Advanced Base-Catalyzed Decarboxylation for Commercial Scale Production of 4-Hydroxyquinolines
Advanced Base-Catalyzed Decarboxylation for Commercial Scale Production of 4-Hydroxyquinolines
The pharmaceutical and agrochemical industries constantly seek robust synthetic pathways that balance high purity with economic efficiency, particularly for heterocyclic scaffolds like 4-hydroxyquinolines. Patent CN1329598A introduces a transformative methodology for preparing 4-hydroxyquinolines and their tautomeric compounds, specifically addressing the synthesis of critical intermediates such as 5,7-dichloro-4-hydroxyquinoline (DCHQ). Unlike traditional thermal or acidic routes, this invention utilizes a mild base-catalyzed decarboxylation process operating at temperatures not exceeding 200°C, preferably between 90°C and 160°C. This technical breakthrough allows for the efficient conversion of 4-hydroxyquinolinecarboxylic acids and their esters into the desired quinoline core without the severe equipment corrosion associated with strong mineral acids. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles while simplifying the overall manufacturing workflow for complex heterocyclic intermediates.
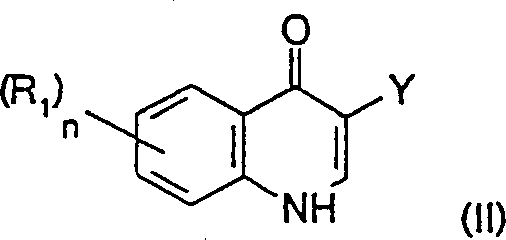
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4-hydroxyquinoline derivatives has been plagued by severe operational constraints and safety hazards inherent to legacy synthetic protocols. Classical methods, such as those described in Organic Syntheses, typically require thermal decarboxylation at extreme temperatures exceeding 230°C, which demands specialized high-energy heating systems and poses significant risks of thermal runaway or product degradation. Alternative approaches, exemplified by U.S. Pat-A-5731440, attempt to lower the temperature to the 120-165°C range but rely heavily on aggressive strong acid media such as concentrated sulfuric, phosphoric, or hydrochloric acid. The reliance on such highly corrosive environments necessitates the use of expensive glass-lined reactors or Hastelloy equipment, drastically increasing capital expenditure and maintenance costs for manufacturing facilities. Furthermore, the generation of large volumes of acidic waste streams creates substantial environmental compliance burdens and complicates downstream wastewater treatment processes, making these conventional routes increasingly unsustainable for modern green chemistry initiatives.
The Novel Approach
The inventive method disclosed in CN1329598A fundamentally shifts the paradigm by employing alkaline conditions to facilitate the decarboxylation of 4-hydroxyquinolinecarboxylic acid precursors at moderate temperatures. By utilizing inorganic bases like potassium hydroxide or sodium hydroxide, or even organic bases like quaternary ammonium hydroxides, the reaction proceeds smoothly at temperatures up to 200°C, with optimal results observed between 90°C and 160°C. This approach effectively eliminates the need for corrosive strong acids, thereby allowing the use of standard stainless steel reactors which are far more cost-effective and durable for long-term commercial production. The process is remarkably versatile, accommodating various precursors including carboxylic acids, esters, and nitriles, and can be conducted in aqueous media, organic solvents, or even under solvent-free conditions depending on the specific substrate requirements. This flexibility not only enhances the safety profile of the operation but also significantly simplifies the work-up procedure, as the product can often be isolated directly via filtration or simple acidification.
Mechanistic Insights into Base-Catalyzed Decarboxylation
The core chemical transformation in this novel process involves the base-promoted removal of the carboxyl group from the 3-position of the quinoline ring, a reaction that is kinetically facilitated by the electron-withdrawing nature of the adjacent nitrogen atom and the 4-hydroxyl group. Under the alkaline conditions described, the carboxylic acid precursor is initially deprotonated to form a carboxylate salt, which then undergoes decarboxylation upon heating to release carbon dioxide and generate the stabilized quinoline anion. This anionic intermediate exists in a dynamic tautomeric equilibrium, as illustrated in the patent documentation, where the negative charge is delocalized between the oxygen at the 4-position and the nitrogen atom within the heterocyclic ring system. The presence of alkali metal cations (M+) stabilizes these anionic forms, preventing premature protonation and ensuring high conversion rates even at relatively mild thermal conditions compared to neutral thermal decarboxylation. Understanding this equilibrium is crucial for process optimization, as the final isolation step often involves shifting this equilibrium back to the neutral lactam or lactim form through controlled acidification.
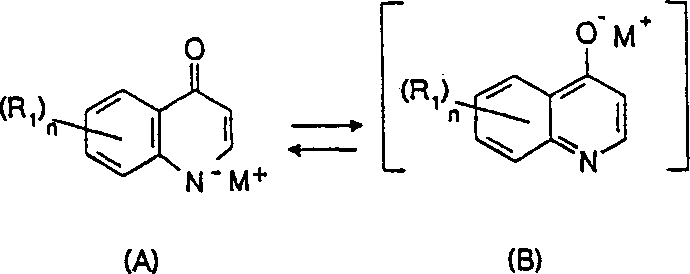
From an impurity control perspective, the basic medium offers a distinct advantage by suppressing acid-catalyzed side reactions that often lead to chlorinated byproducts or ring-opening degradation in halogenated substrates like 5,7-dichloro derivatives. The patent data indicates that conversion rates (TT) can exceed 99% with yields (RR) reaching up to 99% under optimized conditions, such as using a KOH/substrate molar ratio of 1.5 to 3.5 at 150°C. The mechanism also allows for the simultaneous hydrolysis of ester precursors if present, meaning that ethyl or methyl esters of the quinoline carboxylic acid can be directly converted to the final hydroxyquinoline in a one-pot sequence without isolating the free acid intermediate. This telescoping capability reduces the number of unit operations, minimizes solvent usage, and lowers the overall material loss typically associated with multi-step isolation and purification procedures, thereby enhancing the overall mass balance of the synthesis.
