Advanced Synthesis of Enantiomerically Pure Piperazine Derivatives for Commercial API Manufacturing
Introduction to Novel Chiral Intermediates in Patent CN1742002A
The pharmaceutical industry constantly seeks more efficient pathways to produce enantiomerically pure active ingredients, particularly for blockbuster drugs like Levocetirizine. Patent CN1742002A introduces a groundbreaking class of novel piperazine derivatives that serve as robust synthetic intermediates for preparing pharmaceutically active compounds. These compounds, characterized by the general Formula (I), feature a specific asymmetric center that is crucial for biological activity. The innovation lies not just in the molecule itself, but in the stability of its stereochemistry during subsequent chemical transformations. By utilizing these intermediates, manufacturers can bypass the cumbersome and often yield-limiting resolution steps typically required at the end of a synthesis line. This patent represents a significant leap forward in process chemistry, offering a reliable pharmaceutical intermediate supplier pathway that prioritizes both purity and operational simplicity.
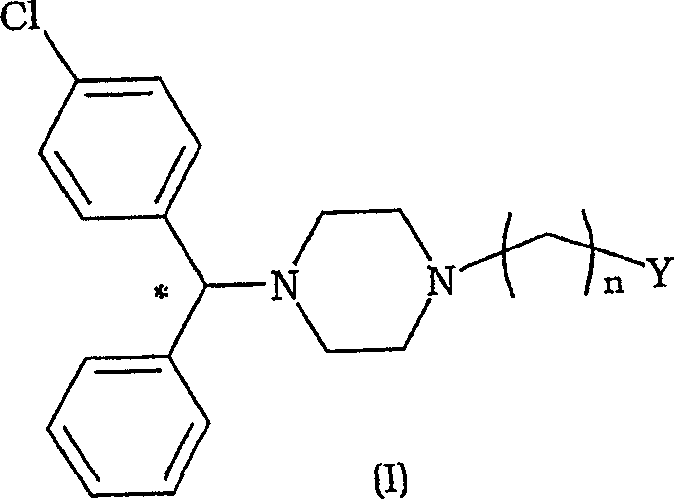
The core structure involves a chlorophenyl-phenyl-methyl group attached to a piperazine ring, which is further substituted with an alkyl chain terminating in a hydroxyl or leaving group. This specific architecture allows for versatile downstream modifications while maintaining the integrity of the chiral center. For R&D directors focusing on impurity profiles, the ability to introduce the chiral element early in the synthesis and maintain it through strong basic conditions is a critical advantage. It eliminates the formation of difficult-to-remove diastereomeric impurities that often plague traditional routes. Furthermore, the patent details specific preferences for the leaving group Y, such as halogens or sulfonates, providing a toolkit for chemists to optimize reaction kinetics based on their specific equipment and safety protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Levocetirizine and related antihistamines has relied heavily on the resolution of racemic mixtures at a late stage of synthesis. Conventional methods often involve synthesizing the racemic cetirizine first and then attempting to separate the active levo-isomer from the inactive dextro-isomer. This approach is inherently inefficient, as it theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity and cost. Moreover, late-stage resolution often requires expensive chiral resolving agents or preparative HPLC, which are difficult to scale economically. The harsh conditions sometimes needed for these separations can also lead to degradation of the sensitive piperazine core, resulting in lower overall purity and increased waste generation. For procurement managers, these inefficiencies translate directly into higher raw material costs and longer lead times for API manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN1742002A shifts the chiral introduction to an earlier stage using the stable intermediates of Formula (I). By starting with an enantiomerically pure piperazine ethanol derivative, the subsequent alkylation steps proceed with remarkable fidelity. The patent highlights that surprisingly, no racemization occurs even under strongly basic conditions, which is a common pitfall in piperazine chemistry. This allows for the direct synthesis of the target molecule, such as the compound shown in Formula (II), with high optical purity from the outset. 