Advanced Green Synthesis of Risperidone Intermediate for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for synthesizing critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN112794849B introduces a groundbreaking synthetic method for 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one, a pivotal intermediate in the production of risperidone, a widely prescribed atypical antipsychotic medication. This innovation addresses significant bottlenecks in existing manufacturing protocols by eliminating the need for hazardous chlorinating agents such as phosphorus oxychloride (POCl3) and avoiding complex multi-step sequences involving noble metal catalysts. By streamlining the synthesis into a concise two-step process involving alkylation and subsequent cyclization, this technology offers a robust solution for cost reduction in pharmaceutical intermediate manufacturing. For R&D directors and procurement specialists, understanding the mechanistic advantages and scalability of this route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent global quality standards while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of risperidone and its key intermediates has been plagued by inefficient and environmentally taxing processes. Traditional routes often involve the condensation of 2-aminopyridine with 3-acetyldihydrofuran-2(3H)-one, followed by hydrogenation using palladium on carbon (Pd/C) and subsequent halogenation. As illustrated in the conventional pathway below, these methods require multiple isolation steps and the use of expensive transition metal catalysts, which introduce risks of heavy metal contamination in the final drug product.
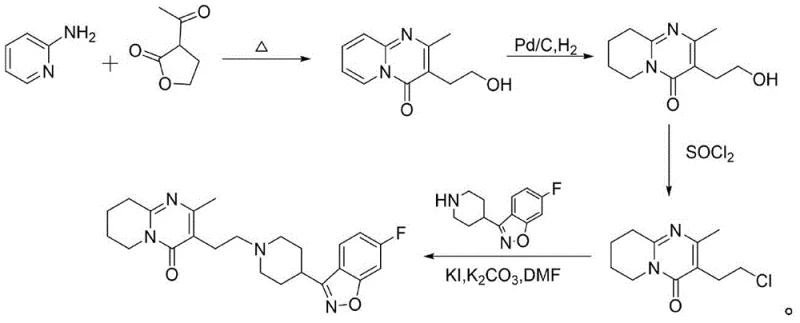
Furthermore, alternative prior art methods, such as those described in CN1705661A, rely heavily on phosphorus oxychloride (POCl3) for the chlorination step. While chemically effective, the use of POCl3 generates substantial amounts of acidic waste gas and liquid effluents, creating severe environmental compliance challenges and increasing waste disposal costs. The handling of such corrosive reagents also necessitates specialized equipment and rigorous safety protocols, complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112794849B proposes a streamlined, two-step synthesis that bypasses the need for toxic chlorinating agents and noble metals entirely. The new strategy initiates with the alkylation of ethyl acetoacetate with 1-bromo-2-chloroethane under mild alkaline conditions to form ethyl 2-acetyl-4-chlorobutyrate. This intermediate is then directly cyclized with 2-aminopyridine using an organic salt catalyst. This approach not only simplifies the operational workflow but also drastically reduces the generation of hazardous byproducts. The reaction scheme below highlights the elegance of this direct construction of the pyrido[1,2-a]pyrimidin-4-one core, offering a clear path toward high-purity pharmaceutical intermediates with minimal downstream purification requirements.
![Novel two-step green synthesis route for 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one](/insights/img/chloroethyl-methyl-pyrido-pyrimidinone-synthesis-supplier-20260305113535-05.webp)
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The success of this novel synthetic route hinges on the precise optimization of reaction conditions, particularly in the second step where the heterocyclic core is formed. The cyclization of ethyl 2-acetyl-4-chlorobutyrate with 2-aminopyridine is facilitated by imidazole hydrochloride, which acts as an effective organocatalyst. Mechanistically, the catalyst likely promotes the nucleophilic attack of the amino group on the carbonyl carbon, followed by intramolecular displacement of the chloride or elimination-addition sequences to close the ring. The patent data indicates that maintaining the reaction temperature between 100°C and 110°C is critical; lower temperatures result in incomplete conversion, while excessive heat can lead to decomposition or polymerization. This thermal sensitivity underscores the importance of precise process control in achieving the reported 95% isolated yield.
