Advanced Synthesis of High-Optical-Purity Tenofovir Alafenamide Intermediates for Commercial Scale-Up
The pharmaceutical landscape for chronic hepatitis B and HIV treatment has been significantly transformed by the advent of nucleotide analog prodrugs, specifically Tenofovir Alafenamide. As the industry demands increasingly efficient and environmentally sustainable manufacturing pathways, patent CN111116655B emerges as a critical technological breakthrough. This patent details a novel preparation method for high-optical-purity tenofovir benzyl ester phosphonamide prodrug intermediates, addressing long-standing inefficiencies in amidation and chiral resolution. By shifting away from toxic solvents and cumbersome purification techniques, this methodology offers a robust framework for the reliable pharmaceutical intermediates supplier seeking to optimize their production lines. The core innovation lies in the strategic application of the Carter condensing agent, which facilitates a cleaner reaction profile and superior yield metrics compared to legacy phosphoramidate synthesis routes. For R&D Directors and Supply Chain Heads, understanding the nuances of this patent is essential for evaluating potential technology transfers or licensing opportunities that could redefine cost structures and supply reliability in the antiviral sector.
The significance of this technology extends beyond mere chemical curiosity; it represents a tangible solution to the scalability bottlenecks that often plague the commercial scale-up of complex pharmaceutical intermediates. Traditional methods often rely on hazardous reagents and energy-intensive purification steps that inflate operational expenditures and complicate regulatory compliance. In contrast, the approach outlined in CN111116655B prioritizes process safety and environmental compatibility without compromising on the stringent purity specifications required for active pharmaceutical ingredients. This alignment with green chemistry principles ensures that manufacturers can meet evolving global regulatory standards while maintaining competitive pricing structures. As we delve deeper into the technical specifics, it becomes evident that this patent provides a comprehensive roadmap for producing high-purity OLED material grade intermediates, although its primary application remains firmly rooted in the life sciences sector where optical purity is non-negotiable for therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tenofovir phosphonamide intermediates has been hindered by the reliance on triphenylphosphine and 2,2-dithiodipyridine as coupling reagents, a combination that necessitates the use of pyridine as a solvent. Pyridine is notorious for its high toxicity, unpleasant odor, and significant environmental impact, posing severe challenges for worker safety and waste management in large-scale facilities. Furthermore, the conventional process typically yields Compound III at rates around 64%, with purity levels hovering near 93%, which often necessitates additional purification steps such as column chromatography. These chromatographic processes are not only solvent-intensive but also difficult to scale, leading to substantial increases in production time and cost. The energy consumption associated with distilling and concentrating large volumes of elution solvents further exacerbates the carbon footprint of the manufacturing process, making it less attractive for modern, sustainability-focused chemical enterprises. Consequently, the industry has long sought an alternative that could bypass these inefficiencies while delivering superior product quality.
The Novel Approach
The methodology presented in patent CN111116655B fundamentally reengineers the synthesis pathway by introducing the Carter condensing agent, also known as BOP, as the primary coupling reagent. This strategic substitution allows for the elimination of pyridine, replacing it with more benign solvents such as dichloromethane, ethyl acetate, or acetonitrile, which are easier to recover and recycle. The new approach achieves yields for Compound III exceeding 80%, with purity levels consistently above 95%, effectively rendering column chromatography obsolete. Instead, the process employs a sophisticated recrystallization technique using organic acids, which simplifies the post-reaction workup and significantly reduces solvent usage. This streamlined workflow not only enhances the overall throughput of the manufacturing line but also drastically lowers the operational costs associated with waste disposal and energy consumption. For procurement managers, this translates into a more stable and cost-effective supply chain for high-purity pharmaceutical intermediates, ensuring that production targets can be met without the volatility associated with complex purification bottlenecks.