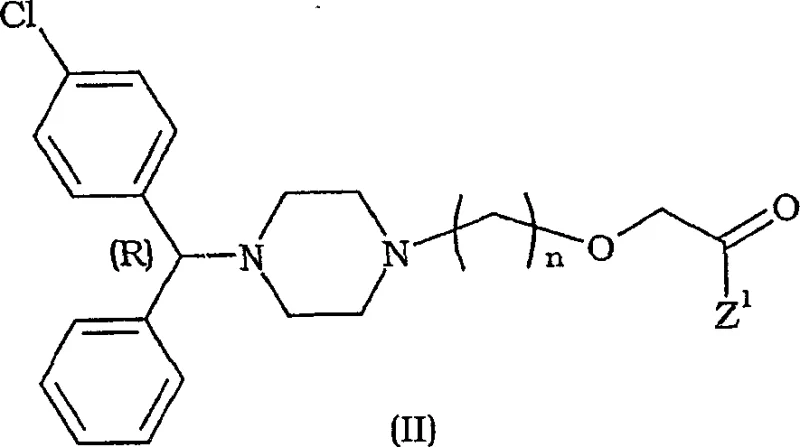 This novel approach effectively decouples the chiral resolution from the final coupling steps, enabling the use of cheaper, achiral reagents for the bulk of the synthesis. The result is a streamlined process that significantly reduces the number of purification cycles required, thereby enhancing the overall throughput and reducing the environmental footprint of the manufacturing process.
This novel approach effectively decouples the chiral resolution from the final coupling steps, enabling the use of cheaper, achiral reagents for the bulk of the synthesis. The result is a streamlined process that significantly reduces the number of purification cycles required, thereby enhancing the overall throughput and reducing the environmental footprint of the manufacturing process.
Mechanistic Insights into Chiral Stability and Alkylation
The mechanistic success of this invention relies on the unique electronic and steric environment surrounding the chiral carbon atom in Formula (I). Typically, benzylic protons adjacent to a nitrogen atom are acidic and prone to racemization via an iminium ion intermediate, especially in the presence of bases. However, the specific substitution pattern in these novel derivatives stabilizes the configuration against epimerization. The patent data indicates that reactions can be conducted at temperatures ranging from 0°C to reflux without compromising the enantiomeric excess (ee). This stability is paramount for ensuring that the final API meets the stringent regulatory requirements for chiral purity, often needing >99% ee. For technical teams, understanding this stability profile allows for broader solvent selection, including polar aprotic solvents like THF or DMF, which can accelerate reaction rates without sacrificing product quality.
Furthermore, the versatility of the leaving group Y enables a wide array of nucleophilic substitutions. Whether reacting with haloacetates to form the acetic acid side chain of Levocetirizine or coupling with protected hydroxylamines to create lipoxygenase inhibitors, the core chiral scaffold remains intact. 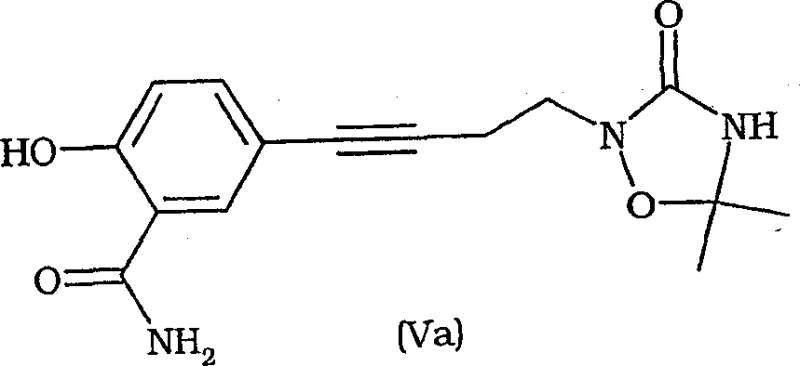 This modularity is demonstrated in the synthesis of Formula (Va) intermediates, where the piperazine core is successfully coupled with complex aromatic systems via Sonogashira-type conditions or direct alkylation. The ability to tolerate such diverse reaction conditions without degrading the chiral center underscores the robustness of this chemical platform. It provides a unified strategy for producing a family of related therapeutics, simplifying inventory management and raw material sourcing for large-scale chemical producers.
