Advanced Catalytic Rearrangement for High-Purity Trimethylhydroquinone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust pathways for the production of critical Vitamin E precursors, specifically 2,3,5-Trimethylhydroquinone (TMHQ). A pivotal advancement in this domain is documented in patent CN1102138C, which outlines a superior method for rearranging 4-oxo-isophorone into TMHQ diesters followed by saponification. This technical breakthrough addresses long-standing inefficiencies in yield and reagent consumption that have plagued traditional manufacturing lines. For R&D Directors and Procurement Managers, understanding the nuances of this catalytic rearrangement is essential for optimizing supply chains and reducing the cost of goods sold (COGS). The process leverages specific protonic acid catalysts to drive the reaction with unprecedented efficiency, marking a significant shift from stoichiometric acid usage to true catalytic cycles. As a reliable pharmaceutical intermediates supplier, analyzing such proprietary data allows us to offer clients superior route feasibility assessments and scalable solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Trimethylhydroquinone has been hindered by processes that are economically and environmentally unsustainable. Early methods, such as the gas-phase rearrangement using zeolites described in DE26 46 172C2, suffered from critically low productivity rates ranging merely from 30% to 50%. Alternative liquid-phase approaches involving sulfuric acid in acetic anhydride solutions yielded even poorer results, often capping at around 31% efficiency, rendering them unsuitable for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the method detailed in DE-OS 2149159, while an improvement, still required excessive amounts of acetic anhydride, specifically 5 to 10 moles per mole of keto-isophorone, alongside a massive 150 mol% of acid catalyst. These stoichiometric excesses not only drive up raw material costs but also create significant waste disposal challenges, complicating the environmental compliance for any manufacturing facility. The maximum yield achievable with these legacy methods hovers around 66%, leaving a substantial portion of valuable starting material unconverted or degraded into impurities. For supply chain heads, these inefficiencies translate into volatile pricing and unreliable lead times for high-purity intermediates.
The Novel Approach
The innovative method disclosed in the patent data introduces a paradigm shift by utilizing a catalytic amount of protonic acid rather than stoichiometric quantities. By employing strong protonic acids such as trifluoromethanesulfonic acid, chlorosulfonic acid, or oleum at concentrations between 0.1-50wt%, the reaction kinetics are significantly enhanced without the burden of excessive reagent load. This approach reduces the required acetic anhydride to a much more economical range of greater than 2 to 4 moles per mole of substrate, effectively halving the chemical consumption compared to prior art. The result is a dramatic improvement in yield, with the new process consistently achieving between 85% and 90% conversion efficiency. This leap in performance is not merely incremental; it represents a fundamental optimization of the reaction thermodynamics and kinetics, allowing for cost reduction in pharmaceutical intermediates manufacturing. The ability to operate with lower acid loads also simplifies the downstream workup, reducing the corrosion burden on equipment and minimizing the neutralization waste streams. Consequently, this method offers a viable pathway for the commercial scale-up of complex organic synthesis that aligns with modern green chemistry principles.
Mechanistic Insights into Acid-Catalyzed Rearrangement
The core of this technological advancement lies in the precise mechanism of the acid-catalyzed rearrangement of keto-isophorone. The reaction initiates with the protonation of the carbonyl oxygen in the 4-oxo-isophorone structure, activating the molecule for a skeletal rearrangement that forms the aromatic ring system. The presence of the acetylating agent, typically acetic anhydride, traps the intermediate enol forms immediately, driving the equilibrium towards the formation of the stable Trimethylhydroquinone diacetate ester. This in-situ protection is crucial, as it prevents the formation of unwanted oxidation byproducts and ensures that the aromatic system is locked in place before the final hydrolysis step. The choice of catalyst is paramount; strong protonic acids like trifluoromethanesulfonic acid provide the necessary acidity to facilitate the rearrangement at moderate temperatures between 25-70°C. This controlled thermal profile prevents the thermal degradation often seen in harsher, high-temperature processes, thereby preserving the integrity of the molecular structure. Understanding this mechanism allows R&D teams to fine-tune reaction parameters for maximum purity and minimal impurity generation.
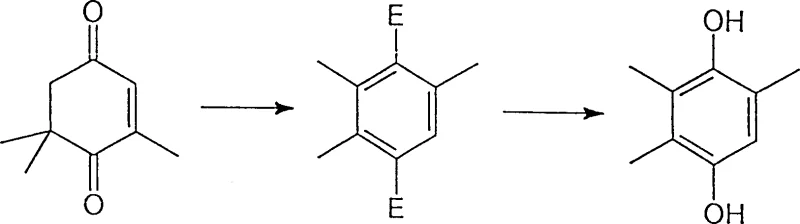
Following the rearrangement, the saponification step is engineered to maximize recovery while minimizing solvent usage. The patent describes a 'one-pot' methodology where water or diluted acid is added directly to the reaction mixture to hydrolyze the diester without prior isolation. This is often facilitated by the addition of phase transfer agents or co-solvents like propyl carbinol or n-butyl acetate, which improve the miscibility of the organic ester and the aqueous hydrolysis medium. The hydrolysis is conducted under boiling conditions, typically using 30% sulfuric acid, which ensures complete cleavage of the acetate groups to reveal the free phenolic hydroxyl groups of the final TMHQ product. The control of impurities during this phase is critical, as residual acids or incomplete hydrolysis can affect the purity profile required for Vitamin E synthesis. By optimizing the phase blender and acid concentration, the process ensures that the final product precipitates cleanly, allowing for easy filtration and washing. This mechanistic control over the hydrolysis step is what guarantees the high purity specifications demanded by downstream applications in the nutraceutical and pharmaceutical sectors.
