Optimizing Antifungal Production: Advanced Nickel-Catalyzed Terbinafine Synthesis for Commercial Scale
Introduction to Next-Generation Terbinafine Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and cost-efficient pathways for active pharmaceutical ingredients (APIs), particularly for widely prescribed antifungal agents like Terbinafine. Patent CN1993369A introduces a transformative synthetic methodology that addresses critical bottlenecks in traditional manufacturing, specifically focusing on the replacement of expensive noble metal catalysts with more abundant transition metals. This technical breakthrough centers on the utilization of divalent Nickel (Ni(II)) salts and complexes to facilitate the key carbon-carbon bond formation steps, offering a viable alternative to conventional Palladium-catalyzed processes. By leveraging this innovation, manufacturers can achieve substantial improvements in process economics without compromising the rigorous purity standards required for global regulatory compliance. The significance of this patent lies not merely in the chemical transformation itself, but in its holistic approach to industrial applicability, encompassing reagent availability, reaction safety, and downstream purification efficiency. For stakeholders evaluating long-term supply strategies, understanding the mechanistic nuances and operational advantages of this Nickel-catalyzed route is essential for maintaining competitiveness in the generic antifungal market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Terbinafine has relied heavily on cross-coupling reactions mediated by Palladium catalysts, such as tetrakis(triphenylphosphine)palladium(0) or dichloro-bis(triphenylphosphine)palladium(II). While these methods are chemically effective, they present severe economic and logistical challenges for large-scale production. The primary constraint is the exorbitant cost of Palladium complexes, which directly inflates the cost of goods sold (COGS) for the final API. Furthermore, conventional routes often necessitate the use of tributylacetylene as a key building block, a reagent that is not only expensive but also poses handling difficulties due to its volatility and stability issues. The reliance on these high-cost inputs creates a fragile supply chain vulnerable to fluctuations in precious metal markets. Additionally, traditional processes may generate significant amounts of heavy metal waste, complicating environmental compliance and increasing the burden on wastewater treatment facilities. The cumulative effect of these factors is a manufacturing process that, while scientifically valid, is suboptimal for high-volume, cost-sensitive generic drug production.
The Novel Approach
The methodology disclosed in CN1993369A fundamentally reengineers the synthesis by substituting Palladium with Nickel-based catalytic systems, specifically highlighting the efficacy of Nickel(II) chloride (NiCl2) and various phosphine-ligated Nickel complexes. This shift represents a paradigm change in process chemistry, enabling the coupling of readily available chlorinated precursors with lithiated alkyne equivalents under milder conditions. 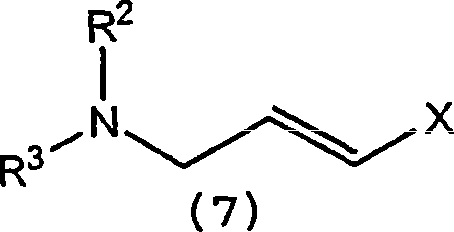
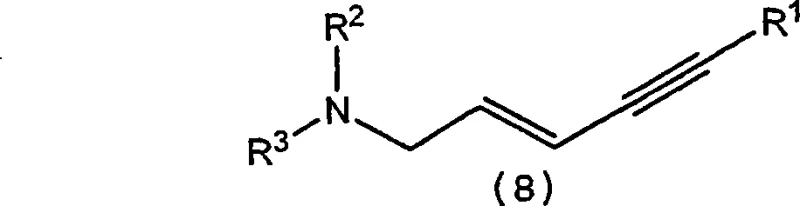 The new approach eliminates the dependency on tributylacetylene by generating the necessary alkyne functionality in situ from 1,1-dichloro-3,3-dimethylbutene, a far more economical starting material. This strategic substitution not only reduces raw material costs drastically but also simplifies the reaction workflow by integrating multiple synthetic steps into a streamlined sequence. The use of Nickel catalysts allows for shorter reaction times and lower operating temperatures compared to their Palladium counterparts, thereby enhancing energy efficiency and reactor throughput. Consequently, this novel route offers a compelling value proposition for manufacturers seeking to optimize their production lines for both economic performance and operational simplicity.