How to Synthesize 5,7-Dichloro-4-hydroxyquinoline Efficiently
Implementing this base-catalyzed decarboxylation route requires careful attention to the stoichiometry of the base and the thermal profile to ensure complete conversion while minimizing energy input. The standardized protocol generally involves mixing the quinoline carboxylic acid precursor with an aqueous alkaline solution, such as 12-30% potassium hydroxide, and heating the mixture in a pressurized reactor to maintain liquid phase conditions above 100°C. Detailed operational parameters, including specific temperature ramps, stirring rates, and precise acidification steps for product recovery, are critical for achieving the high purity levels required for pharmaceutical applications. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized operating procedure outlined below.
- Prepare a reaction mixture containing the 4-hydroxyquinolinecarboxylic acid precursor and an aqueous alkaline solution (e.g., KOH or NaOH) with a molar ratio of 1: 1.5 to 1:3.5.
- Heat the mixture to a temperature between 90°C and 160°C, optionally adding a high-boiling organic solvent like Therminol VP1 to facilitate phase separation.
- Maintain heating until decarboxylation is complete (typically 5-15 hours), then isolate the product via filtration or acidification followed by filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this base-catalyzed technology translates directly into tangible reductions in both capital and operational expenditures across the manufacturing value chain. By eliminating the requirement for corrosive strong acids, manufacturers can utilize standard stainless steel equipment rather than investing in costly glass-lined reactors or exotic alloy vessels, resulting in significant upfront cost savings and reduced maintenance downtime. The milder reaction temperatures (≤200°C) compared to traditional thermal methods (>230°C) also lead to substantially lower energy consumption per kilogram of product, contributing to a reduced carbon footprint and lower utility bills. Furthermore, the ability to recycle alkaline filtrates, as demonstrated in the patent examples, minimizes raw material waste and reduces the volume of effluent requiring treatment, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The substitution of expensive corrosion-resistant infrastructure with standard reactor materials drastically lowers the barrier to entry for scaling this chemistry, while the high yields (>90%) minimize raw material waste. The elimination of strong acids removes the need for specialized acid-resistant piping and valves, further reducing the total cost of ownership for the production facility. Additionally, the potential for solvent-free operation or the use of recyclable high-boiling solvents reduces the recurring cost of solvent purchase and recovery systems.
- Enhanced Supply Chain Reliability: The robustness of the base-catalyzed process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by off-spec material or equipment failure due to corrosion. The use of commodity chemicals like potassium hydroxide and sodium hydroxide ensures a stable and readily available supply of reagents, mitigating the risk of raw material shortages that can plague specialty acid supplies. This reliability allows for more accurate forecasting and inventory management, ensuring continuous availability of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, as evidenced by successful autoclave trials, and generates less hazardous waste compared to acid-based routes, simplifying regulatory compliance and permitting. The ability to operate in aqueous media or with recyclable organic solvents reduces the environmental impact of volatile organic compound (VOC) emissions, facilitating smoother audits and inspections. This environmental friendliness enhances the corporate social responsibility profile of the supply chain, appealing to end-clients who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-catalyzed decarboxylation technology for 4-hydroxyquinoline production. These insights are derived directly from the experimental data and claims presented in patent CN1329598A, providing a factual basis for evaluating the process viability. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this route into existing manufacturing lines or for procurement teams negotiating supply contracts based on this specific intellectual property.
Q: What are the primary advantages of the base-catalyzed method over traditional acid hydrolysis?
A: The base-catalyzed method eliminates the need for highly corrosive strong acids like sulfuric or phosphoric acid, allowing the use of standard stainless steel reactors instead of expensive glass-lined equipment. Additionally, it operates at significantly lower temperatures (≤200°C) compared to thermal decarboxylation (>230°C), reducing energy consumption and thermal degradation risks.
Q: Can this process be scaled for industrial production of 5,7-dichloro-4-hydroxyquinoline?
A: Yes, the patent demonstrates high conversion rates (>99%) and yields (up to 99%) in autoclave experiments, indicating excellent scalability. The ability to recycle alkaline filtrates further enhances the economic feasibility for large-scale manufacturing.
Q: How is the product isolated after the reaction?
A: Depending on the base excess, the product may precipitate directly as a free quinoline compound or remain as a salt in the aqueous phase. If in salt form, acidification with dilute mineral acid precipitates the free product, which is then filtered and washed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxyquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies like the base-catalyzed decarboxylation described in CN1329598A to deliver high-value intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-hydroxyquinoline or 5,7-dichloro-4-hydroxyquinoline meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to technological excellence allows us to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on the quality or consistency of the final product.
We invite you to collaborate with us to leverage this innovative chemistry for your specific project needs, whether you require custom synthesis or large-scale commercial supply. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and to obtain specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable 4-hydroxyquinoline supplier dedicated to optimizing your supply chain through superior process chemistry and unwavering quality assurance.