How to Synthesize Trimethylhydroquinone Efficiently
Implementing this synthesis route requires careful attention to the addition rates and thermal management during the rearrangement phase. The standard protocol involves dripping keto-isophorone into a pre-cooled mixture of acetic anhydride and the selected protonic acid catalyst over a period of 1 to 3 hours. Maintaining the temperature between 0-60°C during this addition is critical to control the exotherm and prevent side reactions. Once the addition is complete, the mixture is heated to 25-70°C and held for 1-7 hours to ensure full conversion to the diester intermediate. The detailed standardized synthesis steps, including specific workup procedures and isolation techniques, are outlined in the guide below for technical reference. Adhering to these parameters ensures that the theoretical yields of 95% for the diester and 89-90% for the final TMHQ are achievable in a production environment.
- Prepare the reaction mixture by adding keto-isophorone to a mixture of acetic anhydride and a catalytic amount of protonic acid (0.1-50wt%) at 0-60°C.
- Heat the mixture to 25-70°C for 1-7 hours to complete the rearrangement into Trimethylhydroquinone diacetate.
- Hydrolyze the diester by adding water or diluted sulfuric acid, heat to boiling, and isolate the final Trimethylhydroquinone product via filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic advantages beyond mere chemical yield. The reduction in reagent stoichiometry directly correlates to a significant decrease in raw material procurement costs, as less acetic anhydride and acid catalyst are required per kilogram of finished product. This efficiency gain is compounded by the simplification of the waste treatment process, as the lower acid load reduces the volume of neutralization salts and aqueous waste that must be processed. Furthermore, the higher yield means that less starting material is wasted, effectively increasing the throughput of existing reactor capacity without the need for capital expenditure on new equipment. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations in raw material pricing. The robustness of the process also reduces the risk of batch failures, ensuring a consistent and reliable supply of high-purity intermediates for downstream Vitamin E production.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in the molar ratio of acetic anhydride required. By lowering the requirement from 5-10 moles in conventional methods to just 2-4 moles, the direct material cost is significantly curtailed. Additionally, the shift from stoichiometric acid usage to a catalytic regime (0.1-50wt%) eliminates the need for purchasing and handling massive quantities of corrosive acids. This reduction in chemical consumption also lowers the operational costs associated with solvent recovery and waste disposal, as there is less organic and aqueous waste to treat. The cumulative effect of these efficiencies results in a markedly lower cost of goods sold, providing a competitive edge in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The simplified reaction profile and reduced dependency on excessive reagents enhance the overall reliability of the supply chain. With fewer raw materials to source and manage, the risk of supply disruptions due to vendor issues is minimized. The higher yield and purity of the process mean that production schedules are more predictable, reducing the likelihood of delays caused by the need for re-processing or purification. This stability is crucial for long-term supply agreements with major pharmaceutical and nutraceutical companies that require just-in-time delivery. Moreover, the use of common and readily available catalysts like oleum or chlorosulfonic acid ensures that the supply of critical reagents remains secure, further stabilizing the production timeline.
- Scalability and Environmental Compliance: From a scalability perspective, the process is designed to transition smoothly from laboratory scale to multi-ton commercial production. The exothermic nature of the reaction is manageable through controlled addition rates, making it safe for large-scale reactors. Environmentally, the reduction in waste generation aligns with increasingly stringent global regulations on industrial effluents. By generating less acidic waste and consuming less solvent, the facility's environmental footprint is significantly reduced, easing the burden on wastewater treatment plants. This compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and customers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Trimethylhydroquinone synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance. Understanding these details is vital for technical teams evaluating the feasibility of adopting this method for their own production lines. The insights provided here cover catalyst selection, yield expectations, and process safety considerations.
Q: What is the primary yield advantage of this new rearrangement method?
A: Unlike conventional methods yielding only 30-66%, this catalytic process achieves yields between 85-90%, significantly reducing raw material waste.
Q: Which catalysts are preferred for the rearrangement of keto-isophorone?
A: The process utilizes protonic acids such as trifluoromethanesulfonic acid, chlorosulfonic acid, or oleum at concentrations between 0.5-25wt% for optimal efficiency.
Q: How does this method improve environmental compliance compared to prior art?
A: By reducing the required molar ratio of acetic anhydride from 5-10mol to just 2-4mol per mole of substrate, the process drastically lowers chemical waste and solvent load.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethylhydroquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply chain for Vitamin E and related nutraceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Trimethylhydroquinone meets the exacting standards required by our partners. Our infrastructure is designed to handle complex catalytic rearrangements safely and efficiently, leveraging the latest process optimization techniques to maximize yield and minimize waste. By partnering with us, clients gain access to a supply chain that is both robust and responsive to market demands.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply strategy. We offer a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this catalytic method for your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments tailored to your project needs. Our goal is to provide not just a product, but a comprehensive solution that enhances your operational efficiency and competitive positioning in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