The new approach eliminates the dependency on tributylacetylene by generating the necessary alkyne functionality in situ from 1,1-dichloro-3,3-dimethylbutene, a far more economical starting material. This strategic substitution not only reduces raw material costs drastically but also simplifies the reaction workflow by integrating multiple synthetic steps into a streamlined sequence. The use of Nickel catalysts allows for shorter reaction times and lower operating temperatures compared to their Palladium counterparts, thereby enhancing energy efficiency and reactor throughput. Consequently, this novel route offers a compelling value proposition for manufacturers seeking to optimize their production lines for both economic performance and operational simplicity.
Mechanistic Insights into Ni(II)-Catalyzed Cross-Coupling
The core of this innovative synthesis lies in the Nickel-catalyzed coupling between a lithiated alkyne species and a chlorinated allylamine derivative. The mechanism initiates with the generation of the nucleophilic alkyne component, typically achieved by treating 1,1-dichloro-3,3-dimethylbutene with an organolithium reagent such as n-butyllithium in a non-polar solvent like toluene or heptane. This step requires precise temperature control, generally maintained between 80°C and 90°C, to ensure complete lithiation while minimizing side reactions. Once formed, the lithiated intermediate undergoes transmetallation with the Nickel(II) catalyst, forming an active organonickel species capable of oxidative addition into the carbon-chlorine bond of the amine precursor. 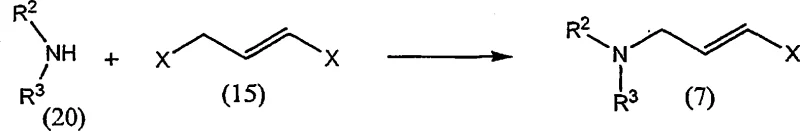 The subsequent reductive elimination step forge the critical carbon-carbon bond, yielding the Terbinafine backbone with high stereoselectivity for the E-isomer. A distinct advantage of the Nickel system is its tolerance to various functional groups and its ability to operate effectively in solvent mixtures such as THF and toluene, which facilitate the solubility of both organic substrates and inorganic salts. Furthermore, the patent details specific workup procedures involving aqueous ammonia or EDTA solutions, which are crucial for chelating and removing residual Nickel ions from the organic phase. This meticulous attention to metal scavenging ensures that the final product meets stringent limits for heavy metal impurities, a critical quality attribute for pharmaceutical intermediates intended for human consumption.
The subsequent reductive elimination step forge the critical carbon-carbon bond, yielding the Terbinafine backbone with high stereoselectivity for the E-isomer. A distinct advantage of the Nickel system is its tolerance to various functional groups and its ability to operate effectively in solvent mixtures such as THF and toluene, which facilitate the solubility of both organic substrates and inorganic salts. Furthermore, the patent details specific workup procedures involving aqueous ammonia or EDTA solutions, which are crucial for chelating and removing residual Nickel ions from the organic phase. This meticulous attention to metal scavenging ensures that the final product meets stringent limits for heavy metal impurities, a critical quality attribute for pharmaceutical intermediates intended for human consumption.
Impurity control is another pivotal aspect of this mechanistic pathway, particularly regarding the suppression of Z-isomer formation and dialkylated by-products. The choice of ligand and catalyst loading plays a significant role in directing the reaction trajectory towards the desired E-configuration. For instance, the use of NiCl2 in combination with triphenylphosphine has been shown to enhance selectivity, whereas unligated Nickel salts might require optimized stoichiometric ratios to prevent over-alkylation. The process also incorporates a protective group strategy in certain embodiments, where silyl groups are employed to mask the amine functionality during the coupling step, subsequently removed via hydrolysis. This modular approach allows for fine-tuning the reaction conditions to maximize yield and purity. By understanding these mechanistic details, process chemists can better troubleshoot potential deviations and implement robust control strategies that ensure consistent batch-to-batch quality, thereby mitigating the risk of costly production failures.
How to Synthesize Terbinafine Efficiently
Implementing this synthesis route requires a disciplined approach to reaction engineering and unit operations. The process begins with the careful preparation of the lithiated alkyne precursor under inert atmosphere, followed by the controlled addition of the Nickel catalyst and the amine electrophile. Detailed standard operating procedures (SOPs) are essential to manage the exothermic nature of the lithiation and coupling steps.
