Advanced Manufacturing of Substituted Imidazopyridines for High-Purity API Intermediates
Advanced Manufacturing of Substituted Imidazopyridines for High-Purity API Intermediates
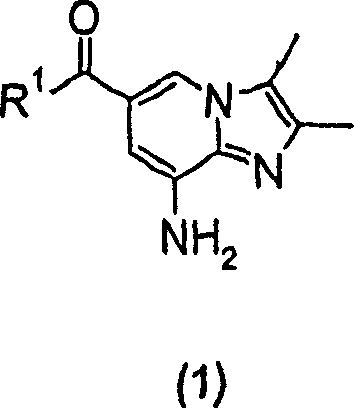
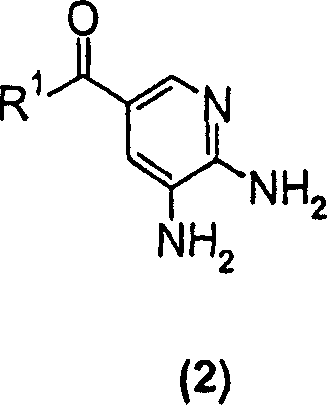
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical backbones for active pharmaceutical ingredients (APIs). Patent CN1255404C discloses a groundbreaking methodology for the large-scale preparation of substituted imidazopyridine compounds, specifically targeting the 2,3-dimethyl-imidazo[1,2-a]pyridine core found in potent gastric acid secretion inhibitors. This technology represents a significant leap forward in process chemistry, addressing the longstanding challenges of low yields and excessive reaction times associated with traditional cyclization methods. By leveraging a novel solvent system based on cyclohexanone, the invention enables the efficient condensation of 5,6-diaminopyridine derivatives with 3-halo-2-butanone compounds. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic and economic advantages of this route is essential for securing a stable supply chain for next-generation gastrointestinal therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in EP33094 and WO99/55706, typically rely on inert solvents like acetone, benzene, N,N-dimethylformamide (DMF), or tetrahydrofuran (THF) to facilitate the cyclization of aminopyridine precursors. These conventional processes are plagued by severe inefficiencies that hinder commercial viability. Specifically, the reaction times are excessively long, often ranging from 16 to 84 hours, which creates a bottleneck in manufacturing throughput and significantly increases energy consumption. Furthermore, these reactions frequently require elevated temperatures that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to remove. Most critically, the isolated yields in these traditional protocols are disappointingly low, typically falling between 22% and 55%. Such poor atom economy not only drives up the cost of goods sold (COGS) but also generates substantial chemical waste, posing environmental compliance challenges for modern facilities aiming for green chemistry standards.
The Novel Approach
In stark contrast, the novel approach detailed in CN1255404C utilizes cyclohexanone as the primary reaction medium, fundamentally altering the kinetics and thermodynamics of the cyclization event. This strategic solvent choice allows the reaction between the formula (2) diaminopyridine compound and the 3-halo-2-butanone reagent to proceed with remarkable speed and efficiency. The reaction time is drastically shortened to a mere 1 to 4 hours, representing a potential tenfold increase in reactor utilization compared to legacy methods. Moreover, the process operates at moderate temperatures of 80-100°C, which are easier to maintain and control on a large scale. The result is a dramatic improvement in productivity, with transformation efficiencies routinely exceeding 95% and isolated yields consistently surpassing 70%. This shift from a sluggish, low-yielding process to a rapid, high-yielding one exemplifies the kind of process intensification that drives cost reduction in API manufacturing.
Mechanistic Insights into Cyclohexanone-Mediated Cyclization
The core of this innovation lies in the condensation mechanism between the 1,2-diamine functionality of the pyridine ring and the alpha-halo ketone moiety of the butanone derivative. While the fundamental organic transformation involves nucleophilic attack followed by dehydration and ring closure, the choice of cyclohexanone appears to play a crucial role beyond simple solvation. It is hypothesized that cyclohexanone provides an optimal polarity environment that stabilizes the transition states involved in the ring closure while effectively solubilizing both the polar diamino starting material and the less polar halo-ketone reagent. This homogeneity likely facilitates better molecular collisions and reduces the activation energy required for the cyclization. Additionally, the solvent properties may assist in the precipitation of the final imidazopyridine product upon cooling, driving the equilibrium forward according to Le Chatelier's principle and simplifying the isolation procedure to a straightforward filtration.
