Optimizing Thiazolidinedione Production: A Strategic Breakthrough for Pharmaceutical Manufacturing
Optimizing Thiazolidinedione Production: A Strategic Breakthrough for Pharmaceutical Manufacturing
The pharmaceutical landscape for Type II diabetes treatment relies heavily on the efficient production of high-purity thiazolidinedione derivatives. Patent CN1231482C introduces a transformative methodology for synthesizing 5-{4-[2-(5-methyl-2-phenyl-oxazol-4-yl)-ethoxy]-benzo[b]thiophen-7-ylmethyl}-2,4-thiazolidinedione and its salts. This innovation addresses critical bottlenecks found in earlier methodologies, such as those described in WO94/27995, which were plagued by excessive reaction steps and suboptimal yields. By streamlining the synthetic route through a novel bromomethylation strategy followed by direct coupling, this technology offers a robust pathway for manufacturing active pharmaceutical ingredients. For R&D directors and supply chain leaders, understanding this process is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex thiazolidinedione derivatives has been hindered by multi-step sequences that erode overall process efficiency. Prior art methods often necessitate rigorous purification between every single transformation, leading to significant material loss and increased solvent consumption. These conventional routes frequently suffer from low yields that render them economically unviable for large-scale commercial production. Furthermore, the reliance on unstable intermediates or harsh reaction conditions in older protocols introduces variability that complicates quality control. For procurement managers, these inefficiencies translate into higher costs of goods sold and potential supply chain disruptions. The inability to utilize crude intermediates forces manufacturers to invest heavily in downstream processing equipment and extended production timelines, creating a barrier to entry for cost-effective manufacturing.
The Novel Approach
The methodology disclosed in CN1231482C revolutionizes this landscape by introducing a concise two-step core sequence starting from a formula II compound. The process initiates with a highly controlled bromomethylation or chloromethylation to generate a reactive formula III intermediate. Crucially, this intermediate does not require exhaustive purification before proceeding to the next stage. The subsequent reaction with a formula IV compound, specifically a thiazolidinedione salt, proceeds with outstanding yield under moderate conditions. This reduction in unit operations not only accelerates the production timeline but also minimizes waste generation. By enabling the use of crude intermediates, the novel approach drastically simplifies the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. This strategic shift allows for a more agile response to market demands while maintaining high purity standards.
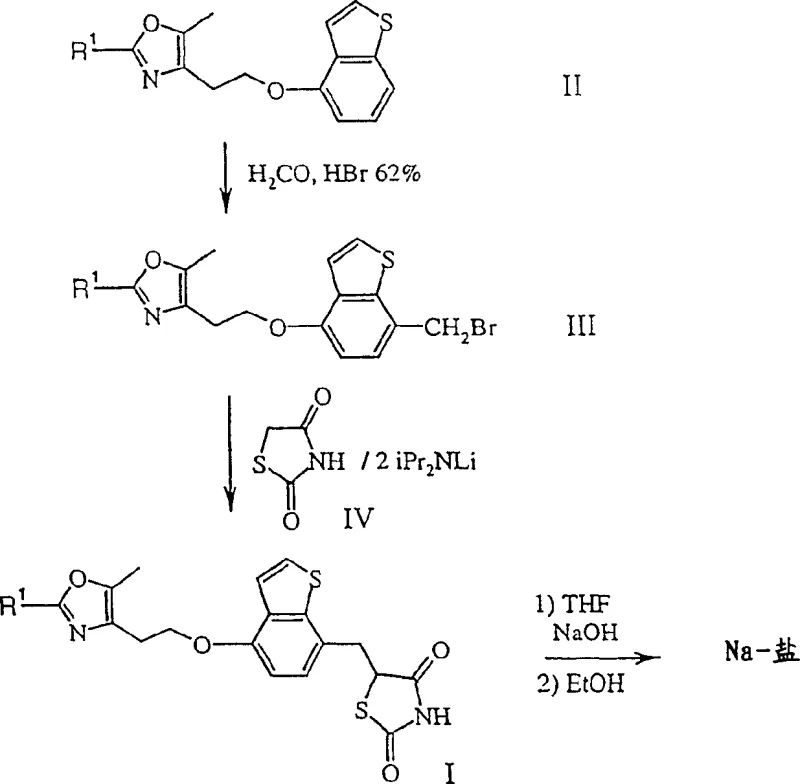
Mechanistic Insights into Bromomethylation and Nucleophilic Substitution
The core of this technological advancement lies in the precise control of the bromomethylation reaction. The patent specifies the use of formaldehyde sources, preferably trioxane, in the presence of hydrobromic acid concentrations between 45% and 62%. Conducting this reaction in solvents like dichloromethane at temperatures ranging from -10 to +10°C ensures the selective introduction of the bromomethyl group without degrading the sensitive oxazole or benzothiophene moieties. This selectivity is paramount for maintaining the integrity of the molecular scaffold. Following this, the generation of the nucleophile is achieved by forming the dilithium salt of 2,4-thiazolidinedione using lithium diisopropylamide (LDA) in THF. This strong base facilitates the deprotonation necessary for the subsequent nucleophilic attack on the benzylic bromide. The mechanistic precision here prevents side reactions such as elimination or over-alkylation, which are common pitfalls in similar alkylations.