This modularity is demonstrated in the synthesis of Formula (Va) intermediates, where the piperazine core is successfully coupled with complex aromatic systems via Sonogashira-type conditions or direct alkylation. The ability to tolerate such diverse reaction conditions without degrading the chiral center underscores the robustness of this chemical platform. It provides a unified strategy for producing a family of related therapeutics, simplifying inventory management and raw material sourcing for large-scale chemical producers.
How to Synthesize Enantiomerically Pure Piperazine Derivatives Efficiently
The synthesis protocol outlined in the patent offers a clear roadmap for producing these high-value intermediates. It begins with the alkylation of a readily available piperazine precursor, followed by a highly efficient chiral separation step. The detailed standardized synthesis steps below provide a guide for implementing this technology in a GMP environment.
- Preparation of the racemic piperazine ethanol precursor via alkylation of 1-[(4-chlorophenyl)phenylmethyl]piperazine with 2-chloroethanol.
- Chiral separation of the racemic mixture using industrial chromatography (e.g., CHIRALPAK AD) to isolate the desired (R)-enantiomer with high optical purity.
- Conversion of the chiral alcohol intermediate into the final active pharmaceutical ingredient or further functionalized derivative via nucleophilic substitution without racemization.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound benefits for the commercial viability of API production. By shifting the resolution step earlier and utilizing stable intermediates, manufacturers can achieve substantial cost savings through improved material efficiency. The elimination of late-stage racemate disposal means that nearly all input materials contribute to the final product yield, drastically improving the atom economy of the process. For supply chain heads, this translates to a more predictable and reliable supply of critical intermediates, reducing the risk of stockouts caused by low-yielding batch failures. The use of common industrial solvents and reagents further mitigates supply chain risks associated with specialty chemicals.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive dynamic kinetic resolution catalysts or complex enzymatic steps often required in traditional routes. By relying on robust chemical alkylation with high yields (reported up to 80-90% in specific steps), the overall cost of goods sold (COGS) is significantly lowered. The high purity achieved through simple crystallization rather than repeated chromatography reduces solvent consumption and waste treatment costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The intermediates described are chemically stable and can be stored for extended periods without degradation, allowing for strategic stockpiling. This stability ensures that production schedules are not disrupted by the short shelf-life issues common with reactive chiral building blocks. Additionally, the scalability of the chiral chromatography step using SMB technology ensures that supply can be ramped up quickly to meet market demand fluctuations without requiring extensive new capital investment.
- Scalability and Environmental Compliance: The synthetic route avoids the use of heavy metal catalysts in the key chiral-defining steps, simplifying the purification process and ensuring the final API meets strict limits for residual metals. The reduced solvent usage and higher yields inherently lower the E-factor (environmental factor) of the process. This aligns with modern green chemistry principles, making it easier for pharmaceutical companies to meet their sustainability goals and regulatory compliance standards regarding waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy.
Q: What is the primary advantage of using Formula (I) intermediates for Levocetirizine synthesis?
A: The primary advantage is the exceptional stability of the chiral center. Unlike conventional methods that risk racemization during harsh reaction conditions, this novel route ensures the (R)-enantiomer remains intact, delivering high optical purity (>98% ee) essential for therapeutic efficacy.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly describes the use of scalable techniques such as Simulated Moving Bed (SMB) chromatography and standard crystallization processes. The reaction conditions utilize common solvents like THF and isopropanol, facilitating easy transition from laboratory to commercial scale.
Q: Does this process apply to other therapeutic areas besides antihistamines?
A: Absolutely. Beyond Levocetirizine, these intermediates are versatile precursors for lipoxygenase inhibitors and other 1,4-substituted piperazines. The modular nature of the leaving group (Y) allows for diverse downstream functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of antihistamines and other therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of piperazine derivatives meets the highest international standards. Our commitment to quality ensures that your downstream synthesis proceeds without interruption or unexpected impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply of these novel intermediates can optimize your manufacturing efficiency and reduce time-to-market for your final pharmaceutical products.
