Advanced Enzymatic Synthesis of Thiamphenicol and Florfenicol Key Intermediates for Commercial Scale-up
The pharmaceutical and veterinary industries are constantly seeking more efficient pathways to produce broad-spectrum antibiotics, particularly chloramphenicol derivatives like Thiamphenicol and Florfenicol. Patent CN1743308A introduces a groundbreaking synthetic methodology that fundamentally shifts the paradigm from traditional racemic resolution to highly selective enzymatic asymmetric synthesis. This innovation addresses critical inefficiencies in the manufacturing of these vital veterinary drugs by establishing chiral centers with exceptional precision right from the initial steps. By leveraging specific hydroxynitrile lyases found in natural sources such as Badam seeds, the process achieves enantiomeric excess values exceeding 96 percent without the need for cumbersome separation techniques. This technical advancement not only streamlines the production workflow but also significantly enhances the economic viability of manufacturing high-purity antibiotic intermediates on a commercial scale.
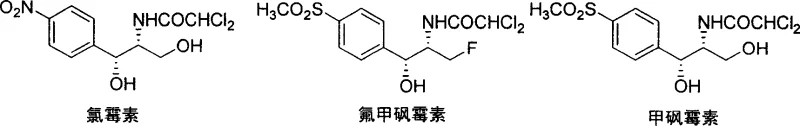
For procurement managers and supply chain directors, understanding the structural nuances and synthetic origins of these compounds is essential for securing reliable long-term contracts. The ability to produce these active pharmaceutical ingredients (APIs) through a convergent and waste-minimized route translates directly into supply chain resilience. As global demand for animal health products continues to rise, manufacturers who adopt this superior synthetic strategy will possess a distinct competitive advantage in terms of cost structure and environmental compliance. This report analyzes the technical depth of Patent CN1743308A to demonstrate why this specific enzymatic route represents the gold standard for modern reliable veterinary drug intermediate supplier capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Thiamphenicol and Florfenicol has relied heavily on the p-thioanisaldehyde route, which inherently suffers from significant material inefficiency due to its dependence on racemic synthesis. As illustrated in traditional schemes, these legacy processes involve the creation of a mixture of D and L-type substituted phenylserine isomers, necessitating a resolution step to isolate the pharmacologically active L-isomer. This resolution process typically utilizes tartaric acid or similar chiral resolving agents, resulting in the discard of approximately half of the synthesized material, which represents a massive economic loss and increased waste disposal burden. Furthermore, the multi-step nature of these conventional routes often involves harsh reaction conditions and expensive reagents that complicate purification and lower overall throughput. The cumulative effect of these inefficiencies is a higher cost of goods sold (COGS) and a larger environmental footprint, which poses challenges for manufacturers aiming to meet increasingly stringent global regulatory standards for green chemistry.
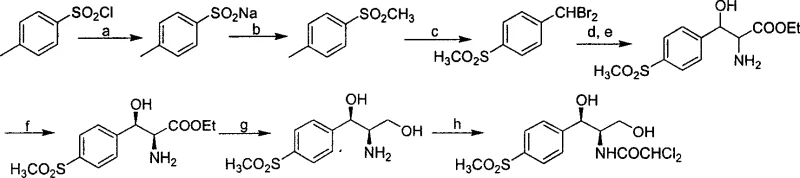
The Novel Approach
In stark contrast, the methodology disclosed in Patent CN1743308A circumvents these historical bottlenecks by employing a chiral pool strategy initiated by biocatalysis. The novel route begins with the enzymatic hydrocyanation of p-methylthiobenzaldehyde, a reaction that installs the first chiral center with high stereoselectivity, thereby eliminating the need for downstream resolution entirely. This approach allows for the theoretical utilization of nearly all starting materials, drastically improving atom economy. The synthesis proceeds through a series of well-controlled transformations, including protection, reduction, and cyclization, to generate a versatile key intermediate (Compound 7) that can be divergently converted into either Thiamphenicol or Florfenicol. This flexibility is a strategic asset for production planning, allowing manufacturers to adjust output ratios based on market demand without retooling the entire upstream process. The integration of enzymatic steps with standard organic synthesis creates a robust hybrid pathway that balances the selectivity of biology with the scalability of chemistry.
