Advanced Manufacturing of Lobaplatin: Streamlined Synthesis for Commercial Scale-up
Advanced Manufacturing of Lobaplatin: Streamlined Synthesis for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing pathways for third-generation platinum antineoplastic agents, specifically targeting the production of Lobaplatin (D19466). Patent CN103467528A introduces a transformative preparation method for Lobaplatin and its stable trihydrate form, addressing critical limitations found in earlier synthetic routes. This technology leverages a direct aqueous reaction between trans-diaminomethylcyclobutane and chloroplatinite salts under an inert atmosphere, bypassing the cumbersome ion-exchange resin steps typical of legacy processes. By shifting the crystallization endpoint to the trihydrate form rather than the hygroscopic anhydrous variant, this method ensures superior product stability and formulation compatibility. For global procurement teams, this represents a significant opportunity for cost reduction in pharmaceutical manufacturing, as the simplified workflow reduces both operational time and the consumption of specialized reagents. The following analysis details the technical superiority and commercial viability of this approach for large-scale API production.
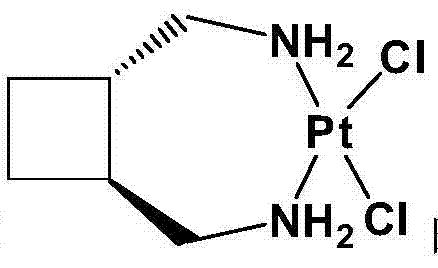
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lobaplatin has been plagued by complex purification requirements and unstable product forms that hinder efficient commercialization. Prior art, such as US5023335, describes a multi-step sequence involving the reaction of the diamine ligand with potassium chloroplatinite, followed by a silver nitrate displacement and a prolonged three-day reaction with L-lactic acid. Crucially, these older methods necessitate the use of alkaline anion exchange columns to purify the intermediate, a step that introduces significant variability and potential contamination risks if the resin is not perfectly regenerated. Furthermore, the final product obtained via these traditional routes is often the anhydrous form of Lobaplatin, which is notoriously hygroscopic, sticky, and difficult to handle during downstream tablet or injectable formulation. Alternative methods, like those disclosed in CN102020679A, attempt to use iodide intermediates but result in excessively long reaction times, high costs due to expensive potassium iodide, and increased impurity profiles that demand rigorous and costly purification protocols.
The Novel Approach
The methodology outlined in CN103467528A fundamentally reengineers the synthesis to prioritize efficiency and product stability. By utilizing water as the primary solvent and conducting the initial coordination under strict inert gas protection, the process generates the dichloro-platinum intermediate with exceptional purity, often exceeding 98% without the need for further refining. The elimination of the anion exchange resin step is a major breakthrough, as it removes a bottleneck that previously extended production cycles and introduced non-metallic impurities. Instead, the process employs a direct silver nitrate replacement in a water-acetone system, followed by immediate coupling with L-lactic acid at a controlled pH range of 5.0 to 8.0. This streamlined approach not only shortens the overall production timeline but also directs the crystallization towards the thermodynamically stable trihydrate form. This shift ensures that the final API is non-hygroscopic and possesses consistent purity, directly addressing the formulation challenges faced by pharmaceutical developers using earlier synthetic generations.
Mechanistic Insights into Platinum Coordination and Ligand Exchange
The core of this synthetic innovation lies in the precise control of the platinum coordination environment during the formation of the key intermediate. The reaction initiates with the dissolution of a chloroplatinite salt, preferably potassium chloroplatinite (K2PtCl4), in water, followed by the addition of trans-diaminomethylcyclobutane. Maintaining an inert atmosphere, typically nitrogen, is critical here to prevent the oxidation of the platinum center or the degradation of the amine ligand, which could lead to colored impurities. The reaction proceeds at mild temperatures between 20°C and 30°C over 8 to 16 hours, allowing for the slow and ordered formation of the dichloro-1,2-diaminomethylcyclobutane platinum complex. This careful control results in yields exceeding 95%, significantly higher than the variable outputs of unprotected reactions. The structural integrity of this intermediate is paramount, as it serves as the scaffold for the subsequent lactate binding.
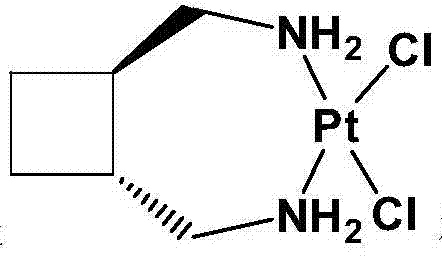
Following the isolation of the dichloro-intermediate, the mechanism shifts to a halide exchange driven by silver chemistry. The addition of silver nitrate in a water-acetone mixture facilitates the precipitation of silver chloride, effectively stripping the chloride ligands from the platinum center to create a reactive aquo-complex in situ. Unlike previous methods that relied on resin columns to capture these chlorides, this precipitation method is quantitative and easily managed via filtration. The resulting filtrate, containing the activated platinum species, is then reacted with L-lactic acid or its sodium salt. The pH is meticulously adjusted to between 5.5 and 6.5 using alkali metal hydroxides, a condition that favors the chelation of the lactate oxygen atoms to the platinum without causing hydrolysis of the amine ligands. This direct coupling mechanism avoids the prolonged reaction times of prior art, completing the coordination in 8 to 12 hours at 40°C to 60°C, thereby minimizing thermal degradation and ensuring a clean impurity profile suitable for oncology applications.
