Advanced Catalytic Synthesis of Risperidone Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for the production of critical antipsychotic agents, with Risperidone standing as a cornerstone therapy for schizophrenia and bipolar disorder. Patent CN112794849A introduces a transformative synthetic methodology for producing 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one, a pivotal intermediate in the Risperidone value chain. This innovation addresses long-standing challenges in process chemistry by replacing hazardous chlorinating agents with a safer, high-yielding catalytic cycle. For R&D Directors and Process Chemists, this patent represents a significant leap forward in impurity control and reaction efficiency, utilizing ethyl acetoacetate and 1-bromo-2-chloroethane as foundational building blocks. The strategic shift away from traditional phosphoryl chloride chemistry not only enhances the safety profile of the manufacturing process but also streamlines the purification workflow, ensuring a consistent supply of high-purity intermediates essential for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrido[1,2-a]pyrimidin-4-one derivatives has relied heavily on the use of phosphorus oxychloride (POCl3) as a dual cyclizing and chlorinating agent, a approach fraught with significant operational and environmental drawbacks. As illustrated in prior art such as CN1705661A, the conventional route involves the condensation of 2-aminopyridine with lactone derivatives in the presence of excessive POCl3 and toluene, generating substantial quantities of acidic waste gas and liquid effluent. This reliance on aggressive halogenating reagents necessitates complex and costly waste neutralization protocols, creating a heavy burden on environmental compliance and increasing the overall cost of goods sold. Furthermore, the harsh reaction conditions often lead to the formation of difficult-to-remove chlorinated byproducts, complicating the impurity profile and requiring extensive chromatographic purification which is impractical at a commercial scale. The logistical challenges of handling and storing large volumes of corrosive POCl3 also pose inherent safety risks to plant personnel and infrastructure, making this traditional pathway increasingly untenable for modern, green chemistry-focused manufacturing facilities.
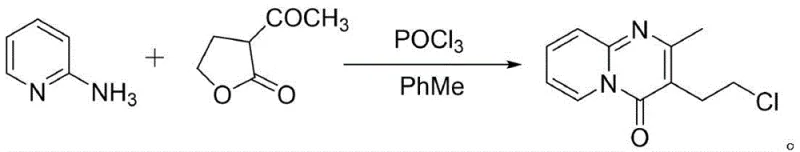
The Novel Approach
In stark contrast to the legacy methods, the novel synthetic strategy disclosed in the patent employs a modular two-step sequence that prioritizes atom economy and operational safety. The process initiates with a nucleophilic substitution reaction between ethyl acetoacetate and 1-bromo-2-chloroethane under alkaline conditions to form ethyl 2-acetyl-4-chlorobutyrate, followed by a catalytic cyclization with 2-aminopyridine. This route completely circumvents the need for phosphorus-based chlorinating agents, thereby eliminating the generation of hazardous phosphorous waste streams and significantly reducing the environmental footprint of the production facility. The use of imidazole hydrochloride as a mild organic salt catalyst in the second step facilitates the ring closure under thermal conditions that are far more manageable than the aggressive reflux required by POCl3 methods. By leveraging readily available commodity chemicals and avoiding the complexities of handling toxic gases, this new approach offers a scalable and economically superior alternative for the production of high-purity pharmaceutical intermediates.
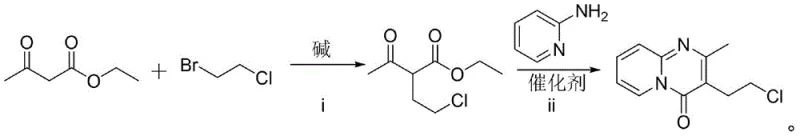
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The core of this technological advancement lies in the precise optimization of the cyclization step, where the interaction between ethyl 2-acetyl-4-chlorobutyrate and 2-aminopyridine is mediated by a specific organic salt catalyst. Mechanistic analysis suggests that imidazole hydrochloride acts as an effective proton shuttle and potentially a mild Lewis acid promoter, facilitating the nucleophilic attack of the pyridine nitrogen onto the carbonyl carbon of the ester moiety. This catalytic environment lowers the activation energy required for the ring-closing condensation, allowing the reaction to proceed efficiently at temperatures between 100°C and 110°C. Unlike metal halides such as lithium chloride or ferric chloride, which showed inferior performance in screening studies, the imidazole system provides a unique balance of acidity and solubility that drives the equilibrium towards the desired bicyclic product while minimizing side reactions. The result is a clean reaction profile that yields the target 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one with exceptional purity, reducing the burden on downstream purification units.
