Scalable Production of Trimethylhydroquinone via Novel Isophorone Oxidation for Global Vitamin E Supply Chains
Scalable Production of Trimethylhydroquinone via Novel Isophorone Oxidation for Global Vitamin E Supply Chains
The global demand for Vitamin E continues to drive innovation in the synthesis of its critical intermediates, specifically 2,3,5-Trimethylhydroquinone (TMHQ). Recent advancements in fine organic synthesis have introduced a robust preparation method detailed in patent CN108911954B, which leverages isophorone as a primary starting material. This technical breakthrough addresses long-standing challenges in the industry, such as low yield, complex purification, and significant environmental pollution associated with legacy processes. By oxidizing isophorone into oxoisophorone and subsequently undergoing acylation and hydrolysis, this novel route offers a streamlined pathway to high-purity TMHQ. For R&D directors and procurement specialists, understanding this shift is crucial for optimizing supply chains and ensuring the consistent quality required for pharmaceutical and nutraceutical applications. The method not only simplifies the reaction steps but also enhances the recoverability of solvents, marking a significant step forward in sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the artificial synthesis of trimethylhydroquinone has relied on routes such as m-cresol methylation or the 1,2,4-trimethylbenzene process, both of which present substantial technical and environmental hurdles. The m-cresol methylation method, while achieving high conversion rates, often utilizes catalysts like chromium nitrate and potassium nitrate, which are notoriously difficult to separate and recover from the final product matrix. This limitation frequently results in lower product purity and necessitates expensive downstream purification steps to meet stringent pharmaceutical standards. Furthermore, the 1,2,4-trimethylbenzene route involves a multi-step sequence including sulfonation, nitration, reduction, and oxidation, creating a long process flow that inherently accumulates impurities and generates significant hazardous waste. The use of sodium dichromate in the oxidation step of this traditional pathway introduces heavy metal contamination risks, complicating waste treatment and increasing the overall environmental footprint of the manufacturing facility.
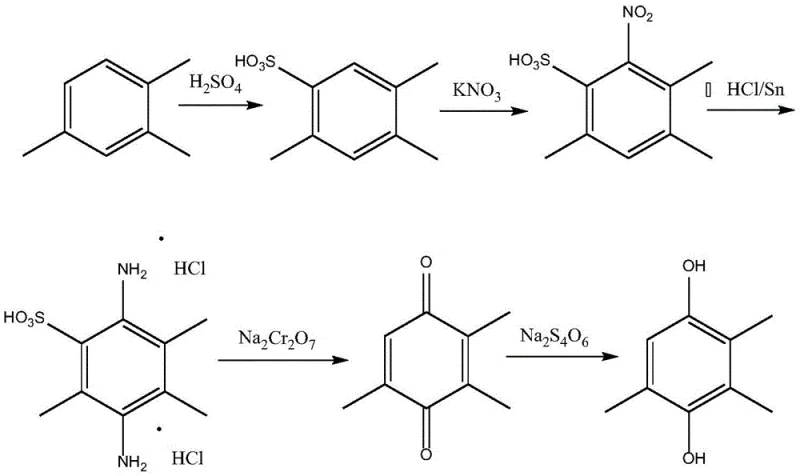
The Novel Approach
In stark contrast to these legacy methods, the novel isophorone-based synthesis route introduced in the patent data offers a dramatically simplified and cleaner alternative. By utilizing isophorone, a cheap and easily obtained raw material, the process bypasses the need for complex methylation or nitration steps entirely. The core innovation lies in the direct oxidation of isophorone to oxoisophorone using oxygen, followed by a controlled acylation reaction. This approach eliminates the reliance on toxic heavy metal oxidants and reduces the consumption of strong acids and alkalis, which are major contributors to industrial wastewater pollution. The streamlined nature of this synthesis not only improves the overall yield but also facilitates the recovery and reuse of organic solvents, thereby lowering the operational costs associated with raw material consumption. For supply chain managers, this translates to a more reliable and sustainable source of TMHQ that is less susceptible to regulatory changes regarding hazardous chemical handling.
Mechanistic Insights into Isophorone Oxidation and Acylation
The core of this technological advancement rests on a sophisticated catalytic system that enables the selective oxidation of isophorone under mild conditions. The reaction system employs a polar organic solvent, such as DMF or DMSO, in conjunction with an organic base and a catalytic amount of indole-3-acetic acid supplemented by an organic metal compound. Maintaining the reaction temperature at 20 +/- 5°C is critical for controlling the selectivity of the oxidation, ensuring that isophorone is converted almost completely into oxoisophorone (KIP) without significant over-oxidation or byproduct formation. The periodic supplementation of the catalyst during the reaction ensures sustained activity over the 4 to 8-hour duration, driving the conversion to completion. This precise control over the reaction environment minimizes the formation of impurities, which is a key concern for R&D directors focused on the impurity profile of API intermediates.
