purification to meet the stringent heavy metal specifications required for pharmaceutical ingredients. Additionally, alternative reduction methods using catalytic hydrogenation with palladium or platinum often lead to the undesirable saturation of the ethylene double bond or high isomerization to the inactive E-isomer, further compromising the overall process efficiency. These technical limitations collectively drive up the cost of goods and introduce significant supply chain volatility for manufacturers relying on outdated synthetic protocols.
The Novel Approach
In stark contrast to the deficiencies of prior art, the novel approach detailed in CN1291970C introduces a strategic substitution of zinc with iron powder for the reduction of nitro-stilbene derivatives, resulting in a dramatic improvement in reaction performance and product quality. This innovative method allows for the reduction of 2-methoxy-5-[2-(3,4,5-trimethoxyphenyl)vinyl]nitrobenzene to proceed with much higher conversion rates, achieving yields of approximately 60% relative to the aldehyde, which is nearly double the efficiency of the traditional zinc method. The use of iron in an ethanol-water solvent system with hydrochloric acid effectively minimizes the formation of azo-coupling byproducts, thereby simplifying the purification process and enhancing the overall purity profile of the intermediate. Moreover, the patent describes alternative pathways (V03 and V04) that utilize amino-starting materials directly, effectively bypassing the reduction step entirely and eliminating the generation of CMR (carcinogenic, mutagenic, or teratogenic) products associated with nitro-reduction. This dual strategy of optimizing reduction conditions and redesigning the synthetic route provides a robust framework for manufacturing that is both economically superior and environmentally more sustainable.
Mechanistic Insights into Iron-Catalyzed Reduction and Wittig Condensation
The core chemical innovation lies in the precise control of the reduction mechanism and the stereoselectivity of the Wittig condensation, which are critical for ensuring the biological activity of the final combretastatin product. The iron-mediated reduction operates through a single-electron transfer mechanism that is sufficiently mild to reduce the nitro group to an amine without affecting the sensitive olefinic double bond, a common failure point in catalytic hydrogenation methods. By carefully controlling the stoichiometry of the iron powder, preferably using more than 2 equivalents per mole of nitro derivative, the process ensures complete conversion of the starting material while preventing the accumulation of hydroxylamine intermediates that could lead to side reactions. The reaction conditions, typically involving heating to 77±2°C in an ethanol-water mixture, are optimized to maintain the solubility of the intermediates while facilitating the precipitation of iron oxides, which can be easily removed by filtration. This mechanistic clarity allows process chemists to scale the reaction with confidence, knowing that the critical quality attributes of the molecule, specifically the Z-configuration of the stilbene backbone, are preserved throughout the transformation. The ability to isolate the Z-amino isomer via crystallization of the hydrochloride salt further underscores the robustness of this chemical design.
Complementing the reduction step, the Wittig condensation mechanism is tuned to maximize the formation of the cis (Z) isomer, which is the pharmacologically active configuration for combretastatins. The patent details the use of specific bases such as sodium methoxide in solvents like toluene or THF at controlled low temperatures (0-10°C) to influence the stereochemical outcome of the olefination. The reaction between the phosphonium salt and the aldehyde proceeds through a betaine intermediate, and the choice of base and solvent system is crucial for minimizing the formation of the trans (E) isomer. In the novel pathways V03 and V04, the use of amino-substituted starting materials requires careful protection group strategy to prevent interference with the ylide formation, yet the patent demonstrates that high purity intermediates can be achieved through standard chromatographic and crystallization techniques. This deep understanding of the reaction kinetics and thermodynamics enables the design of a manufacturing process that consistently delivers high-purity intermediates, reducing the burden on downstream purification and ensuring a reliable supply of material for API synthesis.
How to Synthesize Combretastatin Intermediates Efficiently
The synthesis of these high-value pharmaceutical intermediates requires a disciplined approach to reaction engineering, leveraging the specific conditions outlined in the patent to ensure reproducibility and safety at scale. The process begins with the preparation of the key phosphonium salt or amino-aldehyde precursors, followed by the critical Wittig condensation step which establishes the carbon-carbon double bond with the desired stereochemistry. Subsequent steps involve the reduction of nitro groups using the improved iron protocol or the direct coupling of amino-intermediates with protected serine derivatives to form the final amide bond. Each stage of the synthesis is designed to minimize impurity carryover and maximize yield, utilizing standard unit operations such as filtration, crystallization, and solvent exchange that are well-suited for commercial manufacturing environments. The detailed standardized synthesis steps see the guide below.
- Prepare the phosphonium salt or amino-aldehyde starting materials using standard halogenation and phosphination techniques.
- Perform Wittig condensation using sodium methoxide in toluene or THF to generate the stilbene backbone with high Z-isomer selectivity.
