Advanced Manufacturing of Atorvastatin Intermediates via Safe Organocatalytic Routes
Advanced Manufacturing of Atorvastatin Intermediates via Safe Organocatalytic Routes
The pharmaceutical industry continuously seeks robust and safe methodologies for producing high-value statin intermediates, particularly for blockbuster drugs like Atorvastatin. Patent CN102827030A introduces a groundbreaking preparation method for (3R,5R)-3,5-dihydroxy-6-cyanohexanoate, a pivotal chiral building block in the synthesis of lipid-lowering agents. This innovation addresses critical pain points in traditional manufacturing by replacing hazardous reagents with safer alternatives while maintaining exceptional stereocontrol. The disclosed route leverages asymmetric organocatalysis to achieve high yields and purity, positioning it as a superior choice for reliable pharmaceutical intermediates supplier networks aiming to optimize their production portfolios. By eliminating the need for toxic cyanide salts typically used in ring-opening procedures, this technology not only enhances worker safety but also simplifies waste management protocols, making it an attractive solution for modern green chemistry initiatives in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral dihydroxy nitrile intermediates has relied heavily on pathways involving chiral epoxy butyrate esters as starting materials. These conventional routes, such as those described in U.S. Pat 5155251A and various World Patent publications, necessitate the use of metal cyanides like sodium cyanide or potassium cyanide for ring-opening reactions. The reliance on these hypertoxic prussiate salts presents severe challenges for industrial scale-up, including stringent equipment requirements, complex labor protection measures, and difficult wastewater treatment processes. Furthermore, enzymatic approaches, while selective, often suffer from the difficulty in obtaining stable and cost-effective aldolase enzymes, limiting their commercial viability. The cumulative effect of these drawbacks results in higher operational costs, increased regulatory scrutiny, and potential supply chain disruptions due to the restricted handling of hazardous chemicals, creating a compelling need for safer synthetic alternatives in the competitive API intermediate market.
The Novel Approach
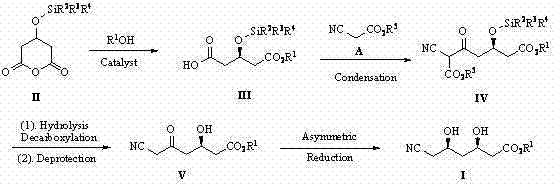
The methodology outlined in patent CN102827030A offers a transformative solution by utilizing 3-silyloxycyclopentanoic anhydride as the primary starting material. This novel route bypasses the dangerous cyanide ring-opening step entirely, instead employing a condensation reaction with cyanoacetate to introduce the nitrile functionality safely. The process is characterized by mild reaction conditions, typically ranging from -78°C to 100°C, and utilizes readily available organic solvents such as MTBE, THF, and toluene. Crucially, the chiral catalysts used, which are quinine-derived thiourea or sulfonamide derivatives, can be recovered quantitatively, significantly reducing raw material costs. This approach not only streamlines the synthetic sequence but also ensures high stereoselectivity and yield, providing a commercially viable pathway that aligns with modern environmental standards and cost reduction in pharmaceutical intermediates manufacturing goals.
Mechanistic Insights into Quinine-Derived Organocatalytic Alcoholysis
The cornerstone of this synthetic strategy lies in the initial asymmetric catalytic alcoholysis of the cyclic anhydride. In this step, a chiral quinine-derived organocatalyst, such as a thiourea or sulfonamide derivative, activates the anhydride substrate through hydrogen bonding interactions. This activation facilitates the nucleophilic attack by the alcohol (R1OH), leading to the regioselective ring opening of the anhydride. The chiral environment provided by the cinchona alkaloid backbone ensures that the reaction proceeds with high enantioselectivity, yielding the (R)-3-silyloxy-5-alkoxy-5-oxopentanoic acid intermediate with excellent optical purity. This organocatalytic approach is superior to traditional metal-catalyzed methods as it avoids heavy metal contamination, simplifying downstream purification and ensuring the high-purity pharmaceutical intermediates required for subsequent steps in the drug synthesis value chain.
Following the establishment of the first chiral center, the synthesis proceeds through a condensation reaction with cyanoacetate, followed by decarboxylation and desilylation to generate the keto-nitrile intermediate. The final stereochemical outcome is determined during the asymmetric reduction of the ketone moiety. Using reagents like diethylmethoxyborane or triethylborane in combination with a reducing agent such as sodium borohydride or lithium borohydride, the ketone is reduced to the secondary alcohol with precise stereocontrol. This step converts the (R)-3-hydroxy-5-oxo-6-cyanocaproic acid ester into the final (3R,5R)-diol product. The mechanism relies on the formation of a chelated transition state where the boron species coordinates with the carbonyl oxygen and the adjacent hydroxyl group, directing the hydride delivery to the desired face of the molecule, thereby ensuring the formation of the syn-diol configuration essential for biological activity.