Impurity control is another vital aspect of this mechanism, especially for pharmaceutical applications where regulatory limits are strict. The first step, involving the alkylation of ethyl acetoacetate with sodium hydride (NaH) in DMF, must be carefully managed to prevent over-alkylation or hydrolysis of the ester group. The patent specifies an activation temperature of -20°C followed by a reaction temperature of 100°C, which maximizes the formation of the desired mono-alkylated product while suppressing side reactions. By isolating this intermediate or proceeding with a telescoped process, manufacturers can ensure that the final API contains negligible levels of genotoxic impurities, thereby satisfying the rigorous demands of reducing lead time for high-purity pharmaceutical intermediates without compromising safety.
How to Synthesize 3-(2-Chloroethyl)-2-Methyl-4H-Pyrido[1,2-a]Pyrimidin-4-One Efficiently
Implementing this synthesis requires adherence to specific operational parameters to replicate the high yields and purity described in the patent. The process begins with the preparation of the alkylated beta-keto ester, followed by the cyclization step. Detailed standard operating procedures regarding stoichiometry, solvent drying, and quenching protocols are essential for successful technology transfer. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol outlined below.
- Perform substitution reaction between ethyl acetoacetate and 1-bromo-2-chloroethane under alkaline conditions (NaH/DMF) to obtain ethyl 2-acetyl-4-chlorobutyrate.
- React the resulting ethyl 2-acetyl-4-chlorobutyrate with 2-aminopyridine using imidazole hydrochloride as a catalyst at 100-110°C.
- Purify the final product through recrystallization to achieve high purity suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical efficiency. The elimination of phosphorus oxychloride and palladium catalysts removes two major cost drivers and supply chain vulnerabilities associated with traditional methods. Sourcing and disposing of hazardous chlorinating agents often incur significant logistical overhead and regulatory scrutiny, whereas the reagents used in this new method—such as ethyl acetoacetate and imidazole salts—are commodity chemicals with stable global supply chains. This shift translates directly into cost reduction in pharmaceutical intermediate manufacturing by lowering raw material expenses and waste treatment fees.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts like Pd/C eliminates the need for costly recovery processes and extensive heavy metal testing, which are mandatory for API release. Furthermore, the avoidance of POCl3 reduces the capital expenditure required for corrosion-resistant reactor linings and scrubber systems. The high yields reported (up to 97% in step one and 95% in step two) mean that less raw material is wasted per kilogram of product, significantly improving the overall mass balance and economic viability of the process.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials rather than specialized, hazard-classified reagents, manufacturers can mitigate the risk of supply disruptions. The simplified two-step sequence also shortens the production cycle time, allowing for faster response to market demand fluctuations. This agility is crucial for maintaining continuous supply of critical psychiatric medications like risperidone, ensuring that downstream formulation partners receive their intermediates without delay.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions and generates minimal hazardous waste, aligning perfectly with modern green chemistry principles and increasingly strict environmental regulations. This ease of compliance facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to environmental violations. The robustness of the reaction conditions suggests that scaling from pilot plant to multi-ton commercial production can be achieved with minimal re-optimization, ensuring a stable long-term supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the detailed experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the practical implications of adopting this technology for large-scale production.
Q: Why is this new synthesis method considered greener than traditional routes?
A: Unlike traditional methods that rely on highly toxic phosphorus oxychloride (POCl3) or expensive palladium catalysts, this novel route utilizes benign alkylating agents and organic salt catalysts, significantly reducing hazardous waste generation.
Q: What are the key yield improvements observed in this patent?
A: The patent reports isolated yields of up to 97% for the intermediate alkylation step and 95% for the final cyclization step, demonstrating superior efficiency compared to older multi-step processes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids harsh conditions and uses commercially available raw materials like ethyl acetoacetate, making it highly scalable and economically viable for ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-Chloroethyl)-2-Methyl-4H-Pyrido[1,2-a]Pyrimidin-4-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the synthetic route described in CN112794849B and confirmed its potential for robust, large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required for FDA and EMA filings.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced synthesis technology. By partnering with us, you gain access to a secure supply of this key risperidone intermediate, optimized for both cost and quality. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to support your next project milestone.
