Advanced Catalytic Reduction of Thiazolidinedione Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly for high-volume anti-diabetic agents like pioglitazone and rosiglitazone. Patent CN1491218A introduces a transformative methodology for preparing compounds derived from thiazolidinedione, oxazolidinedione, or hydantoin structures, specifically addressing the reduction of exocyclic double bonds found in formula (II) precursors. This innovation shifts the paradigm from hazardous traditional reduction methods to a safer, highly selective catalytic process utilizing formic acid as a hydrogen source. By leveraging transition metal catalysts, this technique achieves complete conversion with minimal byproduct formation, directly addressing the purity concerns that often plague the manufacturing of complex heterocyclic intermediates. The strategic implementation of this technology allows for significant operational flexibility, supporting both transfer hydrogenation and direct hydrogenation pathways depending on the specific infrastructure available at the production site.
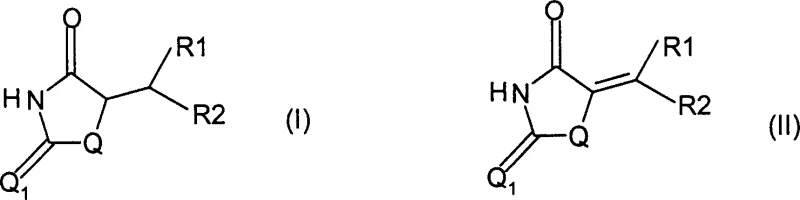
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of saturated thiazolidinedione derivatives from their unsaturated precursors has relied on chemically aggressive reagents that pose substantial risks in large-scale operations. Conventional techniques, such as those described in WO 9837073, frequently employ metal hydrides or magnesium in methanol, which are notorious for generating excessive impurity profiles, sometimes exceeding 10% in critical syntheses like that of pioglitazone. These legacy methods suffer from poor selectivity, often reducing other sensitive functional groups within the molecule, thereby complicating downstream purification and drastically lowering overall yield. Furthermore, the use of stoichiometric amounts of reducing agents generates significant quantities of inorganic waste, creating heavy environmental burdens and increasing the cost of waste disposal. The difficulty in isolating the target compound from the reaction mixture often necessitates complex chromatographic separations, which are economically unviable for multi-ton commercial production of pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN1491218A utilizes formic acid in the presence of a transition metal-based catalyst to effect the reduction, offering a cleaner and more atom-economical alternative. This approach capitalizes on the dual role of formic acid, which serves effectively as both the solvent and the hydrogen donor in transfer hydrogenation reactions, thereby eliminating the need for large volumes of auxiliary organic solvents. The reaction conditions are remarkably mild, operating effectively between 0-150°C, and the process demonstrates exceptional selectivity for the exocyclic carbon-carbon double bond while leaving the sensitive carbonyl groups of the dione ring intact. By avoiding harsh reducing agents, the formation of side products is minimized, leading to a crude product profile that is significantly easier to purify through simple crystallization or filtration. This technological leap not only enhances the chemical efficiency of the synthesis but also aligns with modern green chemistry principles by reducing solvent usage and hazardous waste generation.
Mechanistic Insights into Transition Metal-Catalyzed Transfer Hydrogenation
The core of this innovative process lies in the catalytic cycle facilitated by transition metals such as Iridium, Ruthenium, Rhodium, or Palladium, which activate the formic acid to deliver hydride equivalents to the substrate. In the transfer hydrogenation mechanism, the metal center coordinates with formic acid, promoting its decomposition into carbon dioxide and a metal-hydride species, which then inserts into the electron-deficient double bond of the thiazolidinedione precursor. This pathway avoids the handling of high-pressure hydrogen gas in certain embodiments, although the patent also supports direct hydrogenation using H2 pressures between 0.1-50 bar for accelerated kinetics. The choice of ligand environment around the metal center, whether using phosphine ligands in homogeneous systems like Ru(PPh3)3Cl2 or the surface properties of heterogeneous catalysts like Pd/C, critically influences the turnover frequency and stereoselectivity of the reduction. Understanding this mechanistic nuance allows process chemists to fine-tune the reaction parameters, such as the metal-to-substrate ratio which can range from 1/10000 to 5%, to optimize cost versus reaction speed.
Impurity control is inherently built into this catalytic system due to the specific electronic requirements of the reduction mechanism. Unlike non-selective chemical reducers that might attack the carbonyl oxygens at positions Q and Q1, the transition metal-hydride species generated from formic acid exhibits a strong preference for conjugated olefins. This chemoselectivity ensures that the integrity of the thiazolidinedione or oxazolidinedione ring is preserved, preventing the formation of ring-opened byproducts or over-reduced alcohol species that are common in hydride reductions. Furthermore, the use of formic acid creates an acidic environment that can suppress base-catalyzed degradation pathways often observed in alkaline reduction conditions. The result is a reaction profile where the primary impurity burden is significantly lowered, facilitating a straightforward workup procedure that typically involves simple filtration of the catalyst followed by pH adjustment and crystallization, ensuring the final intermediate meets stringent pharmaceutical quality standards.
