Advanced Z-Isomer Enrichment Technology for High-Purity Risperidone Manufacturing
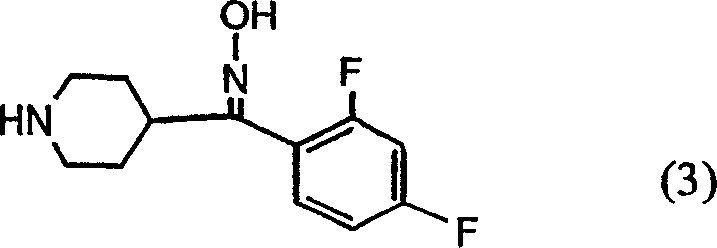
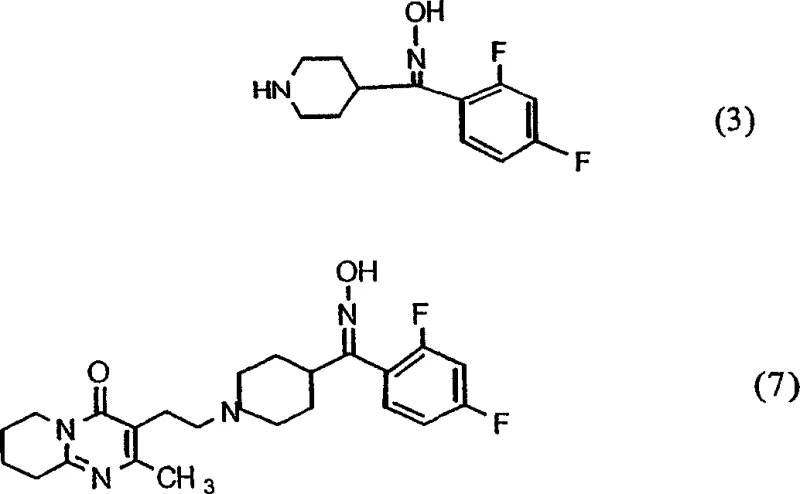
The pharmaceutical industry continuously seeks robust synthetic pathways that maximize yield while minimizing impurity profiles, particularly for high-volume antipsychotic agents like Risperidone. Patent CN1720228A introduces a transformative approach to this challenge by focusing on the stereochemical control of key oxime intermediates. Historically, the formation of the benzisoxazole ring, a critical structural motif in Risperidone, was hindered by the generation of geometric isomer mixtures that complicated downstream processing. This patent discloses a novel methodology utilizing acetate salts to selectively enrich the Z-isomer of formula (3) or (7), thereby streamlining the entire production workflow. By leveraging the differential solubility properties of these acetate salts, manufacturers can achieve superior purity levels that were previously difficult to attain through conventional crystallization techniques alone. This technological advancement represents a significant leap forward for any reliable pharmaceutical intermediates supplier aiming to optimize their production lines for cost and quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Risperidone often relied on the direct oximation of ketone precursors, which invariably resulted in a mixture of Z and E geometric isomers. In these legacy processes, the E-isomer was found to be substantially unreactive during the subsequent cyclization step required to close the benzisoxazole ring. Consequently, the presence of the E-isomer acted as a diluent that not only lowered the overall reaction yield but also introduced complex impurity profiles that required extensive and costly purification efforts. For instance, earlier methods reported cyclization yields as low as 46 percent when starting from non-enriched mixtures, creating significant bottlenecks in commercial scale-up of complex pharmaceutical intermediates. Furthermore, the inability to easily separate these isomers meant that valuable raw materials were effectively wasted, driving up the cost of goods sold and reducing the economic viability of the manufacturing process.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally alters this dynamic by introducing a selective precipitation step based on acetate salt formation. By reacting the crude oxime mixture with acetic acid, the process exploits the fact that the Z-isomer acetate salt possesses markedly lower solubility in polar solvents compared to its E-isomer counterpart. This physical distinction allows for the preferential crystallization of the desired Z-isomer, which can be isolated with high stereochemical purity, often exceeding 90 percent Z-content. This enrichment ensures that the subsequent cyclization reaction proceeds with maximum efficiency, as the reaction vessel is populated almost exclusively by the reactive species. Additionally, the mother liquor containing the residual E-isomer can be recycled through thermal isomerization, further enhancing the atom economy of the process and supporting substantial cost savings in fine chemical manufacturing.
Mechanistic Insights into Acetate-Mediated Isomer Enrichment
The core mechanism driving this process improvement lies in the thermodynamic and kinetic differences between the geometric isomers when converted into their salt forms. When the oxime intermediate is treated with acetic acid, protonation occurs at the imine nitrogen, forming an acetate salt that stabilizes the molecular structure. Crucially, the spatial arrangement of the Z-isomer facilitates a crystal lattice packing that is energetically more favorable for precipitation in solvents like ethanol or n-butanol. In contrast, the E-isomer remains solvated due to steric hindrance and different dipole interactions that prevent efficient crystal growth under the same conditions. This selective phase transition allows for a physical separation that is far more scalable and economically feasible than chromatographic methods, providing a clear pathway for cost reduction in API manufacturing. The result is a highly enriched feedstock that drives the downstream cyclization reaction towards completion with minimal side reactions.