Furthermore, the initial alkylation step is meticulously controlled to prevent the formation of elimination byproducts or dialkylated impurities. The protocol specifies an activation phase at low temperatures (-20°C) using sodium hydride in DMF to generate the enolate of ethyl acetoacetate quantitatively before the introduction of the alkyl halide. This temperature-controlled addition ensures that the nucleophilic substitution occurs selectively at the gamma-position relative to the ketone, preserving the integrity of the beta-keto ester functionality required for the subsequent cyclization. By maintaining strict control over the stoichiometry and thermal profile during this exothermic alkylation, the process achieves yields as high as 97% for the intermediate, establishing a robust foundation for the final cyclization step. This level of mechanistic understanding allows for precise impurity management, ensuring that the final intermediate meets the stringent specifications required for GMP-grade API synthesis.
How to Synthesize 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one Efficiently
The practical implementation of this synthesis requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the alkylated precursor, followed by the catalytic ring closure, with careful attention paid to solvent selection and thermal ramping. Detailed standard operating procedures regarding reagent addition rates, quenching protocols, and crystallization conditions are critical for reproducibility. For a comprehensive guide on the exact molar ratios, stirring speeds, and work-up procedures validated in the patent examples, please refer to the standardized synthesis steps outlined below.
- Perform nucleophilic substitution of ethyl acetoacetate with 1-bromo-2-chloroethane using NaH in DMF at -20°C activation and 100°C reaction temperature to yield ethyl 2-acetyl-4-chlorobutyrate.
- React the resulting ethyl 2-acetyl-4-chlorobutyrate with 2-aminopyridine using imidazole hydrochloride as a catalyst at 100-110°C for 6 hours.
- Purify the crude product via recrystallization from ethyl acetate to obtain the high-purity target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The elimination of phosphorus oxychloride from the supply chain removes a significant regulatory and logistical bottleneck, as this reagent is subject to strict monitoring and requires specialized storage infrastructure. By substituting it with stable, commodity-grade reagents like ethyl acetoacetate and imidazole hydrochloride, manufacturers can drastically simplify their raw material sourcing strategies and reduce inventory holding costs. The simplified waste profile means that effluent treatment costs are significantly reduced, directly improving the margin structure of the final intermediate. Moreover, the high conversion rates and selectivity of the new process minimize the loss of valuable starting materials, ensuring that every kilogram of input translates more efficiently into saleable product, thereby enhancing overall resource utilization.
- Cost Reduction in Manufacturing: The transition to a non-halogenating agent route fundamentally alters the cost structure by removing the need for expensive corrosion-resistant equipment and complex scrubbing systems. Without the requirement to neutralize massive volumes of acidic off-gas, the operational expenditure related to environmental health and safety compliance is substantially lowered. Additionally, the high yields reported in the patent examples indicate a reduction in raw material consumption per unit of output, which serves as a powerful lever for lowering the variable cost of production. The use of imidazole hydrochloride, a cost-effective organic salt, further contributes to economic efficiency compared to precious metal catalysts or exotic reagents.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key raw materials, ethyl acetoacetate and 2-aminopyridine, are widely produced bulk chemicals with established global supply networks. This reduces the risk of supply disruptions that are often associated with specialized or regulated reagents like POCl3. The robustness of the reaction conditions also implies a wider operating window, making the process less susceptible to minor fluctuations in utility supplies or ambient conditions, thus ensuring consistent batch-to-batch delivery performance. This reliability is crucial for maintaining continuous API production schedules and meeting the demanding timelines of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common solvents like DMF and straightforward thermal controls that are easily replicated in large-scale reactors. The absence of hazardous gas evolution simplifies the engineering requirements for reactor venting and containment, allowing for faster technology transfer from pilot plant to commercial manufacturing. From a sustainability perspective, the drastic reduction in toxic waste generation aligns with modern green chemistry principles and corporate ESG goals, making the supply chain more resilient to tightening environmental regulations. This future-proofing of the manufacturing process ensures long-term viability and reduces the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process robustness and quality control. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of this synthesis method over traditional POCl3 routes?
A: This method eliminates the use of highly corrosive and toxic phosphorus oxychloride (POCl3), significantly reducing environmental waste and safety hazards associated with chlorinating agents.
Q: What catalyst is used to achieve high yields in the cyclization step?
A: Imidazole hydrochloride is identified as the optimal catalyst, providing superior yields (up to 95%) compared to metal chlorides like LiCl or FeCl3.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available raw materials like ethyl acetoacetate and operates under relatively mild conditions without requiring complex post-treatment for hazardous waste removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant intermediate synthesis in the global pharmaceutical supply chain. Our team of expert process chemists has extensively evaluated the methodology described in CN112794849A and possesses the technical capability to execute this route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering intermediates that consistently meet the high standards required for antipsychotic drug formulation.
We invite procurement leaders and R&D partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized version of this catalytic cyclization process, we can help you achieve significant reductions in total landed cost while securing a stable supply of high-quality materials. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing excellence can support your long-term strategic goals in the psychiatric therapeutic area.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
