Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN112125856A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing long-standing challenges in synthetic efficiency and operational safety. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing readily available o-iodoaniline and trifluoroethylimidoyl chloride as key building blocks. By replacing toxic gaseous carbon monoxide with a solid surrogate, this innovation not only streamlines the synthetic workflow but also significantly mitigates the safety risks associated with high-pressure gas handling, positioning it as a premier solution for reliable pharmaceutical intermediate supplier networks aiming for greener manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone scaffold, especially with specific trifluoromethyl substitution at the 2-position, has relied on methodologies that present substantial operational hurdles for large-scale manufacturing. Traditional routes often involve the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently require harsh reaction conditions and expensive, pre-activated substrates that drive up the cost of goods sold. Furthermore, alternative strategies utilizing isatoic anhydride or T3P-promoted tandem reactions often suffer from narrow substrate scope and inconsistent yields, limiting their utility in diverse drug discovery programs. Perhaps most critically, classical carbonylation approaches necessitate the direct use of carbon monoxide gas, a colorless and odorless toxin that demands specialized high-pressure equipment and rigorous safety protocols, thereby creating significant bottlenecks in cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
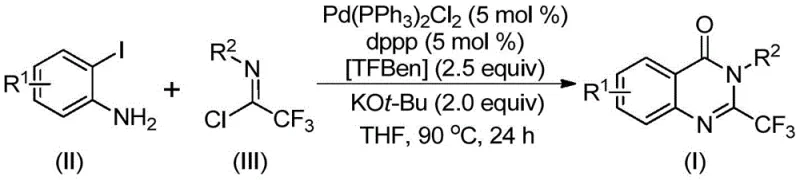
In stark contrast, the methodology disclosed in CN112125856A offers a transformative pathway by employing 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute. This strategic substitution allows the reaction to proceed under atmospheric pressure conditions while maintaining high efficiency, effectively bypassing the need for dangerous gas cylinders. The process demonstrates exceptional compatibility with various substituents on both the aniline and the imidoyl chloride components, enabling the synthesis of a wide array of derivatives without compromising yield. As illustrated in the general reaction scheme below, the convergence of these stable precursors under palladium catalysis provides a direct and atom-economical route to the target heterocycles.
The versatility of this approach is further evidenced by the successful synthesis of diverse derivatives, including those with electron-withdrawing and electron-donating groups, as well as bulky naphthyl substituents. This broad substrate tolerance ensures that medicinal chemists can rapidly access structural analogs for structure-activity relationship studies without being constrained by synthetic limitations. The use of common organic solvents like tetrahydrofuran (THF) further simplifies the workup procedure, facilitating easier isolation and purification of the final high-purity pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The efficacy of this synthesis relies on a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the o-iodoaniline substrate. Following this activation, the solid CO surrogate, TFBen, undergoes thermal decomposition to release carbon monoxide in situ, which subsequently inserts into the carbon-palladium bond to form a crucial acyl-palladium intermediate. This step is pivotal as it introduces the carbonyl functionality required for the quinazolinone ring closure without exposing the reactor to external CO gas. The presence of a strong base, such as potassium tert-butoxide, promotes the intramolecular nucleophilic attack by the nitrogen atom, leading to the formation of a seven-membered palladium intermediate before final reductive elimination releases the product and regenerates the active catalyst species.
Impurity control is inherently managed through the selectivity of the palladium catalyst and the mildness of the reaction conditions, which minimize side reactions such as homocoupling or over-carbonylation. The specific choice of ligands, particularly 1,3-bis(diphenylphosphine)propane (dppp), stabilizes the palladium center and directs the regioselectivity of the insertion steps, ensuring that the trifluoromethyl group remains intact at the 2-position of the quinazolinone ring. This mechanistic precision is essential for producing commercial scale-up of complex pharmaceutical intermediates where strict impurity profiles are mandated by regulatory bodies. The reaction typically proceeds at 90°C, a temperature that balances the kinetic energy required for TFBen decomposition with the thermal stability of the sensitive trifluoromethyl moiety.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and reaction parameters to maximize yield and purity. The process involves charging a reactor with the palladium catalyst, ligand, base, solid CO source, and substrates in an aprotic solvent, followed by heating to the specified temperature for a defined period. While the general procedure is straightforward, attention to detail regarding the quality of reagents and the exclusion of moisture is critical for reproducible results. For a comprehensive, step-by-step guide on executing this synthesis with specific quantities and workup procedures, please refer to the standardized protocol outlined below.
- Combine palladium catalyst Pd(PPh3)2Cl2, ligand dppp, base KOt-Bu, solid CO source TFBen, trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion of starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 2-trifluoromethyl quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers distinct advantages that translate directly into operational resilience and cost efficiency for chemical manufacturers. By shifting away from hazardous gaseous reagents to stable solid surrogates, companies can drastically simplify their safety compliance requirements and reduce the capital expenditure associated with specialized high-pressure reaction vessels. This transition not only lowers the barrier to entry for production but also enhances the overall reliability of the supply chain by minimizing the risk of shutdowns due to safety incidents or regulatory inspections related to toxic gas storage.
- Cost Reduction in Manufacturing: The utilization of cheap and commercially available starting materials, such as substituted o-iodoanilines and trifluoroethylimidoyl chlorides, significantly lowers the raw material costs compared to traditional methods requiring expensive activated esters or anhydrides. Furthermore, the elimination of toxic carbon monoxide gas removes the need for costly gas scrubbing systems and specialized containment infrastructure, leading to substantial overhead savings. The high conversion rates and clean reaction profiles observed in the patent data suggest that downstream purification costs can also be minimized, contributing to a more favorable overall economic model for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents like TFBen ensures that production schedules are not disrupted by the logistical challenges associated with transporting and storing compressed gases. This stability allows for more flexible inventory management and reduces the lead time for high-purity pharmaceutical intermediates, as manufacturers can maintain consistent stock levels of key precursors without expiration concerns. Additionally, the broad substrate scope means that a single production line can be adapted to synthesize multiple derivatives, increasing asset utilization and responsiveness to changing market demands.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process inherently safer and more environmentally friendly, aligning with modern green chemistry principles and stringent environmental regulations. Scaling this reaction from gram to kilogram or ton scale does not require exponential increases in safety measures, facilitating a smoother technology transfer from R&D to commercial manufacturing. The simplified post-treatment process, involving filtration and standard chromatography, further supports efficient waste management and reduces the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this method for industrial partners.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, effectively eliminating the need for handling toxic and hazardous carbon monoxide gas directly in the reactor.
Q: What represents the optimal catalytic system for this transformation?
A: The patent specifies that the combination of Pd(PPh3)2Cl2 as the catalyst and dppp (1,3-bis(diphenylphosphine)propane) as the ligand provides superior reaction efficiency and yield compared to other palladium sources.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses commercially available and inexpensive starting materials, operates at moderate temperatures (90°C), and avoids high-pressure gas equipment, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the global demand for fluorinated heterocycles continues to rise in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards for pharmaceutical applications.
We invite you to engage with our technical team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of adopting this safer, more efficient methodology. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