Furthermore, the patent elucidates a secondary mechanism for maximizing yield through the thermal isomerization of the unwanted E-isomer. By heating the mother liquor containing the E-isomer acetate in the presence of an acid catalyst, such as acetic acid or ammonium acetate, the equilibrium can be shifted towards the more stable Z-configuration. This conversion typically occurs at elevated temperatures, for example above 80 degrees Celsius, allowing the recovered material to re-enter the precipitation cycle. This closed-loop capability ensures that nearly all theoretical material can eventually be converted into the reactive Z-form, drastically reducing waste generation. For R&D teams, understanding this equilibrium is vital for optimizing reaction times and temperatures to ensure that the final product meets stringent purity specifications without requiring excessive solvent usage or energy input.
How to Synthesize Z-Enriched Risperidone Oxime Efficiently
Implementing this synthesis route requires precise control over reaction conditions to ensure the successful formation and isolation of the Z-isomer acetate salt. The process begins with the oximation of the ketone precursor in a suitable solvent system, followed by the careful addition of acetic acid to induce salt formation. Operators must monitor temperature and concentration closely to favor the nucleation of the Z-isomer crystals while keeping the E-isomer in solution. Once the solid is isolated, it is converted back to the free base using a mild alkaline treatment before proceeding to the cyclization step. The detailed standardized synthesis steps see the guide below.
- React the ketone precursor with hydroxylamine in the presence of acetic acid to form a mixture of Z and E oxime isomers.
- Induce preferential precipitation of the Z-isomer acetate salt from the reaction mixture by cooling or adding anti-solvents.
- Separate the solid Z-enriched acetate salt, convert it to the free base, and proceed to cyclization to form the benzisoxazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Z-enrichment technology offers profound advantages in terms of operational stability and cost structure. By eliminating the bottleneck caused by unreactive isomers, the overall throughput of the manufacturing facility can be significantly increased without the need for additional capital investment in reactor volume. The ability to recycle the E-isomer fraction back into the process means that raw material consumption is optimized, leading to a drastic simplification of the supply chain logistics regarding precursor sourcing. Moreover, the reliance on simple crystallization rather than complex chromatography reduces the dependency on expensive consumables and specialized equipment, which translates directly into a more resilient and cost-effective production model. This robustness is essential for maintaining continuous supply in the face of market fluctuations.
- Cost Reduction in Manufacturing: The primary economic driver here is the substantial increase in yield during the critical cyclization step. By feeding the reactor with highly enriched Z-isomer material, the formation of by-products is minimized, and the consumption of reagents per kilogram of final product is drastically reduced. The elimination of expensive purification steps, such as preparative HPLC or multiple recrystallizations, further lowers the operational expenditure. Additionally, the recovery and reuse of the E-isomer fraction ensure that the effective cost of the starting ketone is amortized over a much larger output of final API, creating a leaner manufacturing cost profile that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: A process that is less sensitive to impurity buildup is inherently more reliable for long-term production campaigns. The simplicity of the acetate precipitation step means that batch-to-batch variability is minimized, ensuring consistent quality that meets regulatory standards without deviation. This consistency reduces the risk of batch failures or rejections, which are major disruptors in the pharmaceutical supply chain. Furthermore, the reagents required, such as acetic acid and common alcohols, are commodity chemicals with stable availability, reducing the risk of supply shortages that could halt production lines and delay deliveries to downstream customers.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by maximizing atom economy and minimizing solvent waste. The ability to recycle the mother liquor reduces the volume of hazardous waste requiring disposal, lowering the environmental footprint of the facility. Scalability is also enhanced because crystallization is a unit operation that scales linearly and predictably from pilot plant to commercial tonnage. This makes the technology ideal for meeting growing global demand for Risperidone without the engineering challenges associated with scaling complex separation technologies, ensuring a sustainable and compliant manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this isomer enrichment technology in commercial settings. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation to assist technical teams in evaluating feasibility. Understanding these nuances is critical for successful technology transfer and process validation.
Q: Why is Z-isomer enrichment critical for Risperidone synthesis?
A: Only the Z-isomer of the oxime intermediate effectively undergoes cyclization to form the benzisoxazole ring. The E-isomer is largely unreactive under standard conditions, leading to lower yields and difficult purification if not removed prior to the cyclization step.
Q: How does the acetate salt method improve process efficiency?
A: The acetate salt of the Z-isomer has significantly lower solubility in polar solvents compared to the E-isomer salt. This physical property allows for selective crystallization, enabling the isolation of highly pure Z-isomer material without complex chromatography.
Q: Can the unwanted E-isomer be recycled?
A: Yes, the mother liquor containing the E-isomer can be subjected to thermal isomerization in the presence of an acid catalyst, converting the unreactive E-form back into the reactive Z-form for further processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Risperidone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of modern pharmaceutical synthesis demands a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to full-scale manufacturing is seamless and efficient. We understand the critical importance of maintaining stringent purity specifications for intermediates like the Z-enriched oximes described in CN1720228A, and our rigorous QC labs are equipped to verify every batch against the highest international standards. By partnering with us, you gain access to a supply chain that is not only robust but also technically sophisticated enough to handle the nuances of stereoselective synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific production needs. We are prepared to provide a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of switching to this enriched isomer pathway. Please contact us to request specific COA data and route feasibility assessments that will empower your decision-making process. Let us help you secure a competitive advantage through superior chemical manufacturing solutions.