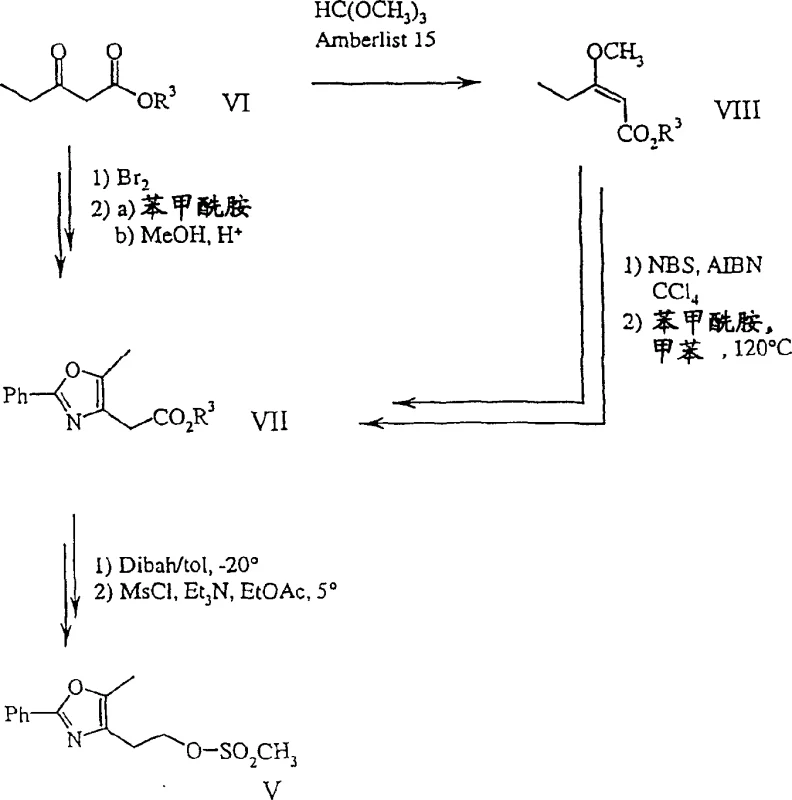
Beyond the final coupling, the patent also details robust pathways for synthesizing the necessary precursors, ensuring a fully integrated supply chain solution. For instance, the preparation of formula V compounds involves the bromination of formula VI compounds, preferably at the gamma-position, followed by condensation with benzamides. Alternative routes utilizing enol ethers (formula VIII) and N-bromosuccinimide (NBS) offer flexibility depending on raw material availability. The ability to switch between brominating a keto-ester directly or via an enol ether intermediate provides R&D teams with contingency plans to mitigate raw material shortages. This versatility in intermediate synthesis underscores the process's resilience. Understanding these mechanistic nuances allows technical teams to optimize reaction parameters further, ensuring consistent batch-to-batch reproducibility essential for regulatory compliance.
How to Synthesize 5-{4-[2-(5-Methyl-2-Phenyl-Oxazol-4-Yl)-Ethoxy]-Benzo[B]Thiophen-7-Ylmethyl}-2,4-Thiazolidinedione Efficiently
Implementing this synthesis requires strict adherence to the specified reaction conditions to maximize yield and purity. The process begins with the preparation of the bromomethylated intermediate, where temperature control is critical to prevent poly-bromination. Once the formula III compound is obtained, it is reacted with the pre-formed dilithium salt of the thiazolidinedione ring. The workup involves careful pH adjustment and crystallization to isolate the final product or its sodium salt. Detailed operational parameters, including stoichiometry and quenching procedures, are essential for successful scale-up. The following guide outlines the standardized synthesis steps derived from the patent examples to assist your technical team in process validation.