- Preparation of the alkyne precursor via lithiation of 1,1-dichloro-3,3-dimethylbutene using n-butyllithium in toluene/heptane.
- In situ coupling of the lithiated species with N-(trans-3-chloro-2-propenyl)-N-methyl-1-naphthylmethylamine using NiCl2 catalyst.
- Workup involving aqueous ammonia or EDTA washes to remove nickel residues, followed by crystallization of the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Nickel-catalyzed synthesis route described in CN1993369A presents a multitude of strategic benefits that extend beyond simple chemical efficiency. The most immediate impact is observed in the reduction of raw material expenditures, driven by the substitution of high-value Palladium catalysts with commodity-grade Nickel salts. This shift decouples production costs from the volatile pricing of precious metals, providing greater financial predictability and margin stability. Moreover, the elimination of tributylacetylene from the bill of materials removes a significant cost driver and sourcing bottleneck, as the new precursors are derived from widely available petrochemical feedstocks. These changes collectively contribute to a leaner, more resilient supply chain capable of withstanding market disruptions. The simplified workflow also translates into reduced operational complexity, lowering the barrier for technology transfer and scale-up across different manufacturing sites.
- Cost Reduction in Manufacturing: The transition from Palladium to Nickel catalysts results in a drastic decrease in catalyst procurement costs, as Nickel salts are orders of magnitude cheaper than Palladium complexes. Additionally, the use of inexpensive starting materials like 1,1-dichloro-3,3-dimethylbutene replaces costly specialty reagents, further driving down the variable cost per kilogram. The process efficiency is enhanced by shorter reaction times and milder conditions, which reduce energy consumption and utility costs associated with heating and cooling cycles. By minimizing the number of isolation and purification steps, particularly through one-pot methodologies, labor costs and solvent usage are also significantly curtailed. These cumulative savings create a highly competitive cost structure that allows for aggressive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved by relying on commoditized chemicals rather than specialized, low-volume reagents. Nickel chloride and common organic solvents like toluene and THF are produced at massive scales globally, ensuring consistent availability and short lead times. This contrasts sharply with the supply risks associated with proprietary Palladium ligands or unstable alkynes, which often suffer from long delivery windows and quality variability. The robustness of the Nickel-catalyzed process also means that production schedules are less prone to delays caused by reagent shortages or specification failures. Consequently, manufacturers can maintain higher inventory turnover rates and respond more agilely to fluctuations in market demand, securing their position as a dependable partner for downstream API formulators.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers distinct advantages by reducing the load of heavy metal waste. Nickel, while still requiring proper disposal, is less toxic and easier to manage than Palladium in many waste streams, simplifying effluent treatment protocols. The ability to run reactions at lower temperatures and pressures enhances process safety, reducing the risk of thermal runaways and facilitating safer scale-up from pilot to commercial plants. The high atom economy of the coupling reaction minimizes the generation of organic by-products, aligning with green chemistry principles and reducing the carbon footprint of the manufacturing operation. These factors make the technology highly attractive for facilities aiming to meet increasingly stringent environmental regulations while expanding production capacity to meet global healthcare needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their own production portfolios.
Q: Why is Nickel preferred over Palladium for Terbinafine synthesis?
A: Nickel salts (like NiCl2) are significantly more economical than Palladium complexes (like Pd(PPh3)4) and allow for milder reaction conditions while maintaining high yields, drastically reducing raw material costs.
Q: What is the expected purity of the final Terbinafine hydrochloride?
A: According to patent data, the process can achieve purity levels exceeding 99% (HPLC A%) after crystallization, meeting stringent pharmaceutical standards.
Q: Does this process avoid expensive tributylacetylene?
A: Yes, the novel route utilizes 1,1-dichloro-3,3-dimethylbutene as a cheaper starting material, which is converted in situ, eliminating the need for costly pre-formed tributylacetylene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbinafine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to drive value in the pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the Nickel-catalyzed Terbinafine synthesis are translated into reality with precision and efficiency. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex organometallic chemistry, including the safe handling of pyrophoric reagents and sensitive catalysts, positions us as a premier partner for companies seeking to optimize their antifungal API supply.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this Nickel-based methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your competitive edge in the global market. Together, we can build a more sustainable and efficient supply chain for essential medicines.