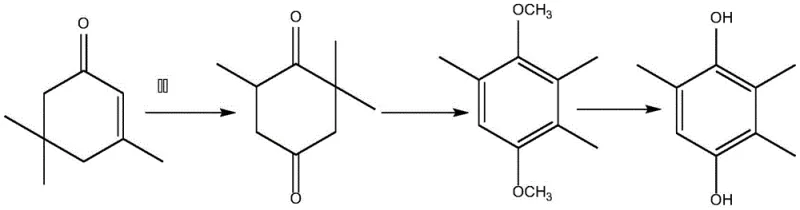
Following the oxidation step, the resulting oxoisophorone undergoes an acylation reaction with acetic anhydride in the presence of an inorganic strong acid at elevated temperatures of 80-85°C. This step generates the trimethylhydroquinone diester intermediate, which is then subjected to hydrolysis using aliphatic alcohol and hydrochloric acid. The hydrolysis conditions are carefully optimized to occur at 80-90°C under nitrogen protection, preventing oxidative degradation of the sensitive hydroquinone structure. The final crystallization step, conducted at low temperatures between -10°C and 5°C, ensures the precipitation of high-purity 2,3,5-trimethylhydroquinone crystals. This mechanistic sequence effectively isolates the target molecule from reaction byproducts, resulting in a product with excellent purity specifications suitable for high-value applications in the vitamin and fine chemical industries.
How to Synthesize 2,3,5-Trimethylhydroquinone Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and safety. The process begins with the preparation of the catalytic solution, followed by the controlled addition of isophorone under an oxygen atmosphere. Operators must monitor the temperature closely and supplement the catalyst at specific intervals to maintain reaction kinetics. Once the oxoisophorone is formed, the acylation and hydrolysis steps must be conducted with strict attention to stoichiometry and thermal conditions to prevent side reactions. The detailed standardized synthesis steps, including specific reagent quantities and timing for laboratory or pilot-scale replication, are provided in the technical guide below for qualified engineering teams.
- Oxidize isophorone using oxygen, an organic base, and indole-3-acetic acid catalyst in a polar solvent at 20 +/- 5°C to form oxoisophorone.
- React oxoisophorone with acetic anhydride and inorganic strong acid at 80-85°C to generate trimethylhydroquinone diester.
- Hydrolyze the diester using aliphatic alcohol and hydrochloric acid at 80-90°C, followed by crystallization to isolate pure 2,3,5-trimethylhydroquinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this isophorone-based synthesis route offers compelling advantages for procurement managers and supply chain heads focused on cost efficiency and reliability. The elimination of expensive and toxic heavy metal catalysts, such as chromium salts, removes the need for costly wastewater treatment processes and heavy metal clearance steps, leading to substantial cost savings in manufacturing operations. Additionally, the ability to recover and reuse organic solvents like DMF or ethanol significantly reduces the consumption of raw materials, further driving down the variable costs associated with production. The use of isophorone, a widely available commodity chemical, ensures a stable supply of starting materials, mitigating the risks associated with sourcing specialized or scarce reagents that often plague traditional synthesis routes.
- Cost Reduction in Manufacturing: The process design inherently lowers operational expenditures by simplifying the reaction sequence and reducing the load on waste treatment facilities. By avoiding the use of large amounts of strong acids and alkalis, the facility can operate with lower corrosion maintenance costs and reduced neutralization chemical requirements. The high conversion rate of isophorone to the target intermediate minimizes raw material waste, ensuring that a greater proportion of input costs are converted into saleable product value. These efficiencies collectively contribute to a more competitive cost structure for the final trimethylhydroquinone product.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials like isophorone and acetic anhydride ensures that production schedules are not disrupted by supply shortages of niche chemicals. The robustness of the reaction conditions, which do not require extreme pressures or temperatures, enhances equipment uptime and reduces the likelihood of unplanned maintenance shutdowns. This stability allows for consistent production output, enabling suppliers to meet long-term contractual obligations and maintain steady inventory levels for downstream Vitamin E manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this process highly amenable to scale-up from pilot plants to commercial tonnage production. The reduced generation of hazardous waste aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against regulatory tightening. This environmental compliance not only avoids potential fines but also enhances the brand reputation of the manufacturer as a sustainable partner in the global chemical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel TMHQ synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity for stakeholders evaluating this technology for adoption. Understanding these details is essential for making informed decisions regarding process integration and supplier selection.
Q: What are the primary advantages of the isophorone route over traditional m-cresol methylation?
A: The isophorone route eliminates the need for heavy metal catalysts like chromium nitrate, significantly reducing toxic waste generation and simplifying the purification process for higher product purity.
Q: How does the new method improve environmental compliance in TMHQ manufacturing?
A: By utilizing oxygen as the oxidant and minimizing the use of strong acids and alkalis during hydrolysis, the process drastically lowers wastewater toxicity and facilitates solvent recovery.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (20-90°C), and the raw materials like isophorone are commercially abundant, making the process highly scalable for commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethylhydroquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the production of essential nutrients like Vitamin E. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthesis routes like the isophorone oxidation method are translated into reliable industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of trimethylhydroquinone meets the exacting standards required by global pharmaceutical and nutraceutical clients. We are committed to delivering not just a product, but a comprehensive supply solution that integrates technical excellence with commercial reliability.
We invite procurement leaders and technical directors to engage with our team to explore how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this greener, more efficient manufacturing method. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your production requirements, ensuring a seamless transition to a more sustainable and cost-effective sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