Mechanistic Insights into Carter Condensing Agent Amidation
The chemical elegance of this patent lies in the mechanism by which the Carter condensing agent activates the phosphonic acid moiety for nucleophilic attack by the amine. Unlike traditional activation methods that may generate significant amounts of phosphine oxide byproducts which are difficult to remove, the Carter reagent facilitates a cleaner conversion through the formation of a highly reactive phosphonium intermediate. This intermediate reacts efficiently with L-alanine isopropyl ester hydrochloride under mild alkaline conditions, typically utilizing bases like triethylamine or diisopropylethylamine. The reaction proceeds smoothly at temperatures ranging from 20°C to 60°C, minimizing the risk of thermal degradation of the sensitive nucleotide scaffold. The use of nitrogen protection throughout the process ensures that moisture-sensitive reagents remain stable, further contributing to the high reproducibility of the reaction. By optimizing the molar ratios of the reactants, specifically maintaining a slight excess of the amine and coupling agent, the process drives the equilibrium towards the desired product, thereby maximizing yield and minimizing the formation of unreacted starting materials that could complicate downstream purification.
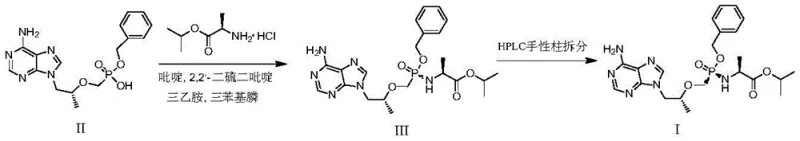
Following the initial amidation, the control of impurities becomes paramount, particularly when aiming for the high optical purity required for the final prodrug. The patent describes a meticulous recrystallization process where the crude Compound III is dissolved in isopropanol and treated with organic acids such as malonic, fumaric, or succinic acid. This step induces the formation of a salt that crystallizes out of the solution, leaving behind soluble impurities in the mother liquor. The choice of organic acid is critical, as it influences the crystal lattice structure and the efficiency of impurity exclusion. Subsequent neutralization with sodium carbonate and extraction with dichloromethane allows for the isolation of the free base form of Compound III with exceptional purity. This method of purification is far superior to chromatographic techniques in terms of scalability and cost, as it relies on standard unit operations that are easily implemented in multi-purpose chemical reactors. The ability to achieve purity levels above 98% through crystallization alone is a testament to the robustness of this chemical design, providing a solid foundation for the subsequent chiral resolution step.
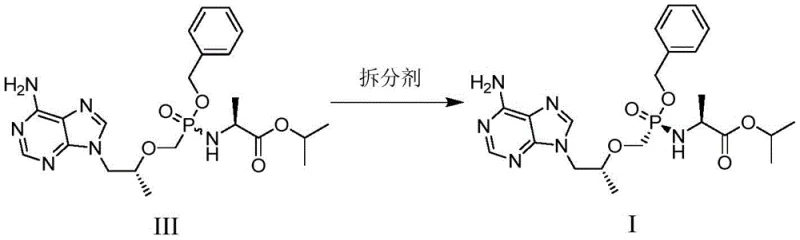
How to Synthesize Tenofovir Alafenamide Intermediate Efficiently
The practical implementation of this synthesis route requires careful attention to reaction conditions and reagent quality to ensure consistent results. The process begins with the condensation of tenofovir benzyl ester and L-alanine isopropyl ester hydrochloride in a suitable solvent, followed by the addition of the Carter condensing agent and a base. Temperature control is vital during the addition of the base to prevent exothermic spikes that could degrade the reagents. Once the reaction is complete, the workup involves a series of aqueous washes to remove inorganic salts and urea byproducts, followed by the critical recrystallization step. For the final resolution to Compound I, the selection of the resolving agent and solvent system must be optimized based on the specific stereochemical requirements. Detailed standardized synthesis steps see the guide below.
- Condense Tenofovir Benzyl Ester with L-alanine isopropyl ester hydrochloride using Carter condensing agent in a non-polar solvent under nitrogen protection.
- Purify the resulting Compound III crude product via recrystallization with organic acids in isopropanol to achieve high purity without column chromatography.