- Perform bromomethylation of the formula II compound using formaldehyde and hydrobromic acid in a solvent like dichloromethane at low temperatures.
- Isolate the resulting formula III intermediate, which can often be used crude in the subsequent step to save time and resources.
- React the formula III compound with the dilithium salt of 2,4-thiazolidinedione (generated using LDA) to form the final formula I product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits beyond mere chemical efficiency. The reduction in reaction steps directly correlates to a decrease in capital expenditure on processing equipment and a reduction in the physical footprint required for manufacturing. By eliminating the need for intermediate purification, the process significantly lowers solvent consumption and waste disposal costs, aligning with modern environmental compliance standards. This streamlined workflow enhances supply chain reliability by shortening the lead time for high-purity pharmaceutical intermediates. Furthermore, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in operating parameters, ensuring a steady flow of materials to downstream API manufacturers.
- Cost Reduction in Manufacturing: The ability to utilize crude intermediates without additional purification steps represents a major driver for cost optimization. This eliminates entire unit operations such as column chromatography or recrystallization of unstable species, thereby saving both time and expensive reagents. The high yields reported in the patent examples mean that less raw material is required to produce the same amount of final product, directly improving the margin structure. Additionally, the use of commercially available reagents like trioxane and standard solvents like dichloromethane ensures that input costs remain stable and predictable. This economic efficiency makes the process highly competitive in the global market for diabetes therapeutics.
- Enhanced Supply Chain Reliability: The synthetic route relies on widely available starting materials and avoids exotic catalysts that might be subject to supply constraints. The modular nature of the intermediate synthesis allows for parallel production streams, reducing the risk of bottlenecks. By simplifying the process flow, the likelihood of batch failures due to operational complexity is minimized, ensuring consistent delivery schedules. This reliability is crucial for maintaining inventory levels and meeting the Just-In-Time delivery expectations of major pharmaceutical clients. The process stability also facilitates easier technology transfer between manufacturing sites, further securing the supply network against regional disruptions.
- Scalability and Environmental Compliance: The reaction conditions, particularly the moderate temperatures and standard pressure requirements, are inherently scalable from pilot plant to commercial tonnage. The reduction in solvent usage and waste generation supports sustainability goals and reduces the burden on effluent treatment plants. The process avoids the use of heavy metal catalysts, simplifying the removal of impurities and ensuring the final product meets stringent residual metal specifications. This environmental compatibility not only reduces regulatory hurdles but also enhances the corporate social responsibility profile of the manufacturing operation. Scalability is further supported by the exothermic nature of the reactions being manageable with standard cooling systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation. They serve to clarify the operational feasibility and strategic value of adopting this method for your production portfolio. Understanding these details is key for making informed decisions about process adoption and vendor qualification.
Q: What are the key advantages of this new synthesis method over prior art?
A: The method described in CN1231482C significantly reduces the number of reaction steps compared to WO94/27995 and achieves outstanding yields, making it suitable for commercial scale production.
Q: Can the intermediates be purified before the next step?
A: While purification is possible, the patent highlights that crude intermediate products can largely be used in subsequent reaction steps without additional purification, enhancing process efficiency.
Q: What specific reagents are preferred for the bromomethylation step?
A: The process preferably utilizes trioxane as the source of formaldehyde and aqueous hydrobromic acid (45-62% concentration) in a solvent such as dichloromethane at temperatures between -10 and +10°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-{4-[2-(5-Methyl-2-Phenyl-Oxazol-4-Yl)-Ethoxy]-Benzo[B]Thiophen-7-Ylmethyl}-2,4-Thiazolidinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of diabetes medication production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of thiazolidinedione derivative meets the highest international standards. Our commitment to process excellence allows us to deliver high-purity pharmaceutical intermediates that facilitate smoother regulatory filings for our partners.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency. Let us be your partner in driving down costs and securing a reliable supply of essential pharmaceutical building blocks.
