Advanced Synthesis of Halofuginone Intermediates for Commercial Veterinary Drug Production
The global demand for effective anticoccidial agents in the poultry industry continues to drive innovation in veterinary pharmaceutical manufacturing. Patent CN103467449A introduces a groundbreaking methodology for the synthesis of halofuginone hydrobromide, a potent quinazolinone alkaloid known commercially as Stenorol. This patent details a novel piperidine derivative intermediate, represented by formula (VIII), which serves as a critical precursor in the production of this broad-spectrum antiparasitic drug. Unlike traditional pathways that suffer from low efficiency and hazardous conditions, this new approach leverages a highly selective copper-catalyzed epoxide ring-opening reaction. By utilizing accessible reagents such as cuprous iodide and tetramethylethylenediamine (TMEDA), the process achieves a remarkable total yield of 75.7% over seven steps, with individual step yields consistently exceeding 90%. This technological leap not only addresses the purity requirements demanded by regulatory bodies but also establishes a robust foundation for cost reduction in veterinary drug manufacturing by eliminating the need for precious metal catalysts.
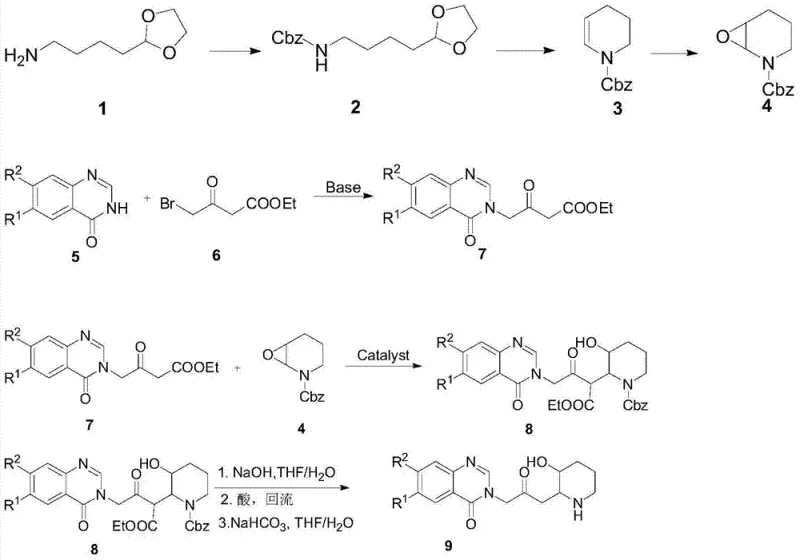
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halofuginone has been plagued by significant technical and economic barriers that hinder efficient commercial scale-up of complex piperidine derivatives. Early methodologies, such as the route reported by Baker BR, relied on high-temperature and high-pressure conversions of 2-acetylfuran, resulting in a dismal total yield of merely 2.1%. Furthermore, subsequent improvements described in patents like CN1651428A utilized 3-hydroxypyridine as a starting material but still achieved a total yield of only 3.7% after more than ten steps. These conventional routes frequently necessitate the use of flammable metallic lithium or prohibitively expensive rhodium catalysts under strictly anhydrous and oxygen-free conditions. Such requirements not only escalate equipment costs and energy consumption but also introduce substantial safety hazards, making industrial implementation difficult and economically unviable for many reliable veterinary pharmaceutical intermediates supplier candidates seeking to enter the market.
The Novel Approach
In stark contrast, the methodology disclosed in CN103467449A offers a streamlined and economically superior alternative that fundamentally reshapes the production landscape. The core innovation lies in the construction of the piperidine ring system via a highly selective reaction between a 1,3-dicarbonyl compound and an epoxy compound under mild alkaline conditions. This novel pathway effectively shortens the synthetic sequence by enabling a one-step reaction that simultaneously completes decarboxylation and the removal of the Cbz protecting group in the final stage. By avoiding the bottlenecks associated with transition metal scarcity and extreme reaction parameters, this approach ensures a consistent supply of high-purity halofuginone intermediates. The operational simplicity, characterized by standard extraction and recrystallization purification techniques rather than complex chromatography, significantly enhances throughput and reduces the overall environmental footprint of the manufacturing process.
Mechanistic Insights into CuI-TMEDA Catalyzed Epoxide Opening
The scientific elegance of this synthesis is rooted in the precise mechanistic control exerted by the copper catalytic system. As illustrated in the reaction mechanism, tetramethylethylenediamine (TMEDA) coordinates with cuprous iodide (CuI) to form a sterically hindered complex, designated as complex 10. This complex subsequently interacts with the N-Cbz-2,3-epoxypiperidine (compound 4) to induce the formation of an activated intermediate, complex 11. The steric bulk of this copper-ligand assembly is crucial, as it directs the nucleophilic attack of the 1,3-dicarbonyl enolate (generated from compound 7 under basic conditions) specifically to the epoxy carbon linked to the nitrogen atom. This regioselectivity is paramount for ensuring the correct stereochemistry and connectivity required for biological activity, effectively suppressing the formation of unwanted regioisomers that would otherwise complicate downstream purification.
