Advanced Catalytic Strategy for Commercial Scale-Up of High-Purity Tiotropium Bromide
Advanced Catalytic Strategy for Commercial Scale-Up of High-Purity Tiotropium Bromide
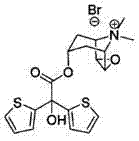
The pharmaceutical landscape for respiratory therapies demands uncompromising quality standards, particularly for long-acting muscarinic antagonists like Tiotropium Bromide. Patent CN101979391B introduces a transformative methodology that addresses the persistent challenge of stereoisomer control during the synthesis of this critical active pharmaceutical ingredient. By implementing a novel mixed catalyst system comprising sodium and sodium methoxide, this technology ensures that the content of the undesirable 2-(2-thienyl) oxyacetic acid scopine ester isomer remains strictly below 0.1%, fully complying with the rigorous tentative standards of the European Pharmacopoeia. This breakthrough represents a significant leap forward for manufacturers seeking to eliminate batch failures associated with high isomer content, thereby securing a more reliable supply chain for high-purity Tiotropium Bromide. The structural integrity of the final molecule, as depicted below, relies heavily on the precision of the initial esterification steps.
Furthermore, the integration of this patented process allows for the utilization of cost-effective starting materials such as scopolamine hydrobromide, which can be efficiently converted into the necessary scopine intermediate. The robustness of this synthetic route is evidenced by its ability to maintain high yields while simultaneously achieving superior purity profiles, a dual objective that is often difficult to balance in complex organic synthesis. For global procurement teams, this translates to a more stable sourcing strategy where the risk of regulatory non-compliance is drastically minimized through inherent process design rather than reliance on extensive downstream purification. As we delve deeper into the technical specifics, it becomes clear that this method offers a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates required for next-generation COPD treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Tiotropium Bromide have historically struggled with the precise control of stereochemistry during the formation of the ester linkage between the scopine moiety and the dithienylglycolic acid derivative. In many established processes, the content of the unwanted isomer frequently exceeds 0.3%, a level that fails to meet the stringent requirements set forth by the European Pharmacopoeia for clinical-grade material. This deficiency forces manufacturers to either accept significant yield losses during aggressive purification attempts or, in worse scenarios, discard entire batches that cannot be brought into specification, leading to substantial economic inefficiencies. The reliance on single-catalyst systems or less optimized reaction conditions often results in a lack of selectivity, allowing competing reaction pathways to generate impurities that are structurally similar and difficult to separate. Consequently, the supply of reliable pharmaceutical intermediates has been periodically disrupted by these quality consistency issues, creating bottlenecks for downstream API production facilities.
The Novel Approach
The innovative approach detailed in the patent data overcomes these historical limitations by introducing a synergistic catalytic environment that fundamentally alters the reaction kinetics in favor of the desired stereoisomer. By employing a mixed catalyst system of metallic sodium and sodium methoxide within a xylene solvent matrix, the process achieves a level of stereocontrol that keeps isomer content consistently between 0.05% and 0.08%, well within the safety margin of regulatory standards. This method not only resolves the critical quality attribute of isomer purity but also streamlines the workflow by reducing the need for complex chromatographic separations that are often costly and time-consuming at scale. The reaction proceeds under moderate thermal conditions, typically between 50°C and 60°C, which enhances operational safety and reduces energy consumption compared to more vigorous traditional methods. This strategic optimization ensures cost reduction in API manufacturing by maximizing the utility of raw materials and minimizing waste generation throughout the synthesis lifecycle.
Mechanistic Insights into Mixed Catalyst Transesterification
The core of this technological advancement lies in the mechanistic behavior of the sodium and sodium methoxide dual-catalyst system during the transesterification reaction. When scopine reacts with methyl 2,2-dithienyl glycolate, the presence of both catalysts creates a highly specific basic environment that likely stabilizes the transition state leading to the desired 6-beta, 7-beta epoxy configuration. The metallic sodium may serve to activate the alcohol group of the scopine more effectively, while the sodium methoxide facilitates the nucleophilic attack on the ester carbonyl, ensuring that the reaction proceeds with high regioselectivity and stereoselectivity. This cooperative catalysis prevents the racemization or epimerization that typically plagues single-catalyst systems, thereby locking the molecular architecture into the therapeutically active conformation required for potent M3 receptor antagonism. Understanding this mechanism is crucial for R&D directors aiming to replicate these results, as the precise mass ratio of the catalysts, preferably around 1:3, is a critical parameter that dictates the final impurity profile.
Impurity control is further enhanced by the specific workup procedures designed to complement the catalytic system, ensuring that any trace byproducts are effectively removed before the final quaternization step. After the transesterification, the reaction mixture is carefully adjusted to an acidic pH of 1-3 using dilute hydrochloric acid, followed by washing with cold water to remove inorganic salts and polar impurities. Subsequent basification to pH 9-10 allows for the selective extraction of the organic product into chloroform, leaving behind water-soluble contaminants. This multi-stage purification strategy, combined with the inherent selectivity of the reaction, results in an intermediate ester of exceptional purity, which is essential for the subsequent methylation step. By rigorously controlling the quality of this key intermediate, the overall process guarantees that the final Tiotropium Bromide API meets the rigorous specifications demanded by global health authorities, minimizing the risk of toxicological issues associated with unknown impurities.