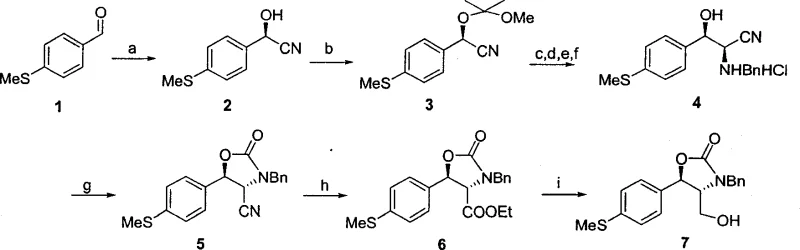
Mechanistic Insights into Enzymatic Hydrocyanation and Chiral Induction
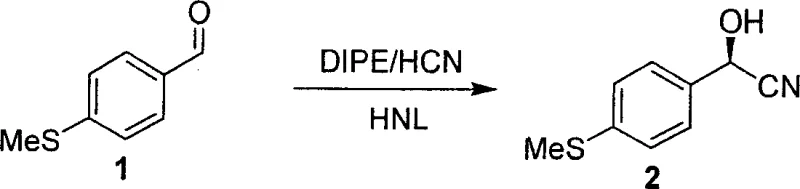
The cornerstone of this innovative synthesis is the enzyme-catalyzed hydrocyanation reaction, which utilizes hydroxynitrile lyase (HNL) to convert the achiral aldehyde substrate into a chiral cyanohydrin. The patent highlights the screening of various natural enzyme sources, identifying that HNL derived from Badam seeds exhibits superior catalytic activity and stereoselectivity for this specific substrate compared to other sources like almonds or apples. Mechanistically, the enzyme stabilizes the transition state of the cyanide addition to the carbonyl group, enforcing a specific spatial orientation that yields the desired (R)-cyanohydrin with an enantiomeric excess (ee) greater than 96 percent. This high level of stereocontrol at the very first step is critical because it dictates the configuration of all subsequent chiral centers in the molecule through substrate-controlled diastereoselective reactions. By establishing the correct absolute configuration early, the synthesis avoids the formation of difficult-to-separate diastereomers later in the sequence, simplifying purification and ensuring high final product purity.
Following the enzymatic step, the synthesis employs a clever strategy of chiral induction to establish the second stereocenter required for the antibiotic activity. The protected cyanohydrin is reduced to an imine and subsequently reacted with benzylamine, where the existing chirality at the alpha-position influences the facial selectivity of the nucleophilic attack. This intramolecular guidance ensures that the new amino group is installed in the correct relative configuration, maintaining the stereochemical integrity of the molecule. The subsequent cyclization with carbonyldiimidazole forms a rigid oxazolidinone ring, which locks the conformation of the two chiral centers and protects the sensitive amine and hydroxyl functionalities during further transformations. This mechanistic design not only ensures high optical purity but also provides a stable scaffold that withstands the rigorous conditions of subsequent oxidation and fluorination steps required to produce Florfenicol. For R&D directors, this level of mechanistic control offers confidence in the reproducibility and robustness of the process when scaling from laboratory to pilot plant.
How to Synthesize Key Thiamphenicol Intermediate Efficiently
The practical implementation of this synthesis involves a sequence of optimized reactions designed for high yield and operational simplicity. The process begins with the preparation of the biocatalyst, followed by the hydrocyanation in a biphasic or organic solvent system containing controlled amounts of water to maintain enzyme activity. Subsequent steps involve standard organic transformations such as silyl or acetal protection, reduction with DIBAL-H, and reductive amination, all of which are well-understood unit operations in fine chemical manufacturing. The formation of the oxazolidinone ring is a pivotal step that consolidates the molecular complexity, after which the side chain can be modified to introduce the dichloroacetamide group for Thiamphenicol or the fluoro-methyl group for Florfenicol. Detailed standard operating procedures for each transformation, including specific reagent ratios, temperature controls, and workup protocols, are essential for ensuring consistent quality. For a comprehensive guide on executing these steps with precision, please refer to the standardized synthesis protocol below.
- Perform enzymatic hydrocyanation of p-methylthiobenzaldehyde using hydroxynitrile lyase (HNL) from Badam seeds to achieve high enantiomeric excess.
- Protect the hydroxyl group and reduce the nitrile to an imine, followed by amination with benzylamine to establish the second chiral center.
