Revolutionizing Mannich Base Production: A Green, Aqueous Superacid Catalytic Route for Commercial Scale-Up
Revolutionizing Mannich Base Production: A Green, Aqueous Superacid Catalytic Route for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing critical nitrogen-containing intermediates. Patent CN101717320B introduces a groundbreaking methodology for the preparation of Mannich bases that fundamentally shifts the paradigm from traditional organic solvent-based systems to an environmentally benign aqueous medium. This innovation leverages liquid superacids as potent catalysts to drive a three-component, one-pot condensation reaction between aldehydes, amines, and ketones or naphthols. By operating under ambient pressure and room temperature conditions, this technology not only addresses the growing regulatory pressure to reduce volatile organic compound (VOC) emissions but also offers a streamlined operational protocol that significantly lowers the barrier to entry for high-volume manufacturing. For R&D directors and supply chain managers alike, this represents a pivotal opportunity to optimize both the purity profile of key intermediates and the overall cost structure of the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mannich bases has been plagued by significant operational inefficiencies and environmental drawbacks inherent to conventional methodologies. Traditional routes often necessitate the use of hazardous organic solvents such as dichloroethane, ethanol, or other volatile media, which impose heavy burdens on waste management systems and increase the risk of workplace exposure to toxic vapors. Furthermore, many established protocols require strict anhydrous conditions to prevent the hydrolysis of reactive intermediates, demanding energy-intensive drying processes and specialized equipment that drives up capital expenditure. In two-component approaches, the necessity to pre-form imines before reacting with ketones adds unnecessary synthetic steps, prolonging cycle times and accumulating yield losses at each stage. These cumulative inefficiencies result in higher production costs and a larger carbon footprint, making legacy methods increasingly untenable in a market that demands both economic and ecological responsibility.
The Novel Approach
In stark contrast, the methodology disclosed in CN101717320B offers a remarkably elegant solution by utilizing water as the sole reaction medium, effectively turning a traditional liability into a strategic asset. This novel approach employs liquid superacids, such as trifluoromethanesulfonic acid or fluorosulfonic acid, to activate the carbonyl and amine components even in the presence of water, a feat that was previously considered chemically challenging. The reaction proceeds efficiently at room temperature and atmospheric pressure, eliminating the need for heating or pressurized reactors, which drastically reduces energy consumption and enhances process safety. By consolidating the synthesis into a single pot where aldehydes, amines, and ketones are mixed directly, the process minimizes handling time and maximizes atom economy. This simplification not only accelerates the time-to-market for new drug candidates but also provides a robust platform for the reliable manufacture of pharmaceutical intermediates on a commercial scale.
Mechanistic Insights into Liquid Superacid-Catalyzed Condensation
The success of this aqueous synthesis relies heavily on the unique ability of liquid superacids to function as powerful proton donors that can overcome the deactivating effects of water. In the catalytic cycle, the superacid protonates the carbonyl oxygen of the aldehyde or the nitrogen of the amine, generating a highly electrophilic iminium ion intermediate that is resistant to hydrolysis under the specific reaction conditions employed. This activated species then undergoes nucleophilic attack by the enol or enolate form of the ketone (or the electron-rich position of the naphthol), leading to the formation of the C-C bond characteristic of the Mannich base. The high acidity ensures that the equilibrium is driven towards product formation despite the presence of water, which would typically quench such reactive intermediates in weaker acid systems. This mechanistic robustness allows for a broad substrate scope, accommodating various electronic environments on the aromatic rings without compromising reaction efficiency.
From an impurity control perspective, the mild reaction conditions play a crucial role in maintaining high product purity. Because the reaction occurs at room temperature, thermal degradation pathways and polymerization side reactions that are common in heated organic syntheses are effectively suppressed. The use of water as a solvent also facilitates the removal of polar impurities and excess reagents during the work-up phase, often allowing the product to precipitate directly or be easily crystallized from ethanol. This inherent selectivity reduces the burden on downstream purification processes, ensuring that the final API intermediate meets stringent quality specifications with minimal effort. The structural versatility of this method is evident in the wide range of substituents tolerated, as illustrated in the general reaction schemes below.