How to Synthesize Tiotropium Bromide Efficiently
The synthesis of Tiotropium Bromide via this patented route involves a sequence of highly controlled chemical transformations that begin with the preparation of the scopine intermediate from commercially available scopolamine hydrobromide. This initial reduction step utilizes sodium borohydride in anhydrous methanol, followed by careful pH manipulation and solvent extraction to isolate the free base, setting the stage for the critical esterification reaction. The subsequent transesterification in xylene with the mixed catalyst system is the pivotal operation where the majority of value is added, requiring precise temperature monitoring and stoichiometric control to achieve the target isomer specifications. Following the isolation of the pure ester intermediate, the final quaternization with methyl bromide and recrystallization from an acetonitrile-methanol mixture yields the final API with high crystallinity and stability. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Prepare Scopine via reduction of Scopolamine Hydrobromide using sodium borohydride in methanol, followed by pH adjustment and extraction.
- Conduct transesterification between Scopine and Methyl 2,2-dithienyl glycolate in xylene using a mixed catalyst of Sodium and Sodium Methoxide at 50-60°C.
- Quaternize the resulting ester intermediate with methyl bromide, followed by recrystallization from an acetonitrile and methanol mixture to obtain pure Tiotropium Bromide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers profound advantages in terms of cost stability and supply continuity, driven by the elimination of yield-killing impurities. The ability to consistently produce material that meets European Pharmacopoeia standards without extensive reprocessing means that production schedules are more predictable, reducing the risk of stockouts that can plague the respiratory therapy market. By utilizing a catalyst system that is both effective and relatively inexpensive, the overall cost of goods sold is optimized, allowing for more competitive pricing strategies in the generic API market. Furthermore, the use of common solvents like xylene and methanol simplifies the logistics of raw material sourcing, as these chemicals are widely available and do not suffer from the supply constraints often associated with exotic reagents. This reliability makes the manufacturer a preferred partner for long-term contracts where consistent quality and delivery are paramount.
- Cost Reduction in Manufacturing: The implementation of the mixed catalyst system significantly reduces the financial burden associated with batch failures and low yields caused by high isomer content. By ensuring that the isomer levels remain below 0.1% directly from the reaction, the need for expensive and yield-losing purification steps such as preparative HPLC is eliminated, leading to substantial cost savings. Additionally, the high conversion efficiency of the transesterification step maximizes the output per unit of raw material input, further driving down the variable costs of production. This economic efficiency is compounded by the reduced consumption of solvents and energy, as the reaction operates at moderate temperatures and requires fewer processing cycles to achieve the desired purity profile.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain reliability by minimizing the variability in production outcomes, ensuring that every batch meets the strict quality criteria required for regulatory filing. The use of stable and readily available starting materials, such as scopolamine hydrobromide and methyl 2,2-dithienyl glycolate, mitigates the risk of supply disruptions caused by raw material shortages. Moreover, the scalability of the process allows for seamless transitions from pilot plant operations to full commercial production, ensuring that supply can be ramped up quickly to meet market demand without compromising on quality. This dependability is crucial for maintaining the trust of downstream partners who rely on a steady flow of high-quality intermediates for their own formulation processes.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is designed to be industrially friendly, utilizing solvents that can be easily recovered and recycled, thereby reducing the overall environmental footprint of the manufacturing operation. The avoidance of heavy metal catalysts or hazardous reagents simplifies waste treatment protocols and ensures compliance with increasingly stringent environmental regulations. The reaction conditions are mild enough to be safely managed in large-scale reactors, reducing the risk of thermal runaways or other safety incidents that could halt production. This combination of safety, scalability, and environmental stewardship makes the process highly attractive for modern pharmaceutical manufacturing facilities aiming to achieve sustainable production goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Tiotropium Bromide. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing the quality credentials of potential suppliers. The focus is on providing transparent, data-backed information to facilitate informed decision-making.
Q: How does the mixed catalyst system improve isomer control in Tiotropium Bromide synthesis?
A: The use of a simultaneous sodium and sodium methoxide catalyst system during the transesterification step creates a specific reaction environment that strictly limits the formation of unwanted stereoisomers, keeping content below 0.1% to meet European Pharmacopoeia standards.
Q: What are the critical purification steps for ensuring API grade quality?
A: Critical steps include washing the reaction mixture with cold water after acidification, followed by extraction and a final recrystallization process using a specific volume ratio of acetonitrile and methanol to remove residual impurities and solvents.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes common organic solvents like xylene and operates at moderate temperatures (50-60°C), making it safe, scalable, and economically viable for commercial manufacturing without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tiotropium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity Tiotropium Bromide that adheres to the most stringent international standards, leveraging advanced catalytic technologies like the one described in CN101979391B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical companies without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of our respiratory API intermediates meets the required isomer limits and impurity profiles. Our commitment to technical excellence ensures that our clients receive a product that is ready for formulation, minimizing their development timelines and regulatory risks.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement costs while enhancing supply security. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your production needs. Let us collaborate to bring high-quality respiratory therapies to patients worldwide through efficient and reliable chemical manufacturing.