How to Synthesize Thiazolidinedione Derivatives Efficiently
Implementing this synthesis route requires careful selection of the catalyst system and reaction conditions to match the specific substitution pattern of the R1 and R2 groups on the precursor molecule. The patent outlines a versatile protocol where the formula (II) compound is treated with formic acid, potentially diluted with water or organic co-solvents like ethyl acetate or toluene to manage solubility and heat dissipation. Operators can choose between homogeneous catalysts for maximum activity in complex substrates or heterogeneous catalysts like 5% Rh/C or PtO2 for ease of separation and catalyst recycling. The reaction progress is typically monitored via HPLC, with completion often achieved within 0.5 to 40 hours depending on the temperature and catalyst loading. Detailed standard operating procedures for scaling this reaction from laboratory benchtop to pilot plant vessels are essential to maintain safety and consistency, particularly when managing the exothermic decomposition of formic acid.
- Prepare the reaction mixture by combining the formula (II) precursor with formic acid (acting as both solvent and hydrogen donor) and a selected transition metal catalyst such as Pd/C or homogeneous Ir complexes.
- Heat the mixture to temperatures between 0-150°C, optionally under hydrogen pressure (0.1-50 bar) if using catalytic hydrogenation, or under reflux for transfer hydrogenation protocols.
- Upon completion, filter off the heterogeneous catalyst, concentrate the filtrate, and isolate the high-purity formula (I) product through pH adjustment and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this formic acid-mediated reduction technology presents a compelling value proposition centered on cost stability and operational safety. By replacing expensive and hazardous stoichiometric reducing agents with catalytic amounts of transition metals and commodity-grade formic acid, the direct material costs of the synthesis are substantially reduced. The elimination of complex purification steps, such as column chromatography, translates into shorter cycle times and higher throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations. Additionally, the reduced solvent footprint lowers the logistical burden of solvent procurement, storage, and recovery, contributing to a leaner and more cost-effective manufacturing operation overall.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with inexpensive formic acid and the ability to recover and reuse heterogeneous catalysts. Since formic acid acts as both the reductant and the solvent, the volume of auxiliary solvents required is drastically minimized, leading to significant savings in solvent purchase and waste treatment costs. The high selectivity of the reaction reduces the loss of valuable starting materials to byproducts, thereby improving the overall mass balance and yield of the process. Furthermore, the simplified isolation procedure reduces labor hours and energy consumption associated with distillation and drying, compounding the financial advantages for large-scale production facilities.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like formic acid and widely available transition metal catalysts mitigates the risk of supply disruptions often associated with specialized or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and pressures, ensures consistent production output even with minor variations in raw material quality. This reliability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates, preventing costly production stoppages. The ability to switch between hydrogenation and transfer hydrogenation modes provides additional flexibility, allowing production sites to utilize existing equipment without requiring massive capital investment in new high-pressure hydrogenation infrastructure.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner alternative that simplifies compliance with increasingly strict waste discharge regulations. The reduction in hazardous waste generation, particularly the absence of heavy metal sludge from stoichiometric reducers and large volumes of organic solvent waste, streamlines the permitting process for manufacturing sites. The scalability of the process is proven by the patent examples which demonstrate effectiveness from gram scale up to multi-kilogram batches, indicating a clear path to multi-ton commercial production. This environmental efficiency not only reduces liability but also enhances the corporate sustainability profile of the manufacturing organization, a key metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented reduction technology in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios for thiazolidinedione-based drugs.
Q: What are the primary advantages of using formic acid over metal hydrides for thiazolidinedione reduction?
A: Using formic acid eliminates the safety hazards associated with pyrophoric metal hydrides like LiAlH4 and significantly reduces impurity profiles, often achieving conversions above 97% without the >10% impurity levels seen in conventional magnesium/methanol reductions.
Q: Can this process be adapted for both homogeneous and heterogeneous catalysis?
A: Yes, the patent demonstrates versatility by successfully employing both homogeneous catalysts like Ir(COD)Cl and heterogeneous systems such as Pd/C, PtO2, and Raney Ni, allowing manufacturers to choose based on cost and separation requirements.
Q: How does this method impact the purification of anti-diabetic drug intermediates?
A: The method simplifies purification by allowing easy filtration of solid catalysts and utilizing formic acid which can be removed or neutralized easily, resulting in high-purity isolates suitable for subsequent API synthesis steps without extensive chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazolidinedione Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of next-generation anti-diabetic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic reduction methods described in CN1491218A are executed with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of thiazolidinedione derivative meets the exacting standards required by global regulatory agencies. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth downstream processing and final API approval.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your pharmaceutical supply chain.