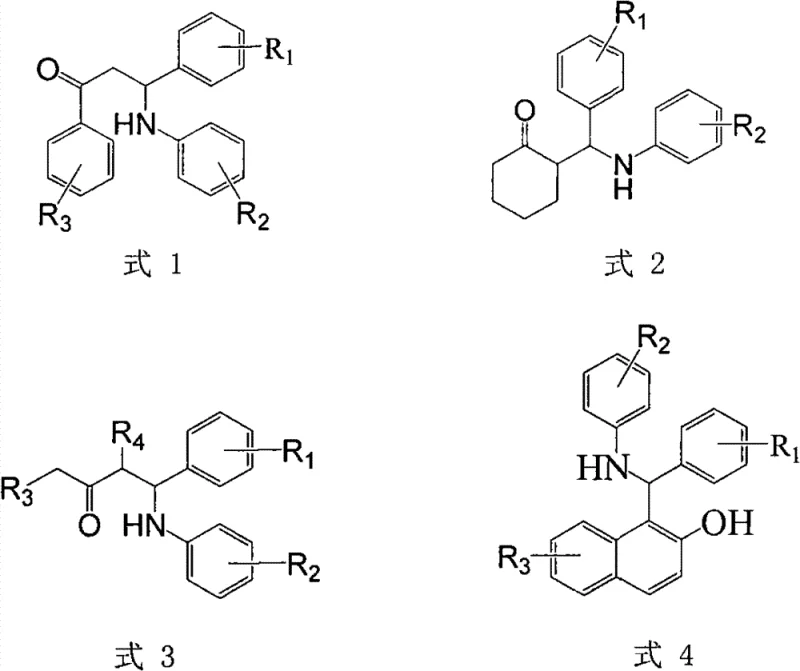
How to Synthesize Mannich Base Efficiently
The implementation of this synthesis route is designed for simplicity and reproducibility, making it an ideal candidate for technology transfer from the laboratory to the pilot plant. The process begins with the precise charging of the three key components—aldehyde, amine, and ketone or naphthol—into a standard reaction vessel equipped with a stirring mechanism. Water is added as the solvent, followed by the careful introduction of the liquid superacid catalyst, which initiates the reaction immediately upon mixing. The mixture is then stirred vigorously at ambient temperature for a duration ranging from 1 to 24 hours, depending on the specific reactivity of the substrates involved. Following the reaction completion, the crude product is isolated, typically through filtration or extraction, and purified via recrystallization using ethanol to achieve the desired high purity levels. Detailed standardized synthetic steps see the guide below.
- Charge a reaction vessel with aldehyde, amine, and ketone (or naphthol) in a molar ratio of 1: 1:1 to 1:1:3.
- Add water as the solvent and introduce a liquid superacid catalyst (such as trifluoromethanesulfonic acid) at 1mol% to 30mol% loading relative to the aldehyde.
- Stir the mixture vigorously at room temperature and atmospheric pressure for 1 to 24 hours, then isolate the product via recrystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous superacid-catalyzed process translates into tangible strategic benefits that extend far beyond simple chemical yield. The elimination of expensive and regulated organic solvents results in a drastic reduction in raw material procurement costs and simplifies the logistics of solvent storage and handling. Moreover, the one-pot nature of the reaction significantly shortens the manufacturing cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations. The mild operating conditions also reduce the wear and tear on reactor vessels and associated infrastructure, leading to lower maintenance costs and extended equipment lifecycles. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of sustaining long-term production volumes.
- Cost Reduction in Manufacturing: The substitution of organic solvents with water represents a fundamental shift in cost structure, removing the need for costly solvent recovery systems and reducing waste disposal fees associated with hazardous chemical waste. By utilizing inexpensive and readily available liquid superacids in catalytic amounts, the process minimizes the consumption of high-value reagents while maintaining high conversion rates. The simplified work-up procedure further reduces labor and utility costs, as there is no need for complex distillation or extensive washing steps to remove toxic residues. Consequently, the overall cost of goods sold (COGS) for these pharmaceutical intermediates is significantly lowered, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on water as a primary solvent mitigates the risks associated with the supply volatility of petrochemical-derived organic solvents, which are often subject to price spikes and availability constraints. Since the reaction does not require specialized anhydrous conditions or inert gas atmospheres, the manufacturing process is less susceptible to disruptions caused by utility failures or equipment malfunctions. The robustness of the chemistry ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to out-of-specification results. This reliability is critical for maintaining uninterrupted supply lines to downstream API manufacturers and ensuring continuity of care for patients.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method align perfectly with increasingly stringent global environmental regulations, facilitating easier permitting and compliance auditing for manufacturing facilities. The absence of VOC emissions and the generation of non-hazardous aqueous waste streams simplify the environmental impact assessment and reduce the burden on effluent treatment plants. From a scalability standpoint, the exothermic nature of the reaction is manageable at room temperature, allowing for safe scale-up from kilogram to multi-ton quantities without the need for complex cooling systems. This ease of scale-up ensures that the technology can meet growing commercial demands without compromising safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of using water as a solvent in Mannich base synthesis compared to traditional organic solvents?
A: Using water eliminates the need for toxic and expensive organic solvents like dichloroethane or ethanol, significantly reducing raw material costs and environmental disposal burdens. Furthermore, water simplifies the work-up process, often allowing for direct crystallization without complex extraction steps, thereby enhancing operational safety and throughput.
Q: Can this superacid-catalyzed method tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates excellent functional group tolerance. As detailed in the patent examples, substituents such as methyl, methoxy, chloro, nitro, and amino groups on the benzaldehyde or aniline rings are well-tolerated, yielding high-purity products without significant side reactions or degradation of sensitive moieties.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely. The process operates under mild conditions (room temperature and atmospheric pressure) and utilizes a one-pot strategy, which minimizes equipment complexity and energy consumption. These factors make it highly scalable for commercial production, offering a robust alternative to multi-step, anhydrous protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mannich Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous superacid-catalyzed synthesis route for producing high-quality Mannich bases. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch produced. We are committed to leveraging this green chemistry innovation to deliver superior value to our partners in the pharmaceutical and agrochemical sectors.
We invite you to collaborate with us to explore how this efficient synthesis method can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and timeline constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can support your journey from development to commercial success.