- Cyclize using carbonyldiimidazole to form the oxazolidinone ring, followed by esterification and reduction to yield the key alcohol intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic route described in Patent CN1743308A offers substantial strategic benefits for procurement and supply chain management, primarily driven by the elimination of wasteful resolution steps and the use of cost-effective biocatalysts. Traditional manufacturing methods incur a hidden tax on production efficiency by discarding half of the synthesized intermediates, whereas this enzymatic approach maximizes raw material utilization. This fundamental improvement in atom economy translates directly into cost reduction in antibiotic manufacturing, as fewer kilograms of starting materials are required to produce the same amount of finished API. Furthermore, the reliance on naturally sourced enzymes reduces dependency on expensive chiral catalysts or resolving agents that are subject to market volatility. The simplified workflow also reduces the number of unit operations, leading to lower energy consumption and reduced solvent usage, which aligns with sustainability goals and lowers waste treatment costs.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the avoidance of racemic resolution, which traditionally limits maximum yield to 50 percent for the desired isomer. By achieving high enantioselectivity through enzymatic catalysis, the process effectively doubles the theoretical yield from the same amount of starting aldehyde. Additionally, the use of Badam seed extract as a biocatalyst source provides a low-cost alternative to proprietary synthetic enzymes or metal complexes. The reduction in processing steps also lowers labor and overhead costs associated with extended reaction times and complex purification sequences. These factors combine to create a significantly leaner cost structure, allowing suppliers to offer more competitive pricing while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials such as p-methylthiobenzaldehyde and common reagents like sodium cyanide and benzylamine. The enzymatic step operates under mild conditions (room temperature), reducing the risk of equipment failure or safety incidents associated with high-pressure or cryogenic processes. Moreover, the convergent nature of the synthesis, where a single key intermediate branches into two different final products, allows manufacturers to respond flexibly to fluctuations in demand for Thiamphenicol versus Florfenicol. This agility minimizes the risk of inventory obsolescence and ensures continuous supply even if market preferences shift. The robustness of the enzymatic reaction also means that production is less susceptible to batch-to-batch variability, ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the compatibility of the enzymatic step with standard reactor configurations and the use of common organic solvents. The high selectivity of the reaction minimizes the formation of byproducts, simplifying downstream purification and reducing the load on wastewater treatment facilities. This aligns with modern environmental regulations that penalize high E-factors (mass of waste per mass of product). The elimination of heavy metal catalysts and toxic resolving agents further enhances the environmental profile of the manufacturing process, making it easier to obtain regulatory approvals in strict jurisdictions. The process is designed to be scalable from 100 kgs to 100 MT annual production capacity without significant re-engineering, ensuring that supply can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for stakeholders evaluating this technology. Understanding these aspects is crucial for making informed decisions about sourcing strategies and process adoption. The answers reflect the proven capabilities of the method as demonstrated in the experimental examples provided in the intellectual property filing.
Q: How does this new synthesis method improve upon conventional resolution processes?
A: Conventional methods rely on resolving racemic mixtures, discarding up to 50% of the material. This patent utilizes enzymatic asymmetric synthesis to directly generate the desired chiral configuration with >96% ee, effectively doubling theoretical yield and reducing waste.
Q: What represents the key chiral intermediate in this pathway?
A: The process generates a versatile oxazolidinone intermediate (Compound 7) which serves as a common precursor for both Thiamphenicol and Florfenicol, allowing for flexible manufacturing based on market demand.
Q: Is the enzymatic step scalable for industrial production?
A: Yes, the patent demonstrates that the hydroxynitrile lyase catalyzed reaction proceeds efficiently at room temperature with simple workup procedures, making it highly suitable for large-scale commercial manufacturing without requiring extreme cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiamphenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic asymmetric synthesis route detailed in Patent CN1743308A for the production of high-value veterinary antibiotics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory chemistry into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of biocatalytic and chemical steps is maintained with precision. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and impurity profiles at every stage of production. By partnering with us, clients can leverage this advanced technology to secure a sustainable and cost-effective supply of critical antibiotic intermediates.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how this synthesis route can optimize your supply chain. We offer a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this enzymatic process for your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments tailored to your project needs. Our goal is to provide engineering solutions that resolve production bottlenecks and enhance your competitive position in the global animal health market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