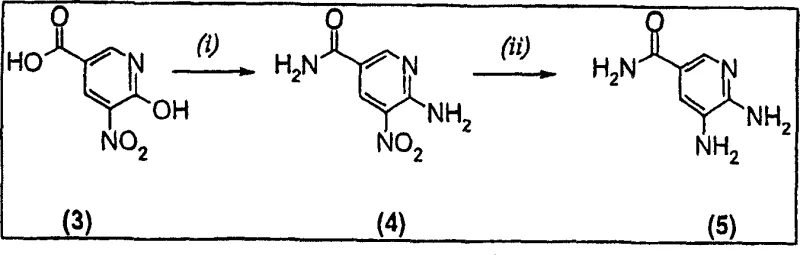
Impurity control is another critical aspect where this mechanism excels. In traditional solvents like DMF, side reactions such as over-alkylation or polymerization of the reactive halo-ketone can occur due to prolonged exposure to heat. By shortening the reaction window to just a few hours, the novel process minimizes the residence time of reactive intermediates, thereby suppressing the formation of high-molecular-weight byproducts and regioisomers. The precursor synthesis, often involving the reduction of nitro-groups as shown in Scheme 1 of the patent, is also optimized to ensure high purity before the cyclization step begins. This holistic approach to impurity management ensures that the final API intermediate meets stringent purity specifications required for regulatory filing, reducing the burden on downstream purification teams.
How to Synthesize 8-Amino-2,3-dimethyl-imidazo[1,2-a]pyridine Efficiently
The synthesis of these valuable heterocycles is streamlined into a concise operational sequence that is highly amenable to standard chemical processing equipment. The process begins with the suspension of the 5,6-diaminopyridine derivative in cyclohexanone, followed by the controlled addition of the 3-halo-2-butanone reagent, such as 3-bromo-2-butanone or 3-chloro-2-butanone. The mixture is then heated to reflux conditions, typically around 100°C internal temperature, where the cyclization proceeds rapidly. Detailed standard operating procedures regarding stoichiometry, addition rates, and specific workup protocols are critical for reproducibility and safety.
- Prepare the 5,6-diaminopyridine precursor via nitro-reduction or amidation of nicotinic acid derivatives.
- Suspend the diamino compound in cyclohexanone and add 3-halo-2-butanone (e.g., 3-bromo-2-butanone).
- Heat the mixture to 80-100°C for 1-4 hours, then cool to precipitate the high-purity imidazopyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary value driver is the substantial reduction in manufacturing costs achieved through process intensification. By eliminating the need for extended reaction times and expensive, hard-to-remove solvents like DMF, the facility can significantly lower its utility bills and solvent recovery costs. The high conversion rates mean that less raw material is wasted, directly improving the margin on every kilogram of product produced. Furthermore, the simplicity of the workup—often requiring only cooling, filtration, and washing with tert-butyl methyl ether (TBME)—removes the need for resource-intensive chromatographic purification steps, which are notoriously difficult to scale and expensive to operate in a GMP environment.
- Cost Reduction in Manufacturing: The elimination of long reaction cycles and the use of cost-effective solvents like cyclohexanone lead to significant operational savings. The high yield ensures that the cost per unit of the active intermediate is minimized, providing a competitive edge in pricing negotiations with downstream API manufacturers. Additionally, the avoidance of complex purification techniques reduces the consumption of silica gel and eluents, further driving down variable costs.
- Enhanced Supply Chain Reliability: The reagents required for this process, including cyclohexanone and various halo-butanones, are commodity chemicals with robust global supply chains. This availability mitigates the risk of raw material shortages that can plague projects relying on exotic or specialized reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram scale to multi-kilogram batches in the patent examples. The reduced reaction time allows for more batches to be run in the same reactor volume per week, effectively increasing capacity without capital expenditure. From an environmental perspective, the reduced solvent usage and higher atom economy align with green chemistry principles, simplifying waste treatment and helping the facility meet increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imidazopyridine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what partners can expect when integrating this route into their supply chain. Understanding these details is vital for risk assessment and project planning.
Q: What are the primary advantages of using cyclohexanone over traditional solvents like DMF?
A: Cyclohexanone significantly reduces reaction time from up to 84 hours to merely 1-4 hours while increasing isolated yields from roughly 50% to over 70%, drastically improving throughput.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes simple filtration and washing steps (e.g., with TBME) rather than complex chromatography, making it highly scalable for multi-kilogram manufacturing.
Q: What is the purity profile of the resulting imidazopyridine intermediates?
A: The method achieves transformation efficiencies usually exceeding 95%, allowing for the isolation of high-purity solids suitable for subsequent coupling reactions without extensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Amino-2,3-dimethyl-imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of imidazopyridine intermediate meets the exacting standards required for global regulatory submissions. Our infrastructure is designed to handle the specific solvent systems and thermal profiles required by this novel cyclohexanone-based process safely and efficiently.
We invite you to collaborate with us to leverage this advanced technology for your gastrointestinal drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this superior synthetic route. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data for our reference standards, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of these critical building blocks for the future of medicine.