How to Synthesize Lobaplatin Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent ratios, temperature gradients, and light protection to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory validation to multi-ton commercial production without the need for specialized chromatography equipment. Operators must ensure that the initial chloroplatinite solution is fully clarified before ligand addition and that all subsequent steps involving silver salts are performed in the dark to prevent photo-decomposition of the sensitive platinum complexes. The detailed standardized synthesis steps, including specific mass ratios and cooling profiles for crystallization, are outlined below to guide process engineers in replicating this high-efficiency route.
- React potassium chloroplatinite with trans-diaminomethylcyclobutane in water under inert gas to form the dichloro-platinum intermediate.
- Perform a replacement reaction with silver nitrate in water/acetone to remove chloride ions as silver chloride precipitate.
- React the filtrate directly with L-lactic acid at pH 5.0-8.0 and recrystallize from water/acetone to obtain Lobaplatin trihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-protected methodology offers tangible strategic benefits beyond mere technical elegance. The most significant advantage is the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply reliability. By removing the dependency on ion-exchange resins, manufacturers eliminate the costs associated with resin procurement, column packing, and the extensive validation required for resin regeneration cycles. Furthermore, the substitution of expensive reagents like potassium iodide with common, commodity-grade chemicals such as silver nitrate and acetone stabilizes the raw material cost base, shielding the supply chain from volatility in specialty chemical markets. This robustness ensures a more predictable pricing structure for long-term API contracts.
- Cost Reduction in Manufacturing: The elimination of the anion exchange resin step provides a substantial reduction in processing costs by removing a labor-intensive and time-consuming unit operation. Traditional methods require significant downtime for column conditioning and washing, whereas this precipitation-based purification allows for continuous or semi-continuous processing. Additionally, the use of water as the primary solvent for the initial coordination step minimizes the volume of organic solvents required, leading to lower waste disposal costs and reduced expenditure on solvent recovery systems. The high yield of the intermediate (>95%) ensures that expensive platinum starting materials are utilized with maximum efficiency, reducing the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available raw materials such as potassium chloroplatinite, silver nitrate, and L-lactic acid mitigates the risk of supply disruptions often associated with exotic or highly specialized reagents. The simplified process flow, characterized by fewer unit operations and shorter reaction times, inherently increases the throughput capacity of existing manufacturing facilities. This agility allows suppliers to respond more rapidly to fluctuations in market demand, ensuring consistent delivery schedules for downstream pharmaceutical partners. The stability of the trihydrate form also extends the shelf-life of the inventory, reducing the risk of product spoilage during storage and transit.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles by prioritizing aqueous media and minimizing the use of volatile organic compounds. The absence of resin waste streams simplifies effluent treatment, as there are no spent resin columns to dispose of as hazardous solid waste. The crystallization process using water and acetone is easily scalable, as the solubility profiles are well-understood and manageable in large reactors. This ease of scale-up facilitates the transition from pilot plant batches to commercial production volumes of 100 kgs to 100 MT annually, ensuring that the supply can meet the demands of global oncology markets without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lobaplatin synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, focusing on the practical implications for quality control and process engineering. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing supplier capabilities.
Q: Why is Lobaplatin Trihydrate preferred over the anhydrous form?
A: The anhydrous form of Lobaplatin is prone to deliquescence and becomes sticky, making it difficult to formulate into stable pharmaceutical preparations. The trihydrate form offers superior physical stability, better reproducibility in purity and content, and improved handling characteristics for industrial manufacturing.
Q: How does this process eliminate the need for ion-exchange resins?
A: Traditional methods require passing the reaction mixture through alkaline anion exchange columns to remove chloride ions, which adds complexity and risk of impurity introduction from incomplete resin regeneration. This novel method utilizes a direct silver nitrate replacement followed by precise pH control during the lactic acid coupling, rendering the resin step unnecessary.
Q: What are the yield advantages of this specific synthetic route?
A: By optimizing the formation of the dichloro-intermediate under inert conditions and avoiding the losses associated with column chromatography or resin exchange, the process achieves a dichloro-intermediate yield of over 95% and a final Lobaplatin trihydrate yield exceeding 40% from the intermediate stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lobaplatin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates and APIs in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated coordination chemistry required for Lobaplatin is executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the structural integrity and impurity profiles of complex platinum compounds, guaranteeing that every batch meets the exacting standards required for clinical and commercial use. Our commitment to quality assurance means that clients can rely on us for a steady supply of material that adheres to the most advanced synthetic protocols available today.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this resin-free manufacturing method. We encourage you to contact us directly to obtain specific COA data for our Lobaplatin batches and to receive detailed route feasibility assessments tailored to your project timelines. Let us collaborate to bring stable, high-quality Lobaplatin to the market more efficiently.
