Scalable Iodine-Catalyzed C3 Selenization of Imidazopyridine Derivatives for Commercial Production
Introduction to Advanced C3 Functionalization Technology
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable synthetic routes for high-value intermediates. A significant breakthrough in this domain is documented in patent CN108191856B, which discloses a novel method for the selenization of the C3 position of imidazopyridine derivatives. This technology represents a paradigm shift from traditional transition-metal catalysis to a more economical and environmentally benign iodine-catalyzed system. Imidazopyridine scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for numerous bioactive molecules targeting various therapeutic areas including oncology and infectious diseases. The ability to selectively functionalize the C3 position with selenium moieties opens new avenues for drug discovery, as organoselenium compounds exhibit unique biological activities and serve as versatile building blocks for further derivatization. This report analyzes the technical merits and commercial viability of this patented process, highlighting its potential to redefine supply chain strategies for pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of imidazopyridine rings, particularly at the C3 position, has been fraught with significant chemical and economic challenges. Traditional protocols often rely heavily on the use of expensive noble metal catalysts such as palladium, copper, or rhodium complexes, which not only inflate the raw material costs but also introduce severe complications in the purification process. The presence of residual heavy metals in the final product is a critical quality attribute that is strictly regulated in the pharmaceutical industry, necessitating additional and costly scavenging steps to meet stringent purity specifications. Furthermore, many existing methods require the assistance of strong protonic acids or harsh reaction conditions that can degrade sensitive functional groups, thereby limiting the substrate scope and forcing chemists to employ complex protecting group strategies. These factors collectively contribute to longer lead times, lower overall yields, and a larger environmental footprint due to increased waste generation, making conventional routes less attractive for large-scale commercial manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the patented technology utilizes elemental iodine as a simple yet highly effective catalyst to drive the selenization reaction. This approach completely bypasses the need for noble metal salts and protonic acid additives, offering a cleaner and more atom-economical pathway. The reaction proceeds smoothly under mild thermal conditions, typically around 85°C, using common organic solvents like dichloroethane and inexpensive inorganic bases such as potassium carbonate. As illustrated in the general structural formula below, this method accommodates a wide variety of substituents on the imidazopyridine core, including alkyl, alkoxy, and halogen groups, demonstrating exceptional functional group tolerance.
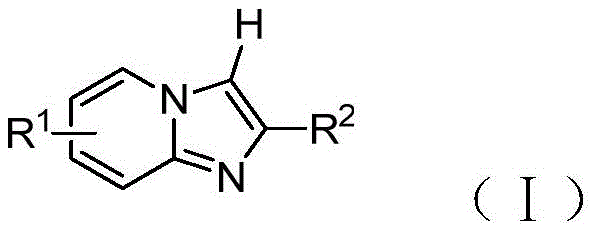
The versatility of this system allows for the use of diverse diselenide reagents, greatly expanding the chemical space accessible to process chemists. By eliminating the reliance on scarce precious metals, this novel approach not only reduces the direct cost of goods but also mitigates supply chain risks associated with the volatility of the noble metal market. The operational simplicity, combined with high conversion rates and minimal byproduct formation, positions this iodine-catalyzed method as a superior alternative for the industrial production of high-purity selenium-containing heterocycles.
Mechanistic Insights into Iodine-Catalyzed C-H Selenylation
The success of this transformation lies in the unique ability of molecular iodine to activate the selenium-selenium bond in diselenide reagents, facilitating a selective C-H functionalization at the C3 position of the imidazopyridine ring. Unlike radical pathways that might lack selectivity or require harsh initiators, the iodine-catalyzed mechanism likely involves the formation of an electrophilic selenium species that attacks the electron-rich C3 site. This electrophilic aromatic substitution or oxidative addition pathway is finely tuned by the basic environment provided by carbonates, which helps to neutralize any acidic byproducts and drive the equilibrium towards the desired selenized product. The absence of strong acids ensures that the nitrogen atoms in the fused heterocyclic system remain unprotonated and available for coordination or electronic modulation, which is crucial for maintaining the integrity of the scaffold during the reaction.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions such as over-selenization or decomposition of the heterocyclic core. The mildness of the iodine catalyst prevents the degradation of sensitive substituents like halogens, which are often prone to dehalogenation under more aggressive transition-metal catalytic conditions. This high level of chemoselectivity results in a cleaner crude reaction profile, significantly reducing the burden on downstream purification units. For R&D teams, understanding this mechanism is vital for troubleshooting and optimizing the process for new analogues, as it suggests that electron-donating groups on the substrate may enhance reactivity while electron-withdrawing groups might require slight adjustments in reaction time or temperature to maintain optimal yields.