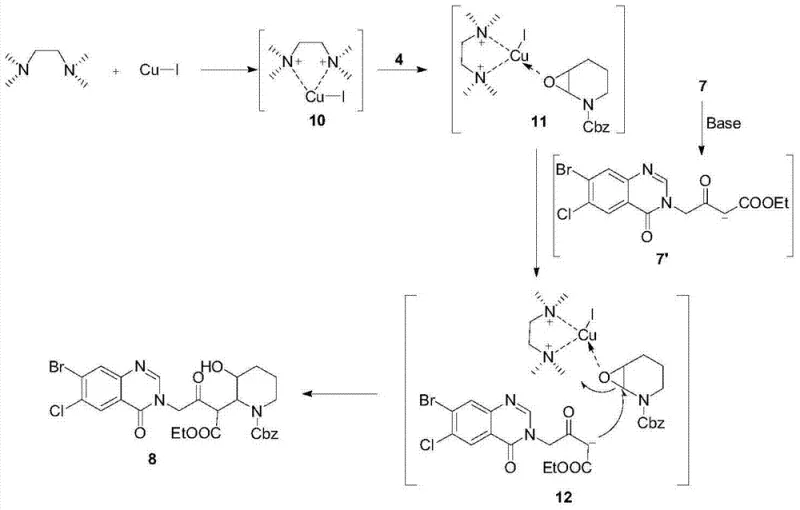
Furthermore, the presence of hexamethylphosphoramide (HMPA) plays a vital role in stabilizing the negative ion generated from the 1,3-dicarbonyl compound, thereby enhancing its nucleophilicity without compromising the selectivity imposed by the copper complex. The result is a reaction with selectivity approaching 100% and an isolated yield of 90.5% for the key coupling step. This level of control is essential for reducing lead time for high-purity anticoccidial agents, as it minimizes the generation of impurities that require extensive remediation. The mechanism ensures that the chiral integrity of the piperidine ring is maintained throughout the transformation, which is a critical quality attribute for the final active pharmaceutical ingredient. Understanding this catalytic cycle allows process chemists to fine-tune reaction parameters for optimal performance on a multi-ton scale.
How to Synthesize Halofuginone Intermediate Efficiently
The practical execution of this synthesis involves a logical sequence of transformations designed for maximum efficiency and safety. The process begins with the preparation of the epoxide building block from readily available 5-aminovaleraldehyde acetal, followed by the critical copper-catalyzed coupling with the quinazolinone fragment. The final step involves a tandem hydrolysis and decarboxylation to reveal the active piperidine moiety. This workflow is specifically engineered to minimize unit operations while maximizing yield, making it an ideal candidate for technology transfer. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which outlines the critical process controls.
- Prepare N-Cbz-2,3-epoxypiperidine (IV) from 5-aminovaleraldehyde acetal via Cbz protection, cyclization, and Oxone oxidation.
- Perform CuI/TMEDA/HMPA catalyzed coupling of compound (IV) with 1,3-dicarbonyl quinazolinone (VII) under alkaline conditions to form compound (VIII).
- Hydrolyze compound (VIII) with NaOH followed by acid reflux to simultaneously remove the Cbz group and decarboxylate, yielding the final halofuginone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented process offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. By shifting away from noble metal catalysts and hazardous reagents, manufacturers can achieve substantial cost savings and mitigate supply risks associated with volatile raw material markets. The robustness of the chemistry ensures consistent batch-to-batch quality, which is essential for maintaining regulatory compliance and avoiding costly production delays. Additionally, the simplified purification protocol reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium and reactive lithium with inexpensive cuprous iodide and organic ligands drastically lowers the direct material cost of goods sold. Furthermore, the high yield per step (>90%) means less raw material is wasted on failed reactions or lost during purification, leading to significant overall economic efficiency. The elimination of high-pressure and cryogenic equipment requirements also reduces capital expenditure and utility costs, making the process financially attractive for long-term production contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as THF, sodium methoxide, and common amines ensures that the supply chain is resilient against disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are globally available from multiple vendors. This diversity in sourcing options empowers procurement teams to negotiate better terms and secure continuous inventory flow, thereby guaranteeing uninterrupted production schedules for critical veterinary medicines.
- Scalability and Environmental Compliance: The mild reaction conditions (room temperature to moderate heating) and the absence of pyrophoric materials make this process inherently safer and easier to scale from pilot plant to commercial manufacturing. The simplified workup, which primarily involves extraction and crystallization rather than complex chromatographic separations, facilitates faster cycle times and higher throughput. Moreover, the reduced use of hazardous reagents simplifies waste treatment protocols, lowering the environmental compliance burden and associated disposal costs for the manufacturing facility.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the feasibility and benefits of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. They address key concerns regarding yield, safety, and scalability that are typically raised during the vendor qualification process.
Q: What are the advantages of the CuI-catalyzed route over traditional lithium or rhodium methods?
A: The CuI-catalyzed route avoids expensive and hazardous metals like lithium and rhodium, operates under mild conditions without high pressure, and achieves a significantly higher total yield of 75.7% compared to less than 4% in older methods.
Q: How does this process improve impurity control for halofuginone production?
A: The use of TMEDA and CuI creates a sterically hindered complex that ensures highly selective attack on the epoxide, resulting in selectivity close to 100% and minimizing side products that are difficult to remove.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process uses readily available catalysts, avoids extreme temperatures or pressures, and simplifies purification to extraction and recrystallization, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halofuginone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive veterinary pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN103467449A are successfully translated into robust industrial processes. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the highest standards required for downstream API synthesis. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this copper-mediated chemistry safely and efficiently.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio and drive value for your organization.