How to Synthesize (3R,5R)-3,5-dihydroxy-6-cyanohexanoate Efficiently
The synthesis of this critical statin intermediate involves a carefully orchestrated four-step sequence that balances reactivity with stereochemical fidelity. The process begins with the asymmetric alcoholysis of the anhydride precursor, followed by chain extension via condensation with cyanoacetate. Subsequent removal of the protecting groups and decarboxylation yields the key keto-nitrile scaffold, which is finally subjected to asymmetric reduction to install the second hydroxyl group. Each step has been optimized in the patent examples to demonstrate robustness across different solvent systems and temperatures, ensuring that the route is adaptable to various manufacturing scales. For detailed operational parameters, stoichiometry, and workup procedures necessary for replicating this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Perform asymmetric catalytic alcoholysis of 3-silyloxycyclopentanoic anhydride using a quinine-derived organocatalyst to form the chiral mono-ester.
- Conduct a condensation reaction between the chiral mono-ester and cyanoacetate under alkaline conditions to introduce the nitrile group and extend the carbon chain.
- Execute decarboxylation and desilylation sequentially or in one pot to yield the keto-nitrile intermediate, followed by asymmetric reduction to establish the final diol stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical feasibility. By eliminating the requirement for highly toxic cyanide salts, the process drastically reduces the costs associated with specialized safety infrastructure, hazardous waste disposal, and regulatory compliance monitoring. The use of inexpensive and readily available starting materials, such as cyanoacetate and simple alcohols, coupled with the quantitative recovery of the chiral organocatalyst, leads to significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the mild reaction conditions minimize energy consumption and equipment wear, contributing to a more sustainable and economically efficient production model that enhances overall margin potential for generic drug manufacturers seeking to optimize their bill of materials.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. Traditional methods often incur high costs due to the need for rigorous containment systems when handling metal cyanides, as well as the expense of chiral epoxy starting materials. In contrast, this method utilizes cheap bulk chemicals and recoverable organocatalysts, which lowers the direct material cost per kilogram. Additionally, the simplified purification processes resulting from the absence of heavy metal residues reduce the consumption of chromatography media and solvents, further driving down the operational expenditure and improving the overall cost competitiveness of the final API.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly improved by relying on raw materials that are commoditized and widely available in the global chemical market. Unlike specialized enzymes or sensitive chiral epoxides that may have limited suppliers and long lead times, the reagents used in this process, such as cyanoacetate and silyl protecting groups, are produced by multiple vendors worldwide. This diversification of the supply base mitigates the risk of shortages and price volatility. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies or environmental controls, ensuring consistent output and reducing lead time for high-purity pharmaceutical intermediates deliveries to downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates. The avoidance of toxic cyanides simplifies the environmental impact assessment and permits acquisition process, accelerating the timeline from pilot plant to full-scale production. The waste streams generated are less hazardous and easier to treat, aligning with increasingly strict global environmental regulations. This compliance advantage reduces the risk of production shutdowns due to regulatory violations and enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for multinational pharmaceutical companies with stringent sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement specialists assessing the long-term viability of the supply source.
Q: How does this patent method improve safety compared to traditional Atorvastatin intermediate synthesis?
A: Traditional routes often rely on highly toxic metal cyanides (such as sodium cyanide or potassium cyanide) for ring-opening reactions. This patented method utilizes cyanoacetate condensation instead, completely avoiding the handling and disposal hazards associated with hypertoxic prussiate salts, thereby significantly enhancing operational safety and environmental compliance.
Q: What type of catalyst is used to ensure high stereoselectivity in the first step?
A: The process employs chiral organocatalysts derived from quinine, specifically quinine thiourea or quinine sulfonamide derivatives. These catalysts facilitate the asymmetric alcoholysis of the cyclic anhydride with high enantioselectivity, establishing the critical chiral center early in the synthesis without requiring expensive transition metals.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the suitability for industrial production due to mild reaction conditions ranging from -78°C to 100°C, the use of common organic solvents like MTBE and THF, and the ability to recover the chiral catalyst quantitatively. These factors contribute to a robust and scalable manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R,5R)-3,5-dihydroxy-6-cyanohexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key statin intermediates for the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of major drug manufacturers. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. By leveraging our expertise in process optimization and quality assurance, we provide a dependable partnership that supports your drug development timelines and commercial launch goals with unwavering consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project needs. Whether you require a Customized Cost-Saving Analysis comparing this organocatalytic method to your current supply chain or need to review specific COA data and route feasibility assessments, our experts are ready to assist. Contact us today to request samples and detailed technical dossiers, and let us demonstrate how our commitment to excellence in fine chemical manufacturing can drive value and efficiency for your organization.