- Execute the critical reduction step using iron powder in acidic ethanol/water media to convert nitro groups to amines without saturating the double bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the synthetic methods described in CN1291970C translates into tangible strategic advantages that directly impact the bottom line and operational reliability. The primary benefit stems from the significant increase in chemical yield achieved by the iron-mediated reduction, which effectively reduces the amount of raw materials required to produce a unit of output, thereby lowering the variable cost of manufacturing. By eliminating the need for expensive and hazardous catalysts like palladium or platinum, and replacing zinc with abundant and cost-effective iron, the process drastically simplifies the supply chain for reagents and reduces exposure to price volatility in the precious metals market. Furthermore, the reduction in CMR substances and hazardous waste generation streamlines regulatory compliance and waste disposal costs, which are often hidden but substantial expenses in fine chemical manufacturing. The robustness of the crystallization steps described in the patent ensures that the final product meets stringent purity specifications without requiring complex and costly chromatographic separations on a large scale. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of meeting the demanding requirements of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The substitution of zinc with iron powder and the elimination of precious metal catalysts fundamentally alter the cost structure of the synthesis, leading to substantial savings in raw material expenditures. The higher yield of the desired Z-isomer means that less starting material is wasted, and the overall throughput of the manufacturing plant is increased without the need for additional capital investment in reactor capacity. Additionally, the simplified workup procedures, which rely on filtration and crystallization rather than extensive chromatography, reduce the consumption of solvents and silica gel, further driving down operational costs. This qualitative improvement in process efficiency allows for a more competitive pricing strategy while maintaining healthy profit margins, making the technology highly attractive for long-term commercial partnerships.
- Enhanced Supply Chain Reliability: By utilizing readily available reagents such as iron powder and common solvents like ethanol and toluene, the manufacturing process becomes less susceptible to supply disruptions that often plague specialized chemical supply chains. The avoidance of CMR reagents reduces the regulatory burden and the risk of production stoppages due to compliance issues, ensuring a continuous and reliable flow of intermediates to downstream API manufacturers. The scalability of the process, demonstrated by the use of standard reaction conditions and equipment, means that production volumes can be ramped up quickly to meet surges in demand without compromising quality. This reliability is crucial for pharmaceutical companies that require guaranteed supply continuity to support their clinical trials and commercial product launches.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily controlled in large-scale reactors and workup procedures that are compatible with existing industrial infrastructure. The reduction in hazardous waste, particularly the elimination of heavy metal sludge associated with zinc reduction, aligns with modern environmental, social, and governance (ESG) goals and simplifies the permitting process for manufacturing facilities. The ability to recycle solvents and the use of aqueous workups further enhance the environmental profile of the synthesis, making it a sustainable choice for forward-thinking chemical enterprises. This alignment with green chemistry principles not only reduces environmental impact but also enhances the brand reputation of the supplier in a market that increasingly values sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology, providing clarity on its practical application and benefits for potential partners. These answers are derived directly from the technical specifications and experimental data presented in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this synthesis route into existing manufacturing portfolios.
Q: How does the iron-mediated reduction improve yield compared to traditional zinc methods?
A: The patent data indicates that replacing zinc with iron powder in the reduction of nitro-stilbene derivatives significantly increases yield from approximately 21-33% to around 60%, while minimizing the formation of azo-coupling byproducts.
Q: Does this synthesis route avoid carcinogenic, mutagenic, or teratogenic (CMR) reagents?
A: Yes, the novel pathways V03 and V04 utilize amino-starting materials directly, eliminating the need for intermediate reduction steps that often generate CMR substances, thereby enhancing industrial safety profiles.
Q: Is the Z-isomer selectivity maintained during the commercial scale-up of this process?
A: The process utilizes specific crystallization techniques and controlled Wittig conditions that favor the formation of the biologically active Z-isomer, with purification steps designed to remove the E-isomer effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Combretastatin Intermediate Supplier
The technological advancements detailed in patent CN1291970C underscore the immense potential of this synthetic route for the commercial production of high-purity combretastatin intermediates, and NINGBO INNO PHARMCHEM is uniquely positioned to bring this innovation to the global market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing scenarios. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the Z-isomer content and impurity profiles to meet the exacting standards of international pharmaceutical clients. We understand that the transition from lab-scale innovation to commercial reality requires not just chemical expertise but also a deep commitment to quality assurance and supply chain integrity, which are the cornerstones of our operational philosophy.
We invite you to engage with our technical procurement team to discuss how this improved synthesis method can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic benefits of switching to this iron-mediated reduction process for your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your projects. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of combretastatin intermediates that will support your drug development and commercialization goals.