- Perform chiral resolution on Compound III using a resolving agent like D-tartaric acid in a refluxing solvent system to isolate the high-optical-purity Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for procurement and supply chain teams tasked with managing the costs and risks associated with antiviral drug production. The elimination of pyridine and column chromatography represents a significant reduction in the consumption of hazardous materials and high-purity solvents, which are often subject to price volatility and strict regulatory controls. By simplifying the purification process to a recrystallization workflow, manufacturers can reduce the cycle time for each batch, thereby increasing the overall capacity of existing facilities without the need for capital-intensive equipment upgrades. This efficiency gain translates directly into lower unit costs, allowing for more competitive pricing in the global market for reliable pharmaceutical intermediates supplier networks. Furthermore, the reduced environmental footprint of the process aligns with the increasing pressure from stakeholders for sustainable manufacturing practices, enhancing the corporate reputation of companies that adopt this technology.
- Cost Reduction in Manufacturing: The substitution of expensive and toxic reagents with the Carter condensing agent and benign solvents leads to substantial cost savings in raw material procurement. The removal of column chromatography eliminates the need for large quantities of silica gel and elution solvents, which are significant cost drivers in traditional purification. Additionally, the higher yield of the reaction means that less starting material is wasted, further improving the overall material efficiency of the process. These factors combine to create a leaner manufacturing model that is less susceptible to fluctuations in raw material prices and more resilient to supply chain disruptions.
- Enhanced Supply Chain Reliability: The robustness of the new synthesis route ensures a more consistent supply of high-quality intermediates, reducing the risk of production delays caused by purification failures or yield variations. The use of common, commercially available solvents and reagents minimizes the dependency on specialized suppliers, thereby diversifying the supply base and mitigating sourcing risks. This reliability is crucial for maintaining the continuity of drug production schedules, particularly for essential medications like those used in the treatment of chronic hepatitis B. A stable supply chain also allows for better inventory management and reduced safety stock levels, freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing unit operations that are standard in the fine chemical industry. The absence of complex chromatographic steps simplifies the technology transfer from laboratory to pilot and commercial scale, reducing the time and cost associated with process validation. Moreover, the reduced generation of hazardous waste simplifies compliance with environmental regulations, lowering the costs associated with waste treatment and disposal. This environmental compliance is increasingly becoming a key differentiator in the global market, where customers are prioritizing suppliers who demonstrate a commitment to sustainability and responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about technology adoption and process optimization.
Q: How does the Carter condensing agent improve the synthesis yield compared to traditional methods?
A: The Carter condensing agent replaces the traditional triphenylphosphine and 2,2-dithiodipyridine system, eliminating the need for toxic pyridine solvents. This change significantly reduces side reactions and simplifies post-reaction workup, leading to yields exceeding 80% for Compound III compared to approximately 64% in conventional processes.
Q: Is column chromatography required for purifying the intermediate Compound III?
A: No, the patented method explicitly avoids column chromatography. Instead, it utilizes a recrystallization technique involving organic acids such as malonic or fumaric acid in isopropanol. This approach not only achieves purity levels above 95% but also drastically reduces solvent consumption and processing time, making it highly suitable for industrial scale-up.
Q: What resolving agents are effective for achieving high chiral purity in Compound I?
A: The process supports various chiral resolving agents including D-tartaric acid, R-mandelic acid, and D-camphoric acid. When optimized, particularly with D-tartaric acid in specific solvent systems like 1,4-dioxane, the method achieves chiral purity levels exceeding 99.9%, ensuring the final prodrug meets stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Alafenamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of pharmaceutical manufacturing. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in patent CN111116655B can be seamlessly integrated into your supply chain. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing you with the confidence that every batch meets the highest standards of quality and consistency. Our dedication to technical excellence allows us to navigate the complexities of chiral resolution and amidation chemistry, delivering solutions that optimize both performance and cost.
We invite you to collaborate with us to explore how this advanced synthesis technology can enhance your production capabilities and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make data-driven decisions about your supply strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to supporting your long-term growth and success in the global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