How to Synthesize C3-Selenized Imidazopyridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction monitoring to ensure consistent quality. The process is designed to be robust, utilizing standard equipment and reagents that are readily available from global chemical suppliers. The following section outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process chemists to replicate the high yields reported in the literature. By adhering to these parameters, manufacturers can achieve reproducible results while maintaining strict control over critical process attributes.
- Combine the imidazopyridine derivative substrate with diphenyl diselenide and elemental iodine catalyst in an organic solvent such as dichloroethane.
- Add an inorganic base like potassium carbonate to the reaction mixture and heat the system to 85-90°C for 6 to 12 hours.
- Upon completion, extract the product with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-catalyzed technology translates into tangible strategic benefits that extend beyond simple unit cost savings. The elimination of noble metal catalysts removes a major variable from the sourcing equation, insulating the production process from the price volatility and geopolitical supply risks often associated with palladium and other precious metals. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, leads to a drastic reduction in processing time and consumable usage, thereby enhancing overall operational efficiency. This streamlined workflow allows for faster batch turnover and improved capacity utilization, which are critical metrics for meeting tight delivery schedules in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive elemental iodine results in a significant decrease in raw material expenditures. Additionally, the removal of heavy metal purification steps reduces the consumption of specialized scavenger resins and solvents, leading to substantial cost savings in the overall manufacturing budget. The high yields achieved, often exceeding ninety percent in optimized examples, mean that less starting material is wasted, further improving the cost-efficiency of the process and maximizing the return on investment for every kilogram of substrate processed.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including diphenyl diselenide and potassium carbonate, are commodity chemicals with stable and abundant global supply chains. This availability ensures that production schedules are not disrupted by raw material shortages, providing a reliable foundation for long-term manufacturing planning. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in reagent quality or environmental factors, ensuring consistent output quality and reducing the risk of batch failures that could delay shipments to downstream customers.
- Scalability and Environmental Compliance: The use of mild reaction temperatures and common solvents makes this process highly amenable to scale-up from benchtop to multi-ton production without requiring specialized high-pressure or cryogenic equipment. From an environmental standpoint, the atom economy of the reaction is superior to many alternatives, generating less hazardous waste and reducing the load on wastewater treatment facilities. This alignment with green chemistry principles not only lowers disposal costs but also supports corporate sustainability goals, making the final product more attractive to environmentally conscious partners and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenization technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this chemistry into their existing production portfolios.
Q: What are the advantages of using elemental iodine over noble metal catalysts for this selenization?
A: Elemental iodine eliminates the need for expensive and toxic noble metal salts like palladium or copper, significantly reducing raw material costs and simplifying downstream purification by removing heavy metal contamination risks.
Q: Does this method tolerate sensitive functional groups on the imidazopyridine ring?
A: Yes, the protocol demonstrates excellent functional group compatibility, successfully accommodating halogens, alkyl groups, and alkoxy substituents without requiring protective groups or harsh acidic conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely, the method utilizes readily available reagents, operates at moderate temperatures (85°C), and achieves high yields (up to 93%), making it highly robust for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-catalyzed selenization technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of imidazopyridine derivative delivered meets the highest industry standards for safety and efficacy. We are committed to leveraging advanced synthetic methodologies to drive innovation and efficiency in the global supply chain.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your specific API intermediate requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your development timelines with speed, precision, and commercial acumen.
